Cell metabolism — WJEC GCSE Study Guide
Exam Board: WJEC | Level: GCSE
Master the fundamental processes that keep cells alive: how enzymes control reactions, how respiration releases energy, and how digestion breaks down food. This topic is heavily tested in exams, particularly linking enzyme activity to temperature and pH graphs.

## Overview
Cell metabolism encompasses all the chemical reactions that occur within a living organism to maintain life. In this topic, we focus on three vital, interconnected processes: the action of enzymes, the release of energy through respiration, and the breakdown of macromolecules during digestion.
Understanding cell metabolism is crucial because it forms the foundation for almost every other topic in biology. Without enzymes, reactions would happen too slowly to sustain life. Without respiration, cells would have no energy to function. Without digestion, the raw materials needed for respiration and growth could never enter our bloodstream.
Examiners love testing this topic because it allows them to assess your understanding of core biological principles alongside your data analysis skills. You can expect questions requiring you to interpret graphs of enzyme activity, compare aerobic and anaerobic respiration, and explain the specific roles of different digestive enzymes. Mastering the precise terminology—such as 'denatured', 'active site', and 'exothermic'—is the key to unlocking the top mark bands.
## Key Concepts
### Concept 1: Enzymes as Biological Catalysts
Enzymes are biological catalysts made of protein. A catalyst is a substance that speeds up a chemical reaction without being changed or used up in the process. Because they are proteins, enzymes are composed of long chains of amino acids folded into specific 3D shapes.
The most important part of an enzyme is its **active site**. This is a uniquely shaped 'pocket' on the enzyme's surface. The molecule that the enzyme acts upon is called the **substrate**.
This relationship is explained by the **Lock and Key Hypothesis**. Just as a specific key only fits a specific lock, only a specific substrate will fit into an enzyme's active site. When they bind together, they form an **enzyme-substrate complex**. The reaction occurs rapidly, the products are released, and the enzyme is left completely unchanged, ready to catalyse another reaction immediately.
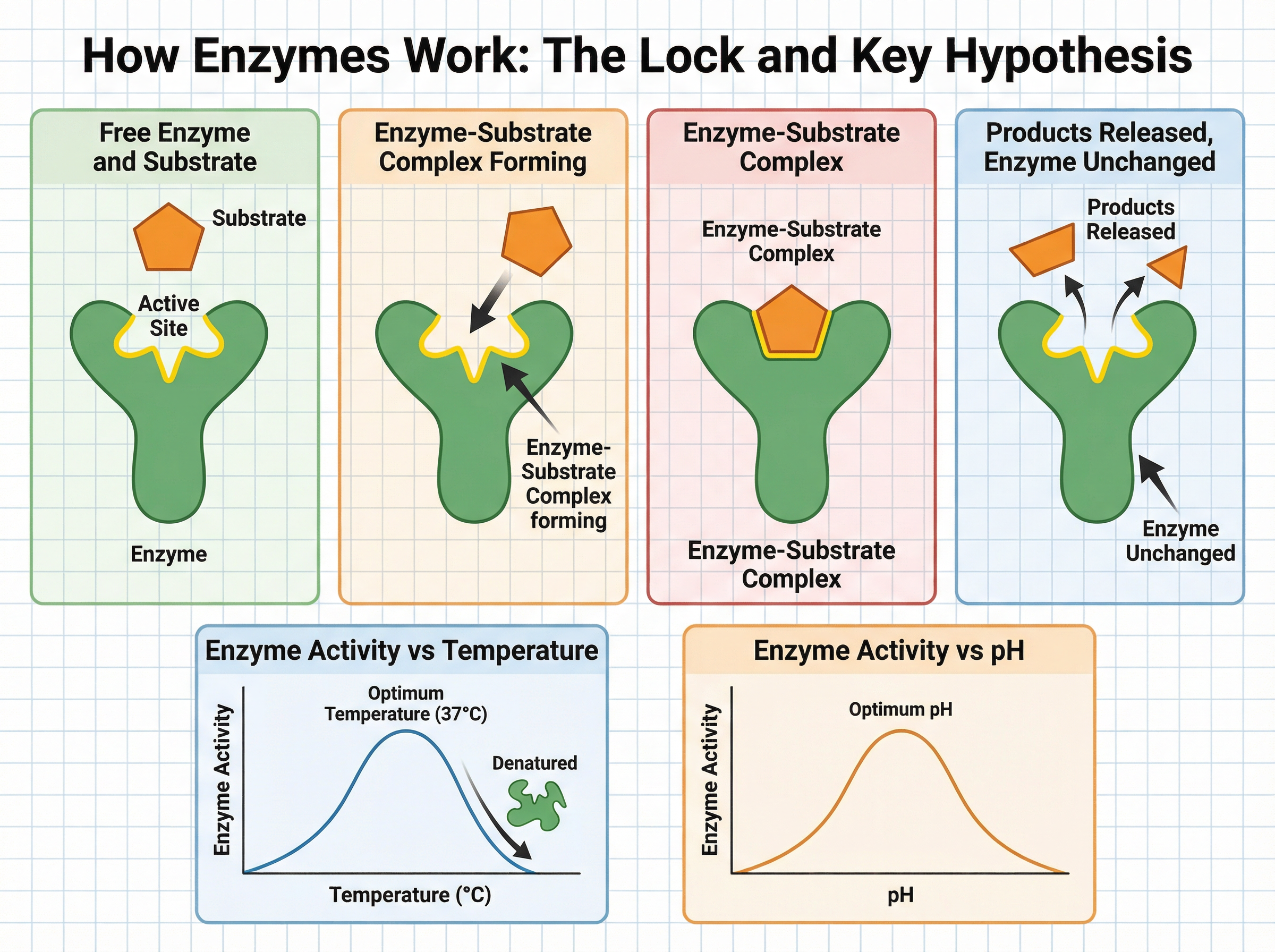
**Example**: Hydrogen peroxide is a toxic byproduct of metabolism. The enzyme catalase, found in liver cells, rapidly breaks it down into harmless water and oxygen. Without catalase, the hydrogen peroxide would build up to lethal levels.
### Concept 2: The Effect of Temperature and pH on Enzymes
Enzymes are highly sensitive to their environment. Every enzyme has an **optimum temperature** and an **optimum pH**—the specific conditions under which it works at its maximum rate.
In humans, the optimum temperature for most enzymes is around 37°C (normal body temperature). As temperature increases towards the optimum, the kinetic energy of the molecules increases, leading to more frequent successful collisions between enzymes and substrates.
However, if the temperature rises *above* the optimum, the strong vibrations break the chemical bonds holding the protein in its specific shape. The active site changes shape, and the substrate can no longer fit. The enzyme has been **denatured**.
*Examiner Tip: Never say an enzyme is 'killed' or 'dies'. Enzymes are molecules, not living things. The only acceptable term is 'denatured'.*
Similarly, if the pH moves too far from the optimum (either too acidic or too alkaline), it interferes with the bonds in the protein, altering the shape of the active site and denaturing the enzyme.
### Concept 3: Aerobic and Anaerobic Respiration
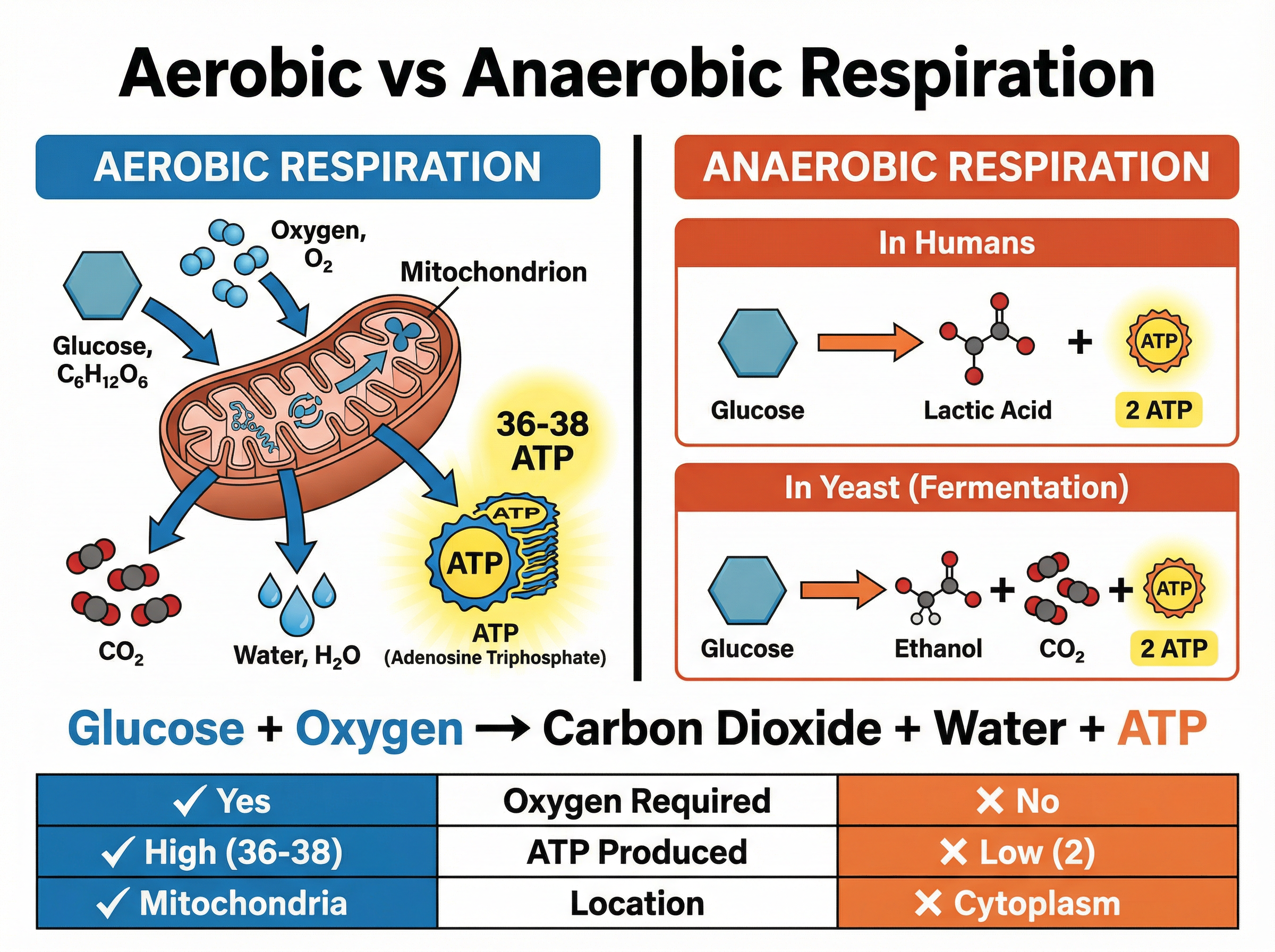
Respiration is the chemical process by which cells release energy from glucose. It occurs continuously in all living cells. It is an **exothermic** reaction, meaning it transfers energy to the environment. This energy is used to synthesise molecules of **ATP** (adenosine triphosphate), the 'energy currency' of the cell.
There are two main types of respiration:
**1. Aerobic Respiration**
This requires oxygen and is highly efficient, releasing a large amount of ATP. It occurs primarily in the mitochondria.
**2. Anaerobic Respiration**
This occurs when oxygen is absent or in short supply. It is much less efficient, producing far less ATP per glucose molecule, and it occurs in the cytoplasm.
The products of anaerobic respiration depend on the organism:
- **In animals (humans):** Glucose is broken down into **lactic acid**. The build-up of lactic acid in muscles during vigorous exercise causes fatigue and an 'oxygen debt'.
- **In plants and yeast:** Glucose is broken down into **ethanol** and **carbon dioxide**. In yeast, this process is called **fermentation** and is economically important in the brewing and baking industries.
### Concept 4: Digestion of Macromolecules
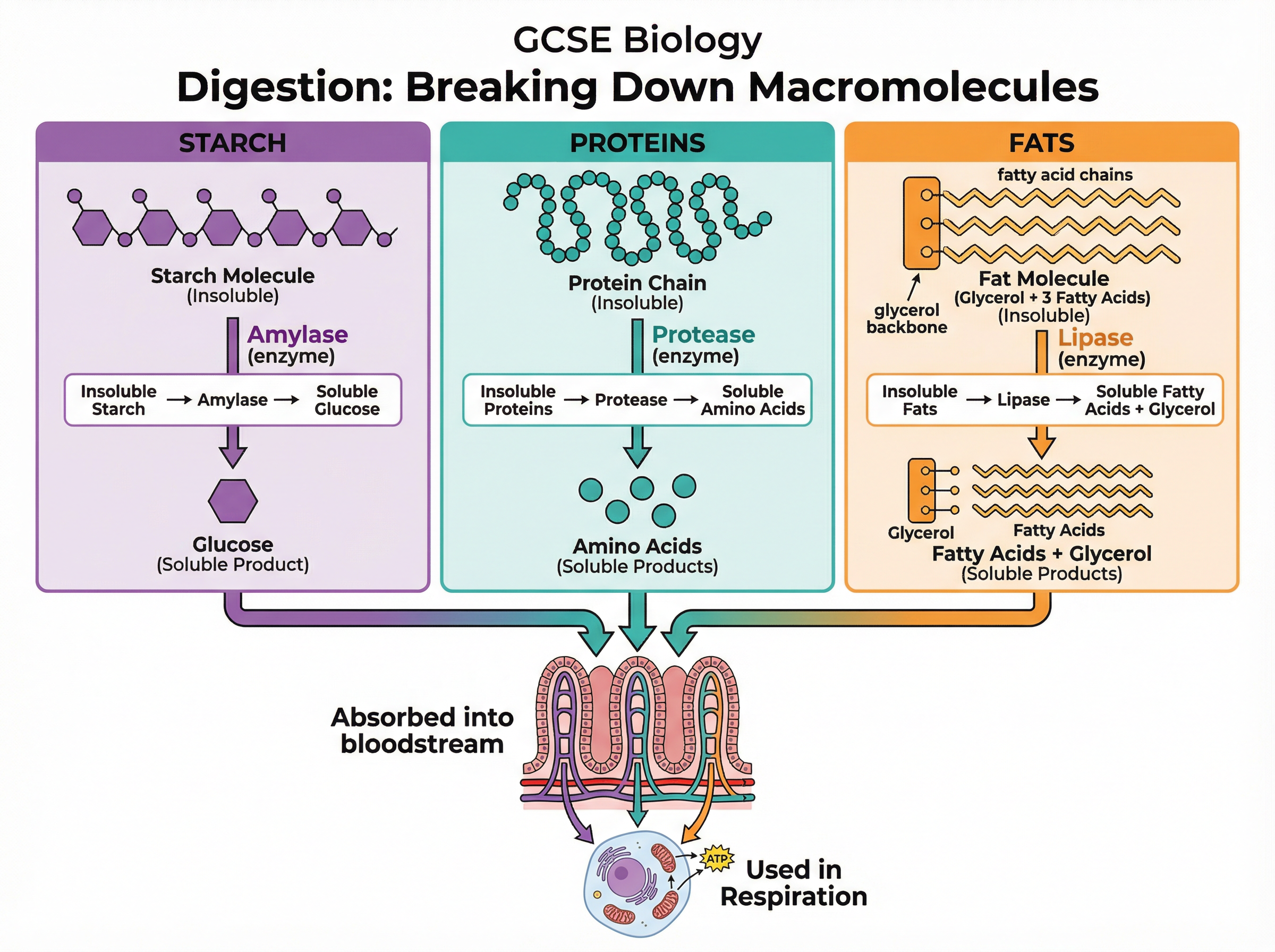
Digestion is the process of breaking down large, insoluble food molecules into small, soluble molecules that can be absorbed into the bloodstream through the wall of the small intestine.
This process relies entirely on specific digestive enzymes:
1. **Carbohydrases (e.g., Amylase):** Break down carbohydrates like starch into simple sugars like glucose. Amylase is produced in the salivary glands and the pancreas.
2. **Proteases:** Break down proteins into amino acids. They are produced in the stomach, pancreas, and small intestine.
3. **Lipases:** Break down lipids (fats and oils) into fatty acids and glycerol. They are produced in the pancreas and small intestine.
Once absorbed, these small soluble molecules (like glucose and amino acids) are transported to cells where they are used for respiration or to build new complex molecules for growth and repair.
---
## Podcast Episode
Listen to our 10-minute deep dive into Cell Metabolism, covering all the core concepts and exam techniques you need to know.

## Mathematical/Scientific Relationships
**Word Equation for Aerobic Respiration:**
Glucose + Oxygen → Carbon Dioxide + Water (+ ATP)
**Symbol Equation for Aerobic Respiration (Must Memorise):**
C₆H₁₂O₆ + 6O₂ → 6CO₂ + 6H₂O
**Word Equation for Anaerobic Respiration (Animals):**
Glucose → Lactic Acid (+ minimal ATP)
**Word Equation for Anaerobic Respiration (Plants/Yeast):**
Glucose → Ethanol + Carbon Dioxide (+ minimal ATP)
**Rate of Reaction Calculation:**
Rate = 1000 / time (or Volume of product produced / time taken)
*Use this when calculating enzyme activity rates from practical data.*
## Practical Applications
**Required Practical: Investigating the effect of pH on the rate of reaction of amylase.**
This practical involves using iodine solution to test for the presence of starch. Iodine turns from orange-brown to blue-black if starch is present. By mixing amylase and starch at different pH levels and testing droplets every 30 seconds, you can determine how long it takes for the starch to be fully broken down. The fastest breakdown time indicates the optimum pH.
*Common Examiner Focus:* They will ask why you must use a water bath (to control temperature as a control variable) and why taking samples every 30 seconds might lead to inaccurate rate calculations (it's an estimate; continuous monitoring or shorter intervals would be more precise).