The principle of material cycling — WJEC GCSE Study Guide
Exam Board: WJEC | Level: GCSE
Master the carbon cycle and the crucial role of decomposers in returning nutrients to the ecosystem. This topic is heavily tested, particularly on how environmental factors like temperature and water content affect the rate of decay.
## Overview
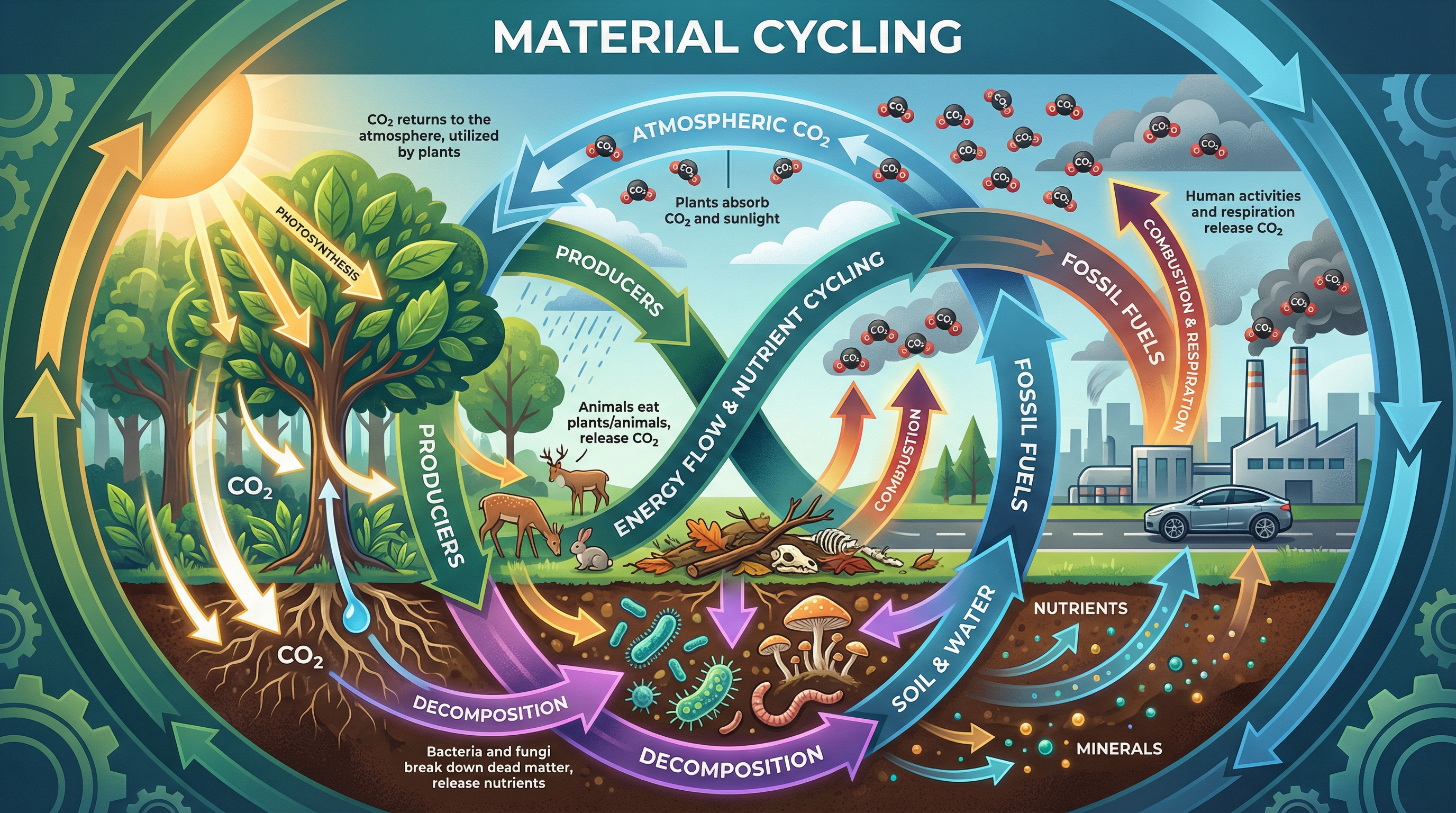
The principle of material cycling is a foundational concept in Biology. In any stable ecosystem, the materials removed from the environment by living organisms must eventually be returned. If they weren't, the ecosystem would run out of essential nutrients like carbon, nitrogen, and water, leading to total collapse.
This topic focuses heavily on the carbon cycle and the vital role of decomposers (bacteria and fungi). You will need to understand how carbon moves between the atmosphere, living organisms, and the soil, and how environmental factors influence the rate at which dead matter is broken down.
Examiners love to test this topic through extended six-mark questions asking you to describe the carbon cycle, or through data analysis questions where you must explain how changes in temperature or moisture affect the rate of decomposition. Mastering this topic requires precision in your language—especially regarding the role of respiration in returning carbon dioxide to the atmosphere.

## Key Concepts
### Concept 1: The Carbon Cycle
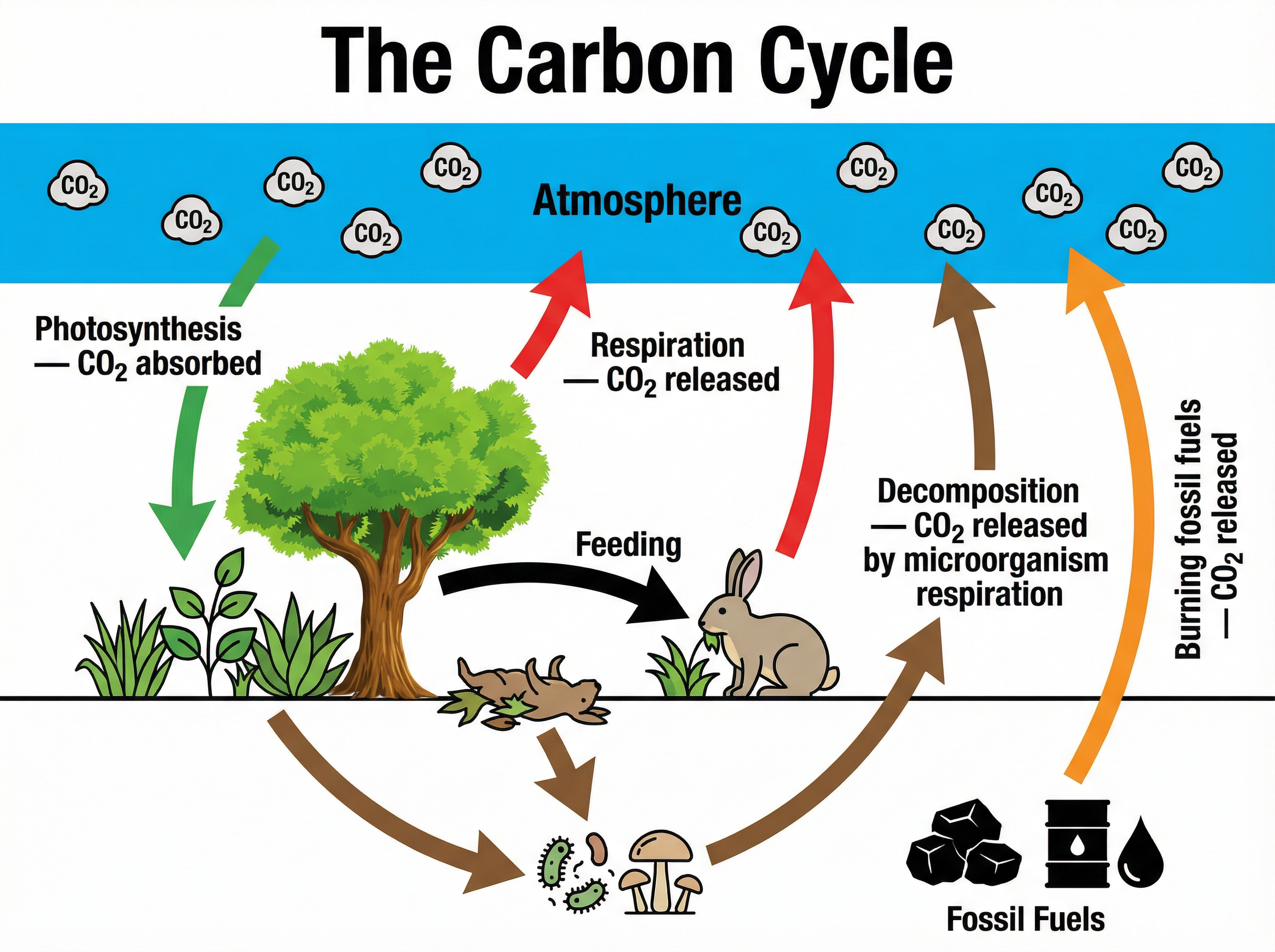
The carbon cycle describes how carbon moves between the abiotic (non-living) environment and biotic (living) organisms. Carbon is the backbone of all organic molecules, including carbohydrates, proteins, and lipids.
**Incorporation:** Carbon enters the biotic world exclusively through **photosynthesis**. Green plants and algae absorb carbon dioxide from the atmosphere and use light energy to convert it into glucose. This carbon is then passed along food chains as animals eat plants, and predators eat other animals.
**Return:** Carbon is returned to the atmosphere through three main processes:
1. **Respiration:** All living organisms (plants, animals, and microorganisms) respire to release energy from glucose, releasing carbon dioxide as a waste product.
2. **Decomposition:** When organisms die, or produce waste, decomposers break down the organic matter. Crucially, the decomposers respire as they do this, releasing carbon dioxide.
3. **Combustion:** The burning of fossil fuels and biomass releases locked-away carbon back into the atmosphere as carbon dioxide.
### Concept 2: The Role of Decomposers
Decomposers are microorganisms, specifically **bacteria and fungi**. They are essential for nutrient cycling. They secrete enzymes onto dead organic matter, breaking down complex molecules into simpler, soluble ones which they then absorb.
As decomposers carry out this process, they release inorganic nutrients (like nitrates and phosphates) into the soil, which can be taken up by plant roots. Simultaneously, they respire, returning carbon dioxide to the atmosphere.
**Examiner Tip:** Never just say "decomposers release carbon." Always state that "decomposers release carbon dioxide through respiration."
### Concept 3: Factors Affecting the Rate of Decomposition
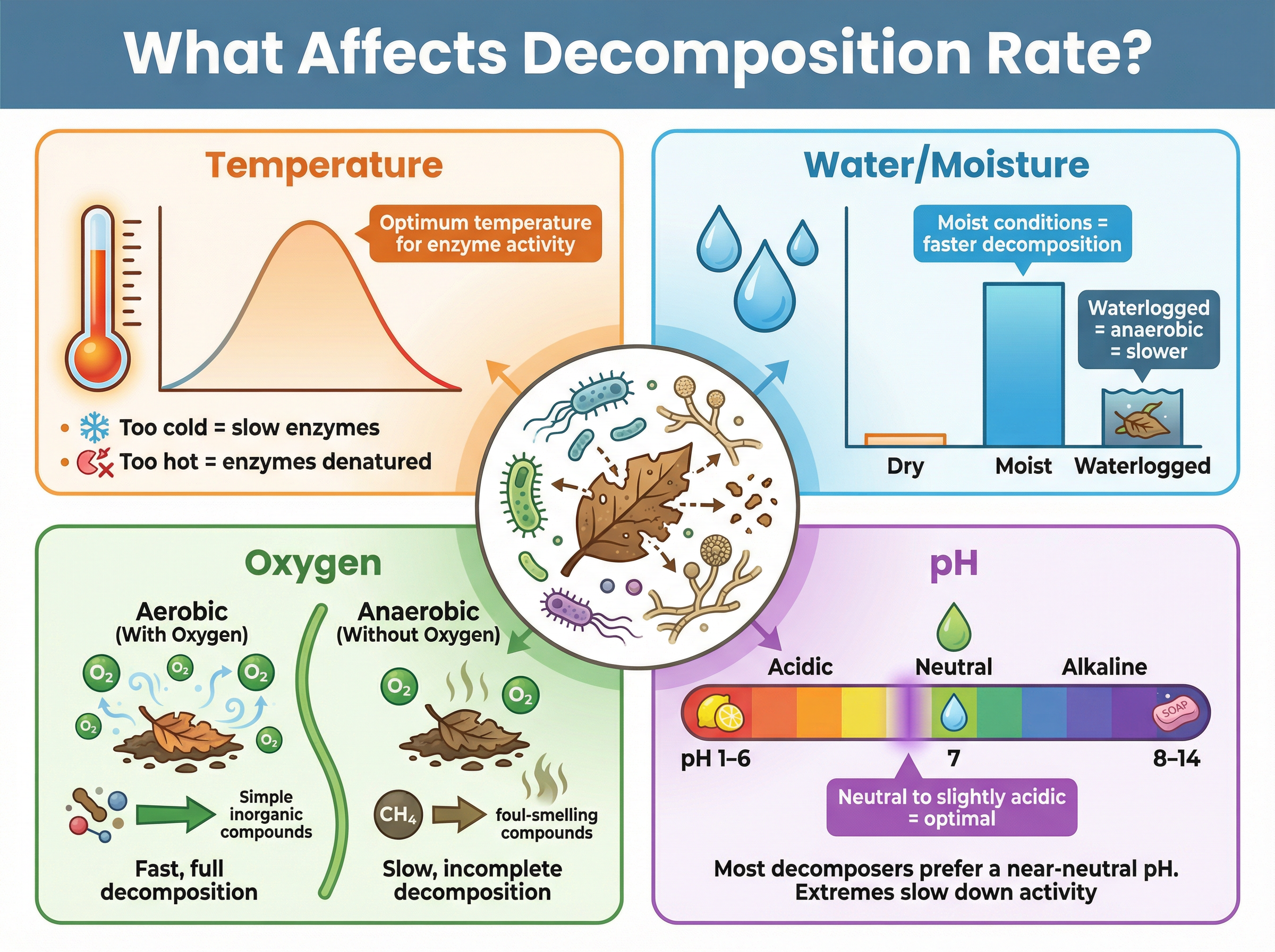
The rate at which decomposition occurs depends heavily on environmental conditions, because decomposition is an enzyme-controlled process carried out by living organisms.
1. **Temperature:** At low temperatures, enzyme activity is slow, so decomposition is slow. As temperature increases to the optimum (usually 30-40°C), enzyme activity peaks and decomposition is rapid. Above the optimum, enzymes denature, and decomposition stops.
2. **Water Content:** Decomposers need moisture to survive and for their enzymes to work. Dry conditions severely slow decomposition. However, waterlogged conditions also slow it down because water displaces oxygen in the soil, creating anaerobic conditions.
3. **Oxygen Availability:** Most decomposers are aerobic. In well-aerated soils, aerobic decomposition is fast and produces carbon dioxide. In anaerobic conditions (like peat bogs), decomposition is slow and produces methane instead.
## Mathematical/Scientific Relationships
While there are no complex mathematical formulas to memorize for this specific section, you must understand the relationship between rate and time.
**Rate of Decay = Change in mass / Time**
If a question provides data on the mass of a decaying compost heap over time, you may need to calculate the rate. Remember that a higher temperature (up to the optimum) will result in a larger change in mass over the same time period.
## Practical Applications
**Compost Heaps:** Gardeners optimize the conditions for decay to produce compost quickly. They keep the heap moist, aerate it regularly with a pitchfork (providing oxygen for aerobic respiration), and the metabolic heat generated by the decomposers keeps the heap warm, maintaining a high rate of enzyme activity.
**Biogas Generators:** In anaerobic conditions, decomposers produce methane gas. Biogas generators deliberately create anaerobic conditions to break down plant and animal waste, capturing the methane produced to use as a renewable fuel for heating or cooking.