Bonding, structure, and the properties of matter — AQA GCSE Study Guide
Exam Board: AQA | Level: GCSE
Master the fundamentals of how atoms bond and arrange themselves to form everything around us. This topic is a cornerstone of GCSE Chemistry, essential for explaining why materials behave the way they do and unlocking high marks across your exams.
## Overview
Welcome to Topic 4.2: Bonding, Structure, and the Properties of Matter. This is arguably the most foundational topic in all of GCSE Chemistry. It explains why a diamond can cut through rock while graphite in your pencil easily flakes off onto paper, even though both are made entirely of carbon atoms.
Understanding bonding is critical because examiners will constantly ask you to explain the physical properties of substances—like melting points or electrical conductivity. To secure those marks, you must be able to identify the type of bonding (ionic, covalent, or metallic) and the resulting structure (giant lattice or small molecules). This topic connects heavily to quantitative chemistry and organic chemistry, making it a high-yield area for your revision.
## Key Concepts
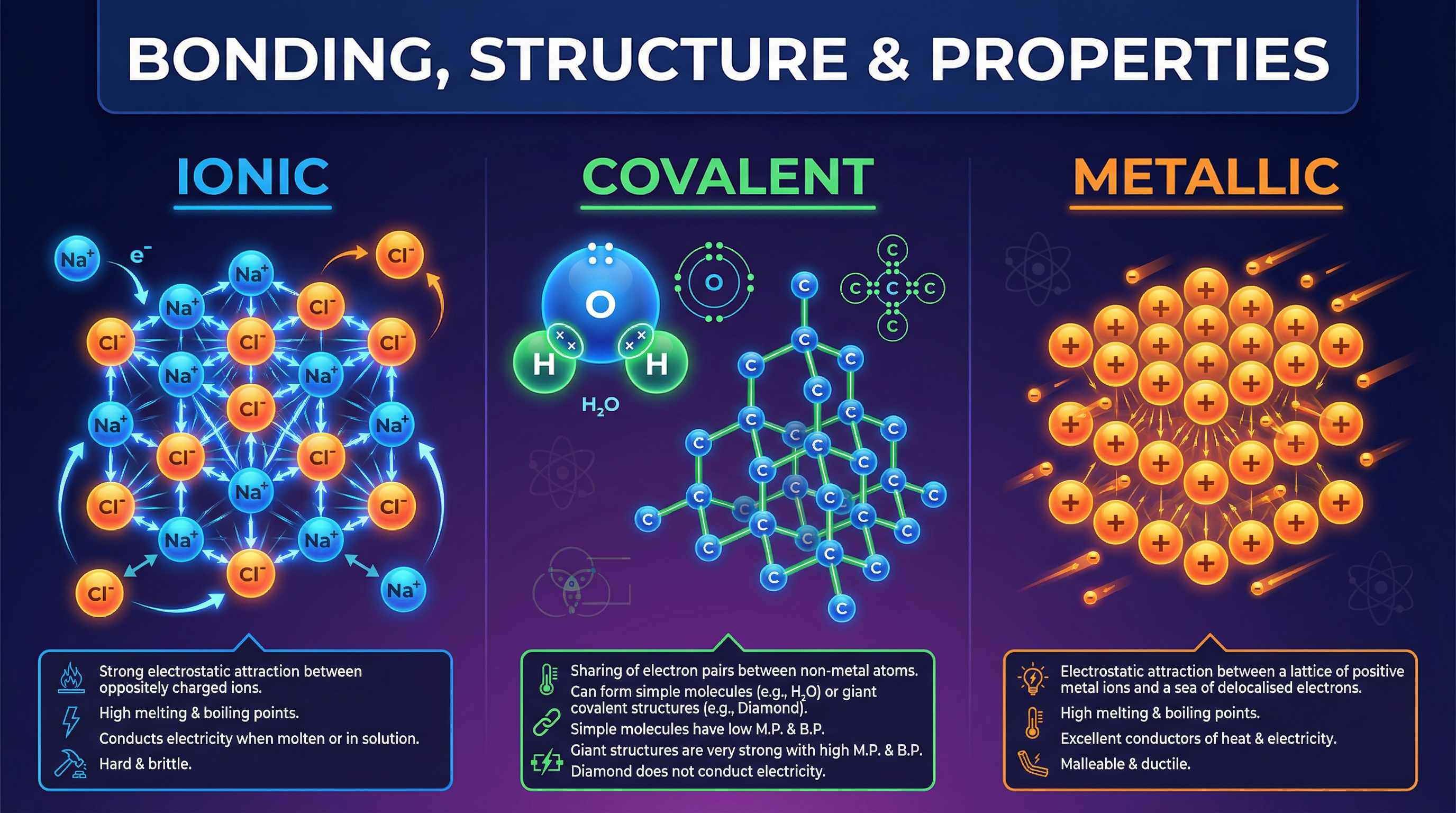
### Concept 1: Ionic Bonding
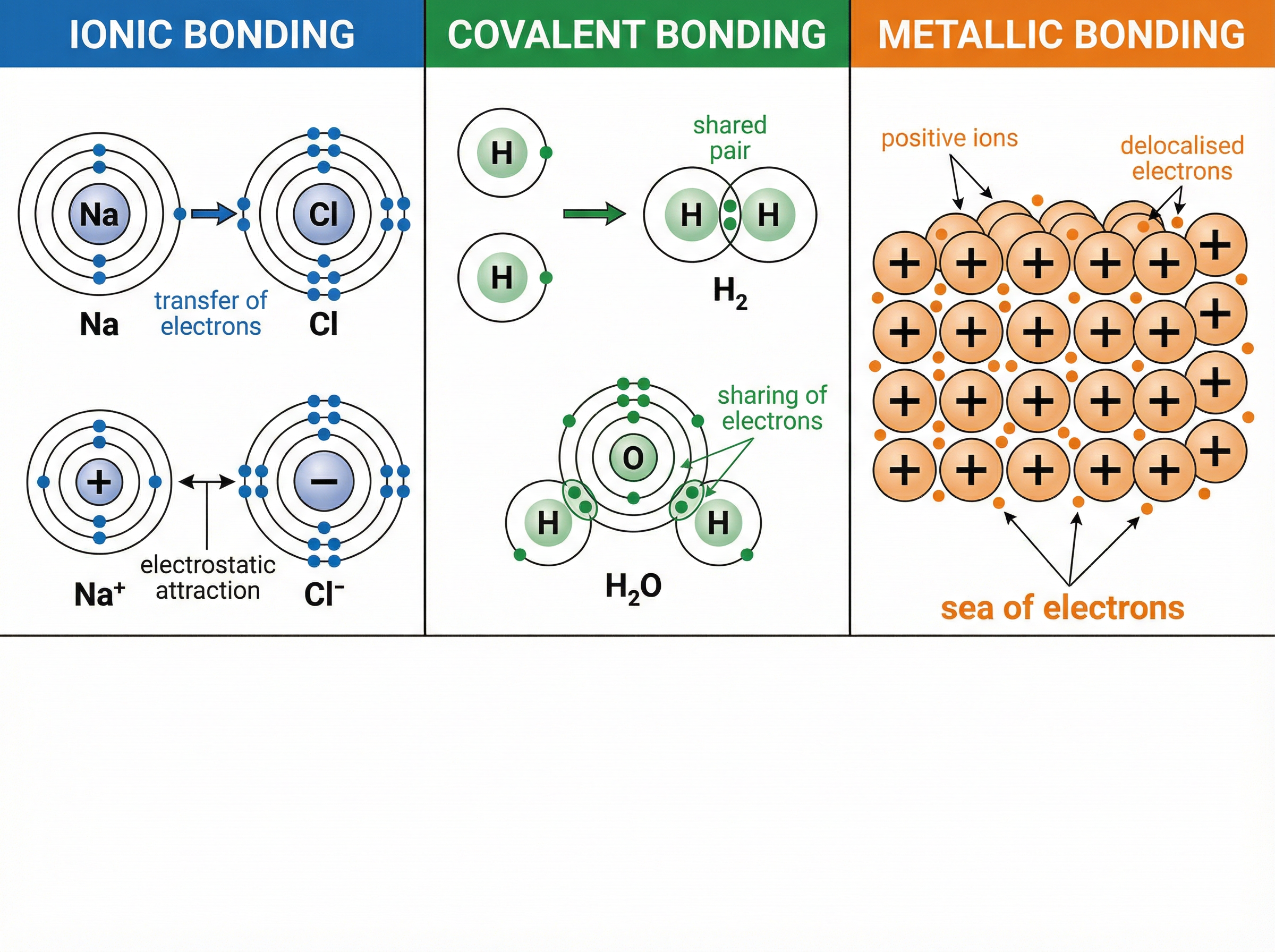
Ionic bonding occurs between metals and non-metals. When these elements react, metal atoms lose electrons to form positively charged ions (cations), and non-metal atoms gain those electrons to form negatively charged ions (anions).
The crucial phrase examiners are looking for is **electrostatic attraction between oppositely charged ions**. This attraction acts in all directions, creating a massive, regular 3D arrangement called a **giant ionic lattice**.
Because these electrostatic forces are incredibly strong, you need a massive amount of thermal energy to overcome them. This explains why ionic compounds like sodium chloride have very high melting and boiling points. Furthermore, solid ionic compounds cannot conduct electricity because the ions are locked in fixed positions. However, when molten or dissolved in water, the lattice breaks down, and the ions are free to move and carry electrical charge.
**Example**: Sodium (Na) transfers its 1 outer electron to Chlorine (Cl), which has 7. This forms Na⁺ and Cl⁻ ions, both with full outer shells, held together in a giant lattice.
### Concept 2: Covalent Bonding (Small Molecules)
Covalent bonding happens between non-metal atoms. Instead of transferring electrons, atoms **share pairs of electrons** to achieve full outer shells. The strong covalent bond is the electrostatic attraction between the positive nuclei of the atoms and the shared pair of negative electrons.
Many covalent substances exist as **small molecules** (e.g., H₂O, CO₂, CH₄). While the covalent bonds *within* the molecules are very strong, the **intermolecular forces** *between* the separate molecules are very weak.
When you melt or boil a simple molecular substance, you are only overcoming these weak intermolecular forces, **not** breaking the strong covalent bonds. This is why small molecules have low melting and boiling points and are often gases or liquids at room temperature. They also do not conduct electricity because they have no overall electrical charge or free electrons.
### Concept 3: Covalent Bonding (Giant Structures)
Some covalently bonded substances form **giant covalent structures** (macromolecules), where every atom is joined to adjacent atoms by strong covalent bonds in a continuous 3D network. There are no separate molecules here.
Because you must break millions of strong covalent bonds to melt or boil these substances, they have exceptionally high melting points and are very hard.
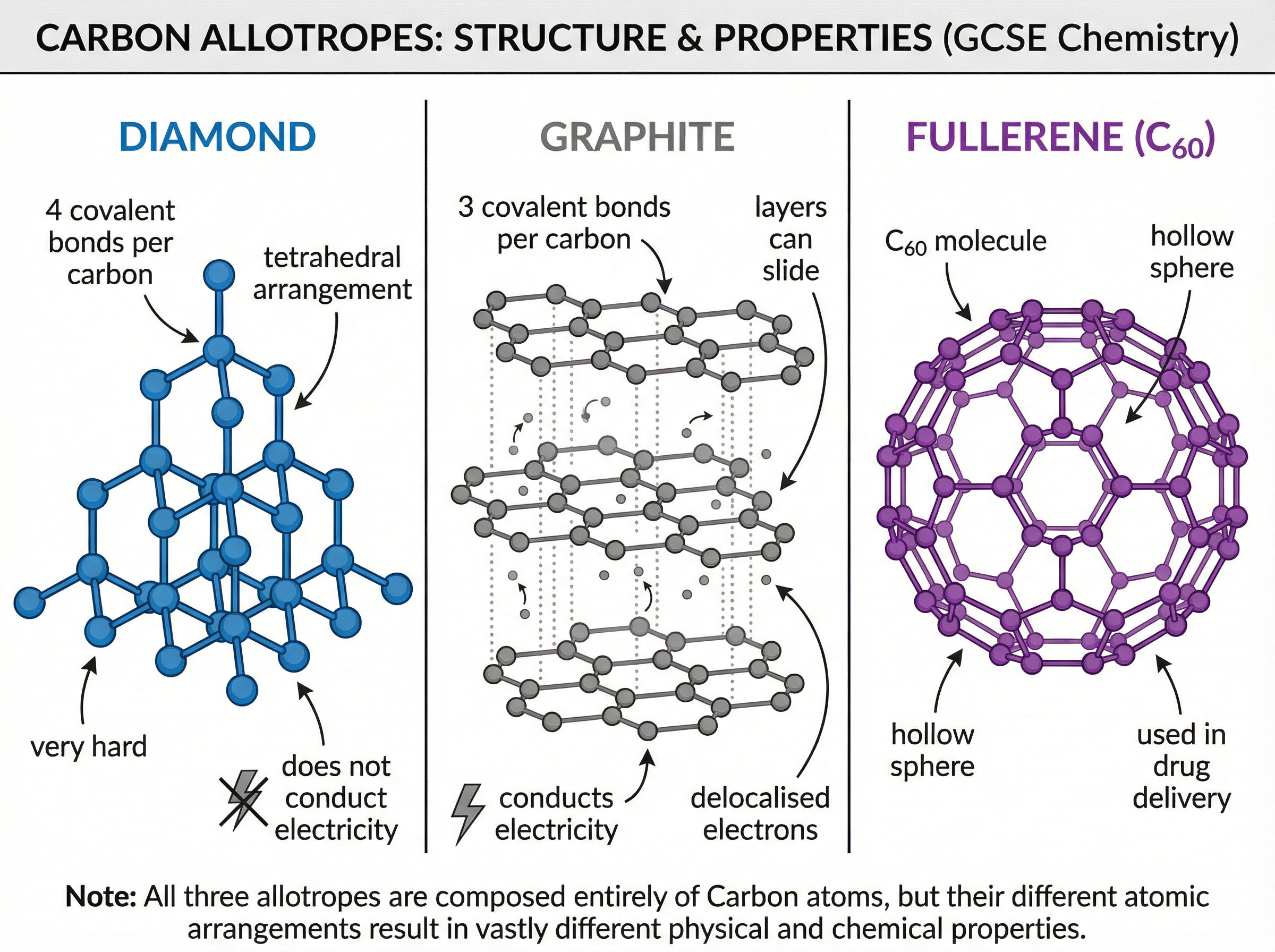
**Carbon Allotropes**:
* **Diamond**: Each carbon atom forms 4 covalent bonds in a rigid tetrahedral structure. It is extremely hard, has a very high melting point, and does not conduct electricity (no free electrons).
* **Graphite**: Each carbon atom forms 3 covalent bonds, creating flat hexagonal layers. The 4th electron from each carbon is **delocalised** between the layers. The layers are held together by weak intermolecular forces, allowing them to slide over each other (making graphite soft/slippery). The delocalised electrons allow graphite to conduct electricity and thermal energy.
* **Fullerenes**: Molecules of carbon with hollow shapes (e.g., Buckminsterfullerene, C₆₀). They are based on hexagonal rings of carbon atoms.
### Concept 4: Metallic Bonding
Metals consist of a giant structure of atoms arranged in a regular pattern. The electrons in the outer shell of metal atoms are **delocalised**—they are free to move throughout the whole structure.
Metallic bonding is the strong electrostatic attraction between the **positive metal ions** and the **sea of delocalised electrons**.
This structure perfectly explains metallic properties:
* **Conductivity**: The delocalised electrons can move through the structure, carrying electrical charge and thermal energy.
* **Malleability**: The layers of positive metal ions can slide over each other without breaking the metallic bonds, allowing metals to be bent and shaped.
* **High Melting Points**: The strong electrostatic attraction between the positive ions and delocalised electrons requires a lot of energy to overcome.
### Concept 5: Nanoparticles
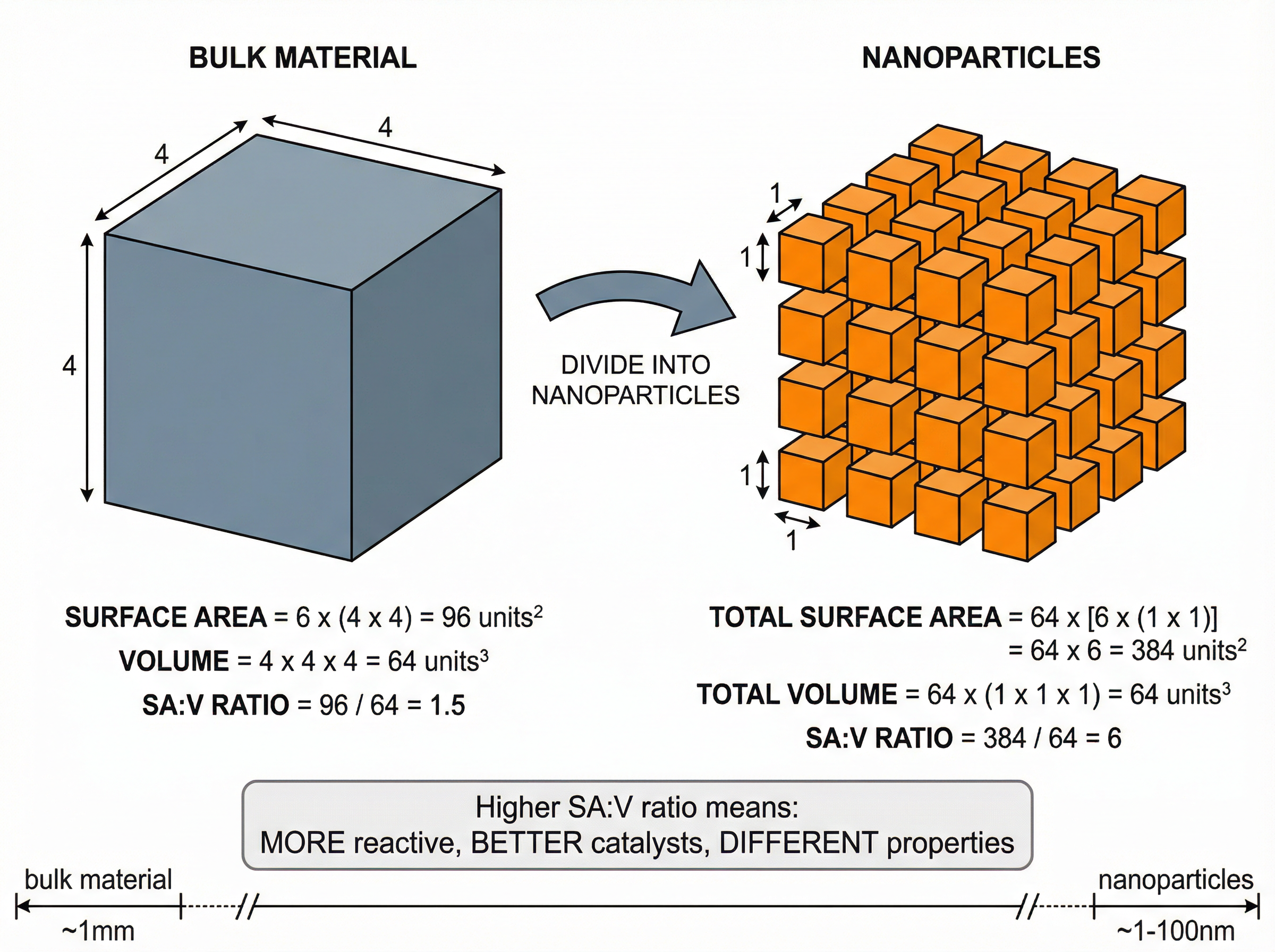
Nanoparticles are extremely small particles, typically 1-100 nanometres (nm) in size. They contain only a few hundred atoms.
The defining feature of nanoparticles is their incredibly high **surface area to volume ratio (SA:V)**. As a particle decreases in size, its surface area to volume ratio increases exponentially. Because chemical reactions happen on the surface of a material, nanoparticles are highly reactive and make excellent catalysts. A much smaller quantity of nanoparticles is needed compared to bulk materials to achieve the same effect.
## Mathematical/Scientific Relationships
* **Surface Area to Volume Ratio**: $ ext{Ratio} =
rac{ ext{Surface Area}}{ ext{Volume}}$
* *Note*: As the side length of a cube decreases by a factor of 10, the surface area to volume ratio increases by a factor of 10.
* **Nanometre Conversion**: $1 ext{ nm} = 1 imes 10^{-9} ext{ m}$ (Must memorise)
## Practical Applications
* **Graphite as a lubricant**: Because the layers can slide over each other easily.
* **Copper in electrical wiring**: Because it is an excellent conductor (due to delocalised electrons) and is ductile.
* **Nanoparticles in sunscreens**: Titanium dioxide nanoparticles provide better UV protection and don't leave white marks on the skin, though their long-term health effects are still being studied.
