Chemical analysis — AQA GCSE Study Guide
Exam Board: AQA | Level: GCSE
Master the detective work of chemistry. Learn how to identify unknown substances, calculate Rf values, and use both chemical tests and modern instrumental methods to secure top marks.
## Overview
Chemical Analysis (Topic 4.8) is where chemistry meets detective work. In this topic, you learn how to identify exactly what a substance is made of, whether it's a pure element or a complex mixture. This is crucial in the real world—from forensic scientists testing crime scene evidence to environmental agencies checking drinking water quality.
Examiners love this topic because it seamlessly blends practical skills with theoretical knowledge. You will be expected to recall specific colour changes, calculate $R_f$ values from chromatograms, and explain the advantages of modern instrumental methods over traditional chemical tests. This topic connects heavily to quantitative chemistry and the properties of transition metals.

---
## Key Concepts
### Concept 1: Purity, Formulations, and Chromatography
In everyday language, "pure" means nothing has been added to it (like "pure orange juice"). However, in chemistry, a **pure substance** contains only one element or one compound.
**Melting and Boiling Points:**
You can test purity by measuring melting and boiling points. A pure substance melts and boils at specific, sharp temperatures (e.g., pure water melts at exactly 0°C). If a substance is impure, its melting point will be **lower** and occur over a range of temperatures, while its boiling point will be **higher**.
**Formulations:**
A formulation is a mixture that has been designed as a useful product. Each component is present in a carefully measured quantity so the product has the required properties. Examples include paints, medicines, fuels, and fertilisers.
**Paper Chromatography:**
Chromatography is used to separate mixtures and help identify substances. It involves a **stationary phase** (the chromatography paper) and a **mobile phase** (the solvent, like water or ethanol). Substances separate because they have different attractions to the two phases. A pure substance will only produce one spot, whereas a mixture will separate into multiple spots.
### Concept 2: Identification of Common Gases
You must memorise the tests for four common gases. Examiners frequently ask for both the test and the positive observation.
1. **Hydrogen ($H_2$):** Hold a burning splint at the open end of a test tube of the gas. Hydrogen burns rapidly with a **squeaky pop** sound.
2. **Oxygen ($O_2$):** Insert a glowing splint into a test tube of the gas. The splint will **relight**.
3. **Carbon Dioxide ($CO_2$):** Bubble the gas through an aqueous solution of calcium hydroxide (limewater). The limewater turns **milky (cloudy)**.
4. **Chlorine ($Cl_2$):** Use damp litmus paper. When put into chlorine gas, the litmus paper is **bleached and turns white**.
### Concept 3: Identification of Metal Ions (Cations)
Metal ions can be identified using flame tests or sodium hydroxide solution.
**Flame Tests:**
Different metal ions produce distinct colours when heated in a flame.
* Lithium ($Li^+$) produces a **crimson** flame.
* Sodium ($Na^+$) produces a **yellow** flame.
* Potassium ($K^+$) produces a **lilac** flame.
* Calcium ($Ca^{2+}$) produces an **orange-red** flame.
* Copper ($Cu^{2+}$) produces a **green** flame.
*Note:* If a sample contains a mixture of ions, the colours of some ions can be masked (hidden) by others (especially sodium's strong yellow flame).

**Sodium Hydroxide Tests:**
When sodium hydroxide ($NaOH$) solution is added to solutions of metal ions, precipitates (insoluble solids) are formed.
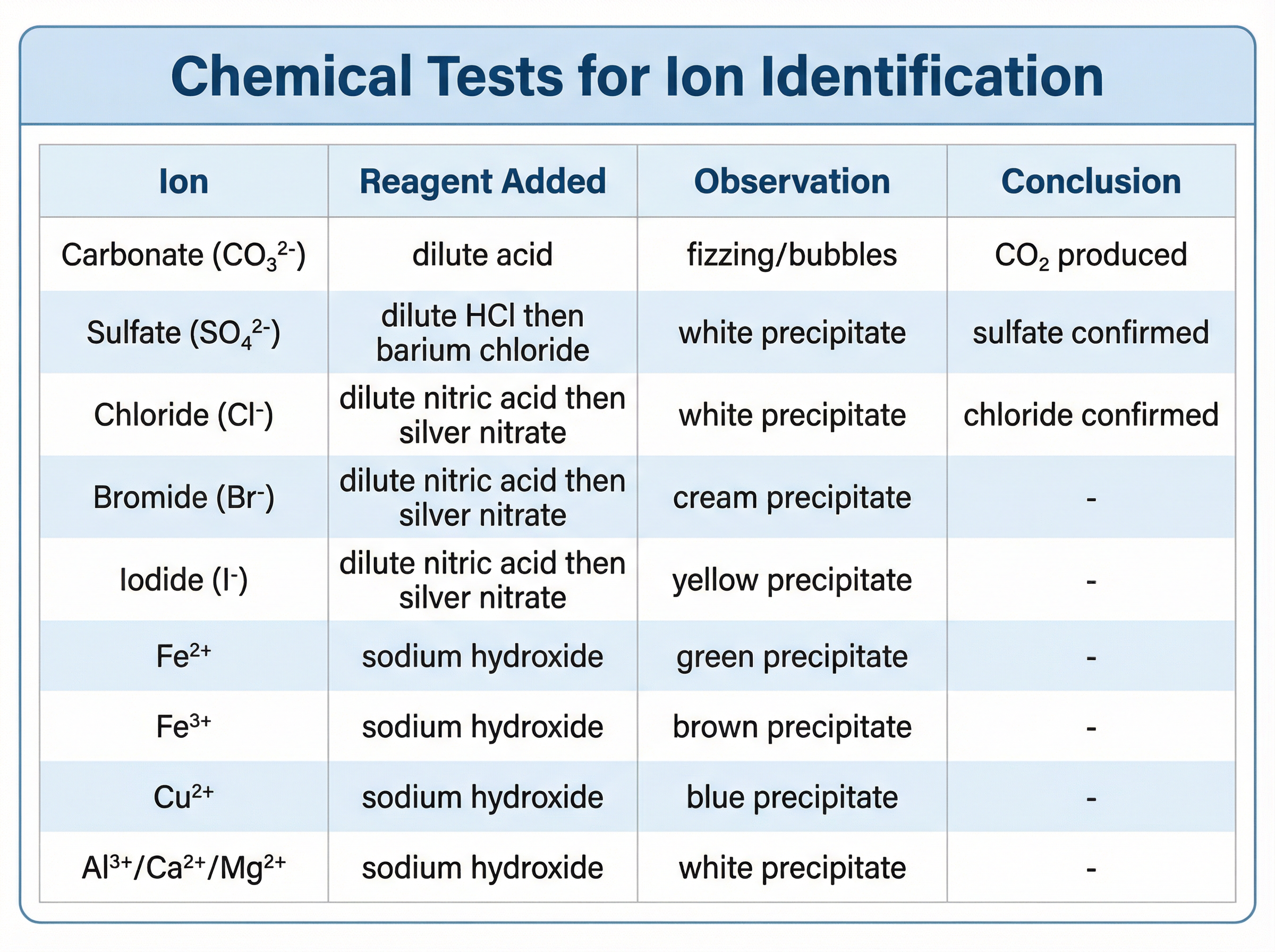
* **Aluminium ($Al^{3+}$), Calcium ($Ca^{2+}$), and Magnesium ($Mg^{2+}$)** all form **white** precipitates. Only the aluminium hydroxide precipitate dissolves when excess sodium hydroxide is added.
* **Copper(II) ($Cu^{2+}$)** forms a **blue** precipitate.
* **Iron(II) ($Fe^{2+}$)** forms a **green** precipitate.
* **Iron(III) ($Fe^{3+}$)** forms a **brown** precipitate.
### Concept 4: Identification of Non-Metal Ions (Anions)
You also need to know the chemical tests for negative ions.
**Carbonates ($CO_3^{2-}$):**
Add dilute acid to the sample. If carbonate ions are present, the mixture will fizz (effervesce) as carbon dioxide gas is produced. You confirm it is $CO_2$ by bubbling it through limewater.
**Halides ($Cl^-$, $Br^-$, $I^-$):**
First, add dilute nitric acid ($HNO_3$), followed by silver nitrate solution ($AgNO_3$).
* Chloride ($Cl^-$) gives a **white** precipitate of silver chloride.
* Bromide ($Br^-$) gives a **cream** precipitate of silver bromide.
* Iodide ($I^-$) gives a **yellow** precipitate of silver iodide.
**Sulfates ($SO_4^{2-}$):**
First, add dilute hydrochloric acid ($HCl$), followed by barium chloride solution ($BaCl_2$). If sulfate ions are present, a **white** precipitate of barium sulfate forms.
### Concept 5: Instrumental Methods
Modern laboratories use instrumental methods (machines) to analyse chemical substances. A key example is **Flame Emission Spectroscopy**, which is used to analyse metal ions in solutions. The sample is put into a flame, and the light given out is passed through a spectroscope. The output is a line spectrum that can be analysed to identify the metal ions and calculate their concentrations.
**Advantages of Instrumental Methods:**
Compared to traditional chemical tests, instrumental methods are:
1. **Highly accurate** (reliable results)
2. **Highly sensitive** (can detect tiny amounts of substances)
3. **Very rapid** (fast results)
---
## Mathematical/Scientific Relationships
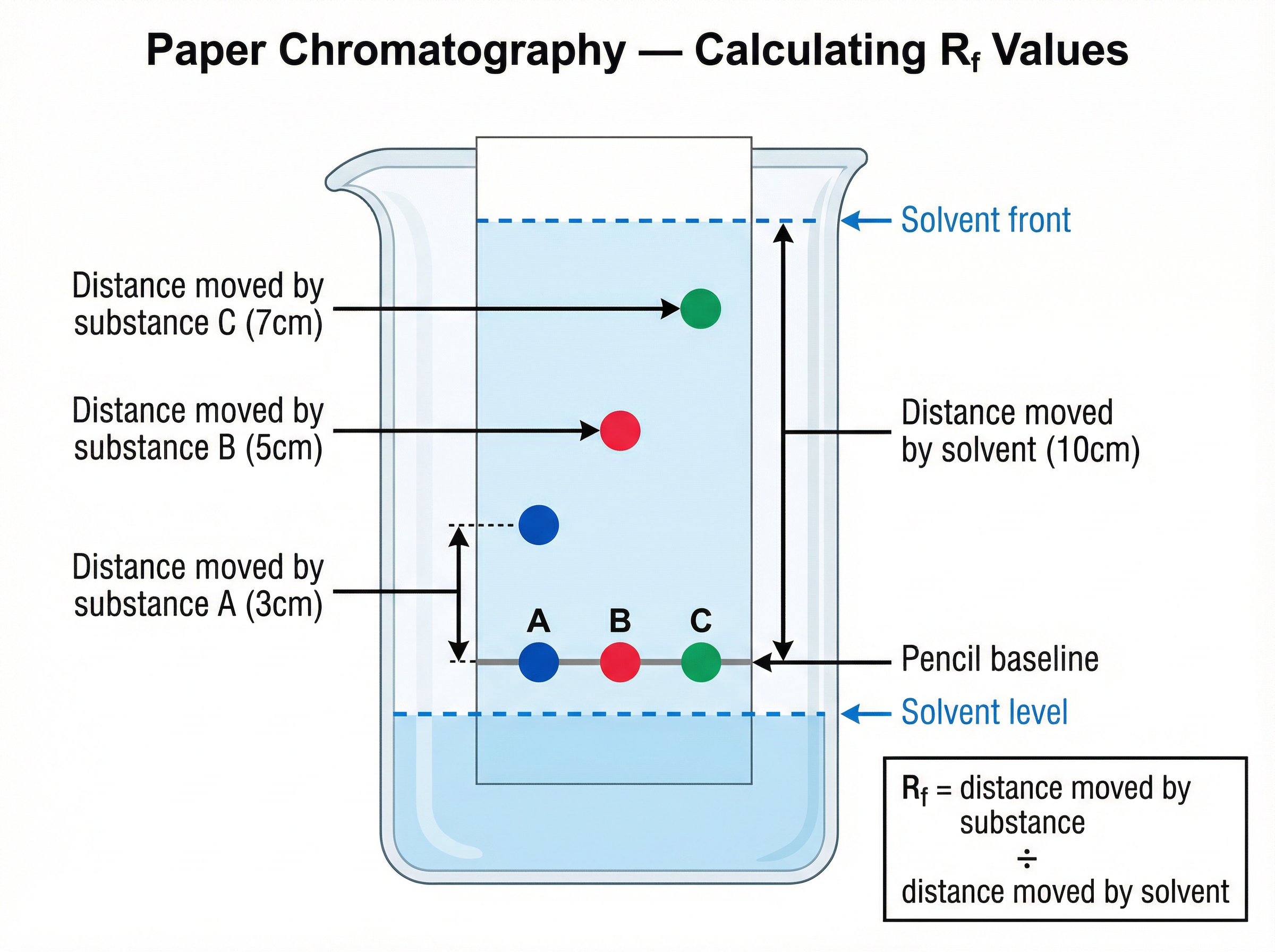
**Calculating $R_f$ Values (Retention Factor):**
$$R_f =
rac{ ext{distance moved by substance}}{ ext{distance moved by solvent}}$$
* **Distance moved by substance:** Measure from the pencil baseline to the centre of the spot.
* **Distance moved by solvent:** Measure from the pencil baseline to the solvent front.
* **Rule:** $R_f$ values are always between 0 and 1. They have no units.
## Practical Applications
**Required Practical: Chromatography**
* **Apparatus:** Chromatography paper, capillary tube, beaker, solvent (e.g., water), pencil, ruler.
* **Method:** Draw a pencil line 2 cm from the bottom of the paper. Use a capillary tube to put a small spot of the mixture on the line. Place the paper in a beaker with a small volume of solvent (the solvent level must be *below* the pencil line). Wait for the solvent to travel up the paper. Remove the paper, mark the solvent front with a pencil, and let it dry.
* **Common Errors:** Using a pen for the baseline (the ink will run and interfere with results). Having the solvent level above the baseline (the spots will wash off into the solvent).
**Listen to the Podcast:**
