Chemical changes — AQA GCSE Study Guide
Exam Board: AQA | Level: GCSE
Master the transformative world of Chemical Changes! This topic covers the reactivity series, metal extraction, acids and bases, and the complex rules of electrolysis, providing the foundation for understanding industrial chemistry and earning high marks in your exams.
## Overview
Welcome to **Chemical Changes**, one of the core pillars of GCSE Chemistry. This topic is fundamentally about how substances interact, transform, and exchange electrons to form new compounds. It bridges the gap between the theoretical atomic structure you learned earlier and the practical industrial processes that shape our modern world.
In this topic, we explore three main areas:
1. **Metals and Reactivity**: Why some metals explode in water while others lie dormant in the ground for millennia, and how we extract them.
2. **Acids, Alkalis, and Salts**: The chemistry of neutralisation, pH, and how to safely produce specific chemical salts.
3. **Electrolysis**: The fascinating process of using electrical energy to rip ionic compounds apart, a technique crucial for producing aluminium, chlorine, and purifying copper.
Examiners love this topic because it tests your ability to link observations to underlying atomic theory. You will frequently encounter questions requiring you to balance equations, predict products of reactions, and explain processes using precise terminology like *oxidation* and *reduction*.
---
## Listen to the Podcast
*Listen to a 10-minute audio summary covering all the key concepts and exam tips.*

---
## Key Concepts
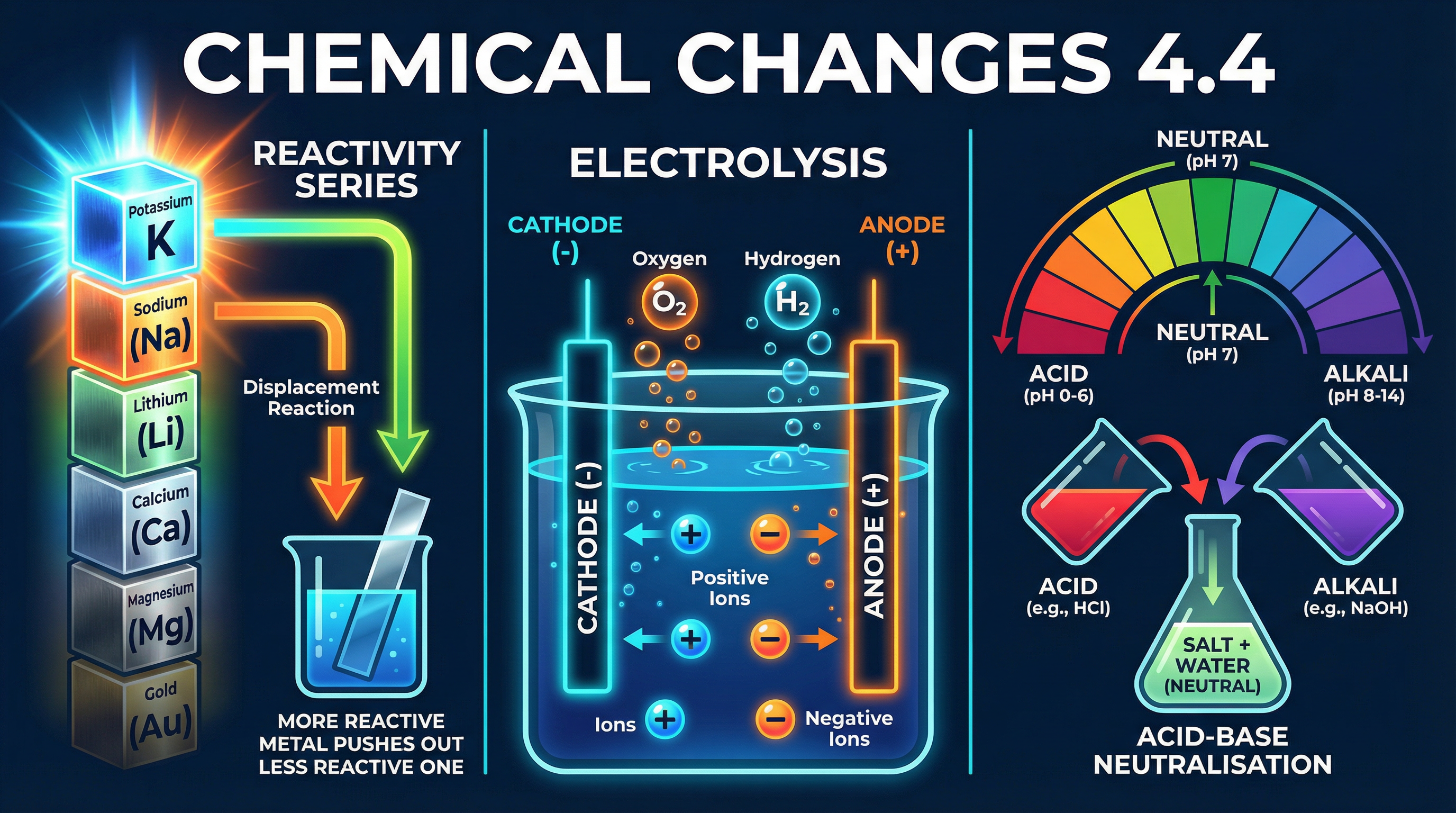
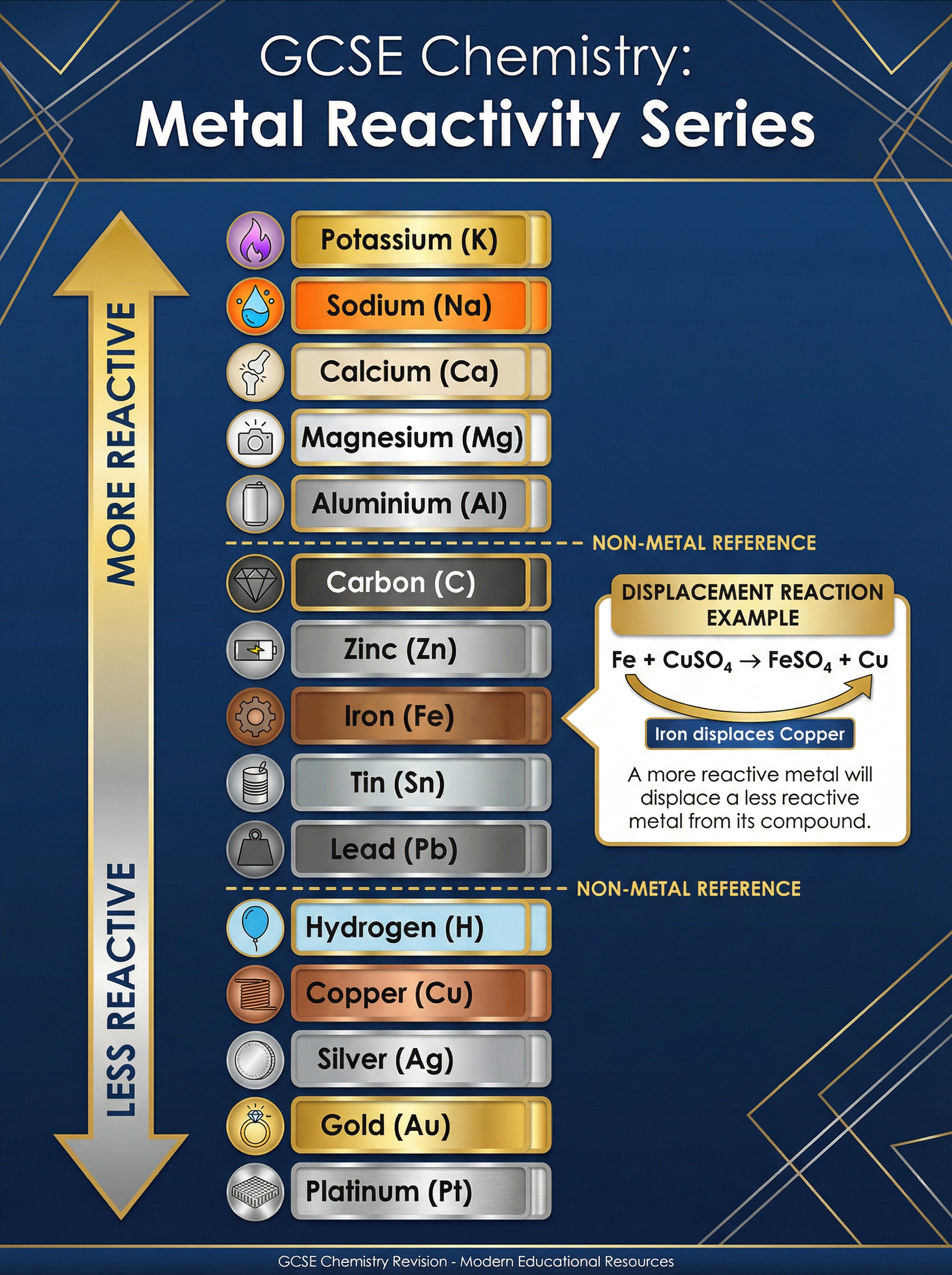
### Concept 1: The Reactivity Series and Displacement
The reactivity series is a list of metals ordered by their tendency to lose electrons and form positive ions. The higher a metal is on the series, the more reactive it is.
**Why does this work?**
Reactivity depends on atomic structure. Metals react by losing their outer shell electrons. Potassium is highly reactive because its outer electron is far from the positive nucleus, shielded by inner electron shells, making it very easy to lose.
**Displacement Reactions**
A fundamental rule: **A more reactive metal will displace a less reactive metal from an aqueous solution of its salt.**
**Example**: Adding zinc powder to copper(II) sulfate solution.
Zinc is more reactive than copper. It "pushes out" the copper from the compound.
Equation: $\text{Zn (s)} + \text{CuSO}_4 \text{ (aq)} \rightarrow \text{ZnSO}_4 \text{ (aq)} + \text{Cu (s)}$
*Observation*: The blue solution fades to colourless (zinc sulfate), and a reddish-brown solid (copper) forms.
### Concept 2: Extraction of Metals
Most metals are found in the Earth's crust combined with other elements (often oxygen) as ores. The method used to extract them depends entirely on their position in the reactivity series.
* **Metals below carbon** (e.g., Zinc, Iron, Lead, Copper): Extracted by **reduction with carbon**. Carbon is more reactive, so it displaces the metal from its oxide.
* *Example*: $\text{2Fe}_2\text{O}_3 + \text{3C} \rightarrow \text{4Fe} + \text{3CO}_2$
* **Metals above carbon** (e.g., Aluminium, Magnesium, Sodium): Extracted by **electrolysis** of the molten compound. They are too reactive to be reduced by carbon. This process requires vast amounts of electricity, making these metals more expensive to produce.
* **Unreactive metals** (e.g., Gold, Platinum): Found native (as the uncombined element) in the Earth's crust.
### Concept 3: Oxidation and Reduction (Redox)
Reactions involving the transfer of oxygen or electrons are called redox reactions. Oxidation and reduction *always* occur together.
**Definitions:**
1. **In terms of oxygen:**
* **Oxidation**: Gain of oxygen.
* **Reduction**: Loss of oxygen.
2. **In terms of electrons (Higher Tier):**
* **Oxidation**: Loss of electrons.
* **Reduction**: Gain of electrons.
* *Memory Hook*: **OIL RIG** (Oxidation Is Loss, Reduction Is Gain).
### Concept 4: Acids, Alkalis, and Neutralisation
* **Acids** produce hydrogen ions ($\text{H}^+$) in aqueous solutions.
* **Alkalis** (soluble bases) produce hydroxide ions ($\text{OH}^-$) in aqueous solutions.
The **pH scale** measures the acidity or alkalinity of a solution, ranging from 0 (strongly acidic) to 14 (strongly alkaline), with 7 being neutral. It is a logarithmic scale: a decrease of 1 on the pH scale represents a 10-fold increase in the concentration of $\text{H}^+$ ions.
**Neutralisation** occurs when an acid reacts with a base to form a salt and water.
Ionic equation for neutralisation: $\text{H}^+\text{(aq)} + \text{OH}^-\text{(aq)} \rightarrow \text{H}_2\text{O(l)}$
**Naming Salts:**
The first part of the name comes from the metal in the base (e.g., Sodium from Sodium Hydroxide). The second part comes from the acid used:
* Hydrochloric acid $\rightarrow$ Chloride
* Sulfuric acid $\rightarrow$ Sulfate
* Nitric acid $\rightarrow$ Nitrate
### Concept 5: Electrolysis
Electrolysis is the breakdown of an ionic compound, molten or in aqueous solution, by the passage of electricity.
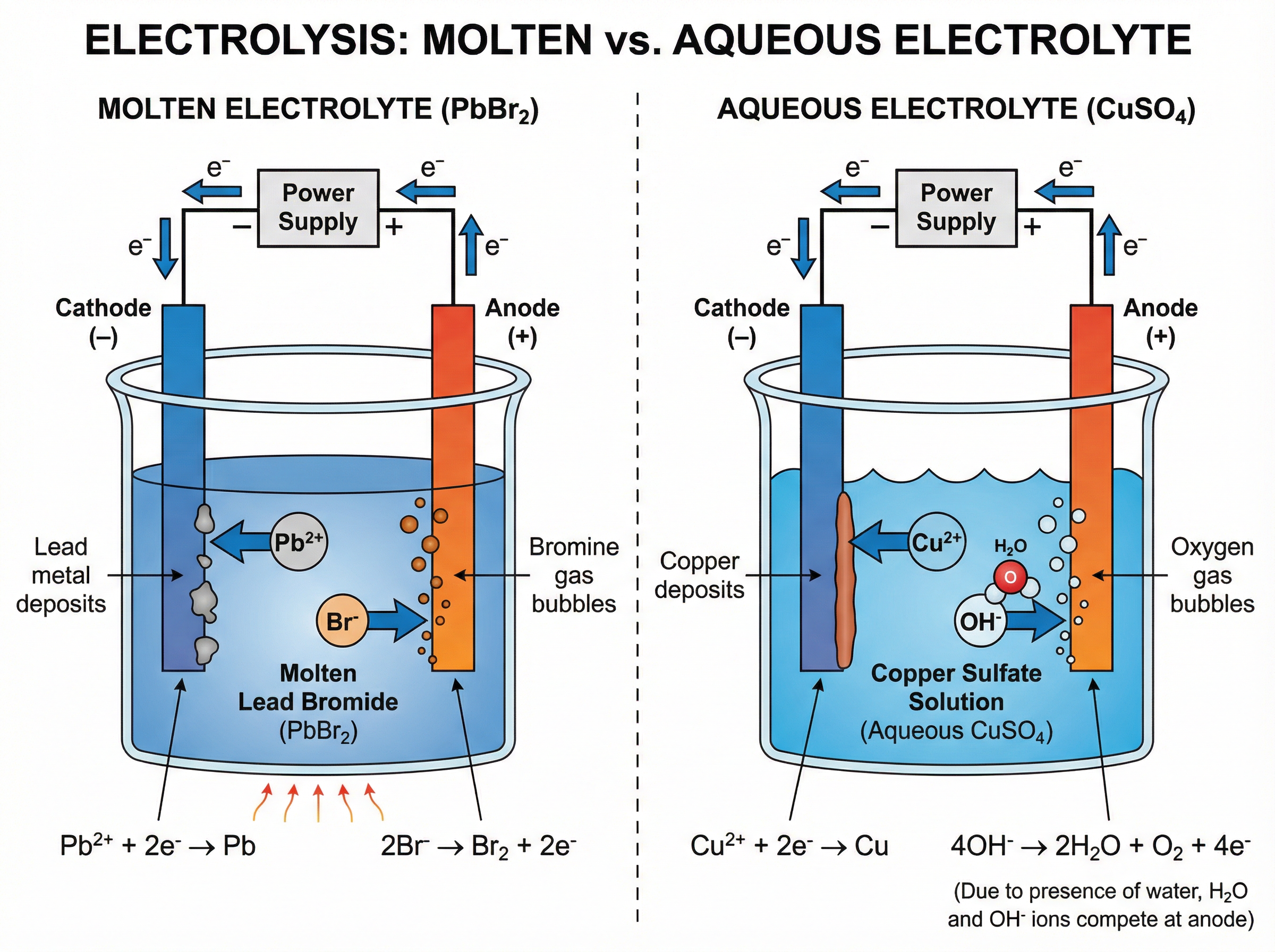
**The Setup:**
* **Electrolyte**: The liquid or solution containing mobile ions.
* **Cathode**: The negative electrode. Attracts positive ions (cations).
* **Anode**: The positive electrode. Attracts negative ions (anions).
**Molten Electrolytes:**
Simple! The metal ion goes to the cathode and is reduced to the metal. The non-metal ion goes to the anode and is oxidised to the non-metal.
*Example: Molten Lead(II) Bromide ($\text{PbBr}_2$)*
* Cathode: $\text{Pb}^{2+} + \text{2e}^- \rightarrow \text{Pb}$ (Lead metal forms)
* Anode: $\text{2Br}^- \rightarrow \text{Br}_2 + \text{2e}^-$ (Bromine gas forms)
**Aqueous Electrolytes (The Tricky Part):**
Because the compound is dissolved in water, water molecules also ionise slightly to form $\text{H}^+$ and $\text{OH}^-$ ions. There is competition at the electrodes!
* **At the Cathode (-):** The *least reactive* positive ion is discharged. If the metal is more reactive than hydrogen (e.g., Sodium), **hydrogen gas** is produced. If the metal is less reactive than hydrogen (e.g., Copper), the **metal** is produced.
* **At the Anode (+):** If halide ions ($\text{Cl}^-$, $\text{Br}^-$, $\text{I}^-$) are present, the **halogen gas** is produced. If no halide ions are present (e.g., sulfates, nitrates), **oxygen gas** is produced from the discharge of hydroxide ions: $\text{4OH}^- \rightarrow \text{2H}_2\text{O} + \text{O}_2 + \text{4e}^-$.
---
## Required Practicals
### 1. Preparation of a Pure, Dry Sample of a Soluble Salt
**Method:**
1. Measure a set volume of acid (e.g., sulfuric acid) into a beaker and gently heat it using a Bunsen burner (speeds up reaction).
2. Add an insoluble base (e.g., copper(II) oxide) in small amounts, stirring constantly, until it is in excess (stops reacting and sits at the bottom). This ensures all the acid is neutralised.
3. Filter the mixture to remove the excess unreacted base. The filtrate is the salt solution.
4. Pour the filtrate into an evaporating basin and heat over a water bath until crystals start to form around the edge (crystallisation point).
5. Leave the solution to cool and crystallise completely. Filter the crystals and pat dry with filter paper.
### 2. Electrolysis of Aqueous Solutions
**Method:**
1. Pour the electrolyte (e.g., copper(II) sulfate solution) into a beaker.
2. Insert two inert electrodes (e.g., graphite or platinum) into the solution.
3. Connect the electrodes to a DC power supply.
4. Observe the electrodes for gas bubbles or metal deposition. Test any gases produced (e.g., damp blue litmus paper bleaches white for chlorine; glowing splint relights for oxygen; lit splint gives a 'squeaky pop' for hydrogen).
---
## Mathematical/Scientific Relationships
* **pH Scale Logarithmic Relationship**: A change of 1 pH unit = a 10x change in $\text{H}^+$ ion concentration.
* *Example*: If a solution's pH drops from 4 to 2, the $\text{H}^+$ concentration has increased by a factor of $10 \times 10 = 100$. (Must memorise)
* **Balancing Equations**: The number of atoms of each element must be the same on both sides of the equation.
* **Balancing Half-Equations**: The total charge on the left side must equal the total charge on the right side. Ensure electrons ($\text{e}^-$) are added to the correct side (left for reduction, right for oxidation).