Chemistry of the atmosphere — AQA GCSE Study Guide
Exam Board: AQA | Level: GCSE
Master the 4.6 billion-year story of our planet's atmosphere, from its fiery volcanic origins to the modern pollution challenges we face today. This topic is heavily tested on extended response questions, so precision with terminology like 'greenhouse effect' and 'incomplete combustion' is essential for securing top marks.
## Overview
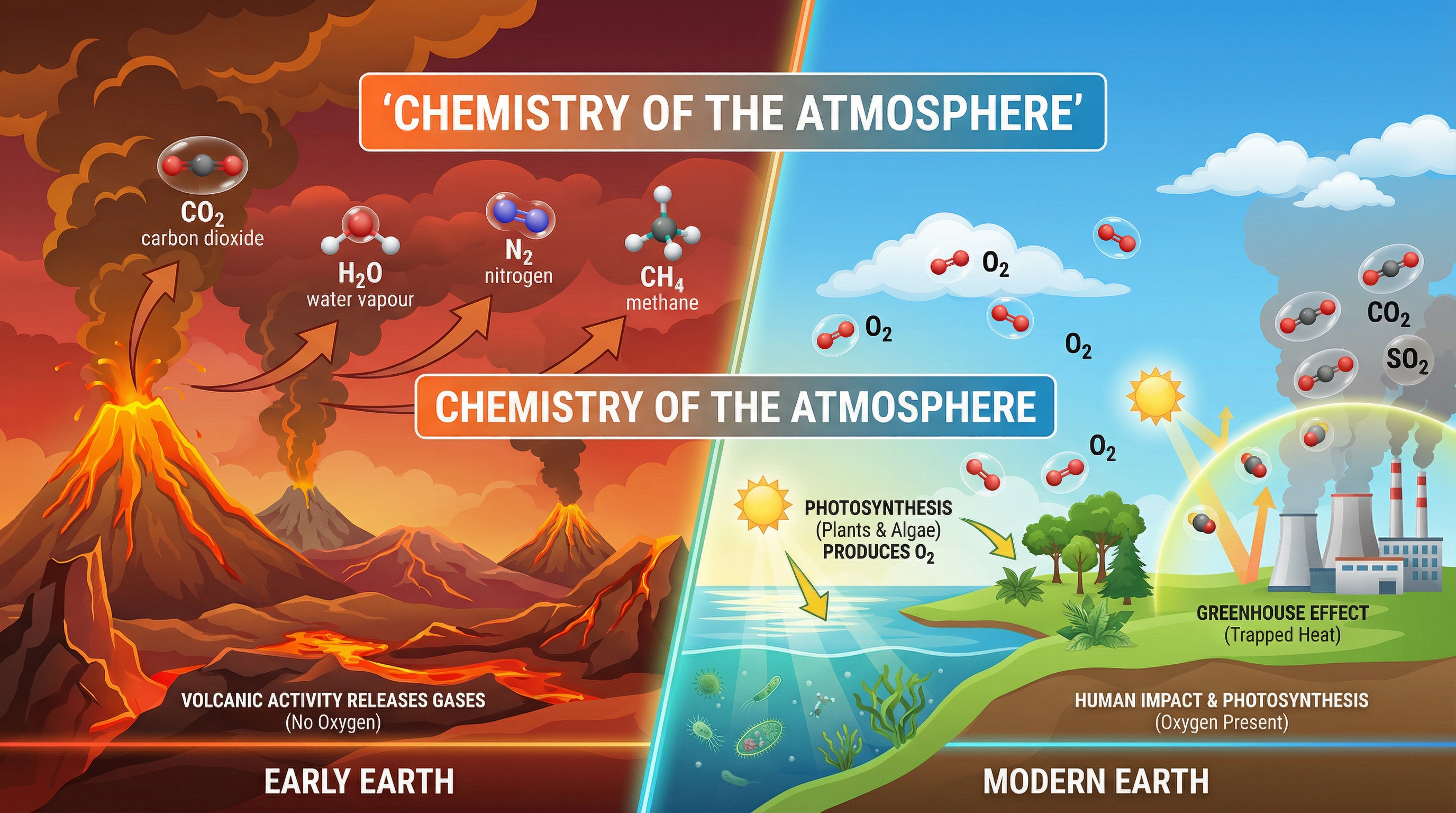
Welcome to the Chemistry of the Atmosphere! This topic tells the incredible 4.6 billion-year story of the air we breathe. It is a vital part of the GCSE Chemistry specification because it connects fundamental chemical principles—like combustion and reversible reactions—to the most pressing environmental challenges of our time, such as climate change and air pollution.
Examiners love this topic for 6-mark extended response questions. They will test your ability to explain *how* the atmosphere evolved and *why* human activities are changing it today. You must be precise with your terminology; for example, candidates frequently lose marks by confusing the greenhouse effect with global warming or ozone depletion.
Before you dive into reading, why not listen to our exclusive podcast episode? It covers everything you need to know in just 10 minutes.

## Key Concepts
### Concept 1: The Early Atmosphere
Around 4.6 billion years ago, the Earth was highly active with volcanoes. These volcanoes erupted constantly, releasing huge volumes of gases that formed the early atmosphere. The main gases were carbon dioxide ($CO_2$) and water vapour ($H_2O$), along with smaller proportions of nitrogen ($N_2$), methane ($CH_4$), and ammonia ($NH_3$). Crucially, there was **no oxygen** ($O_2$). Examiners often expect candidates to compare this early atmosphere to the present-day atmospheres of Mars and Venus.
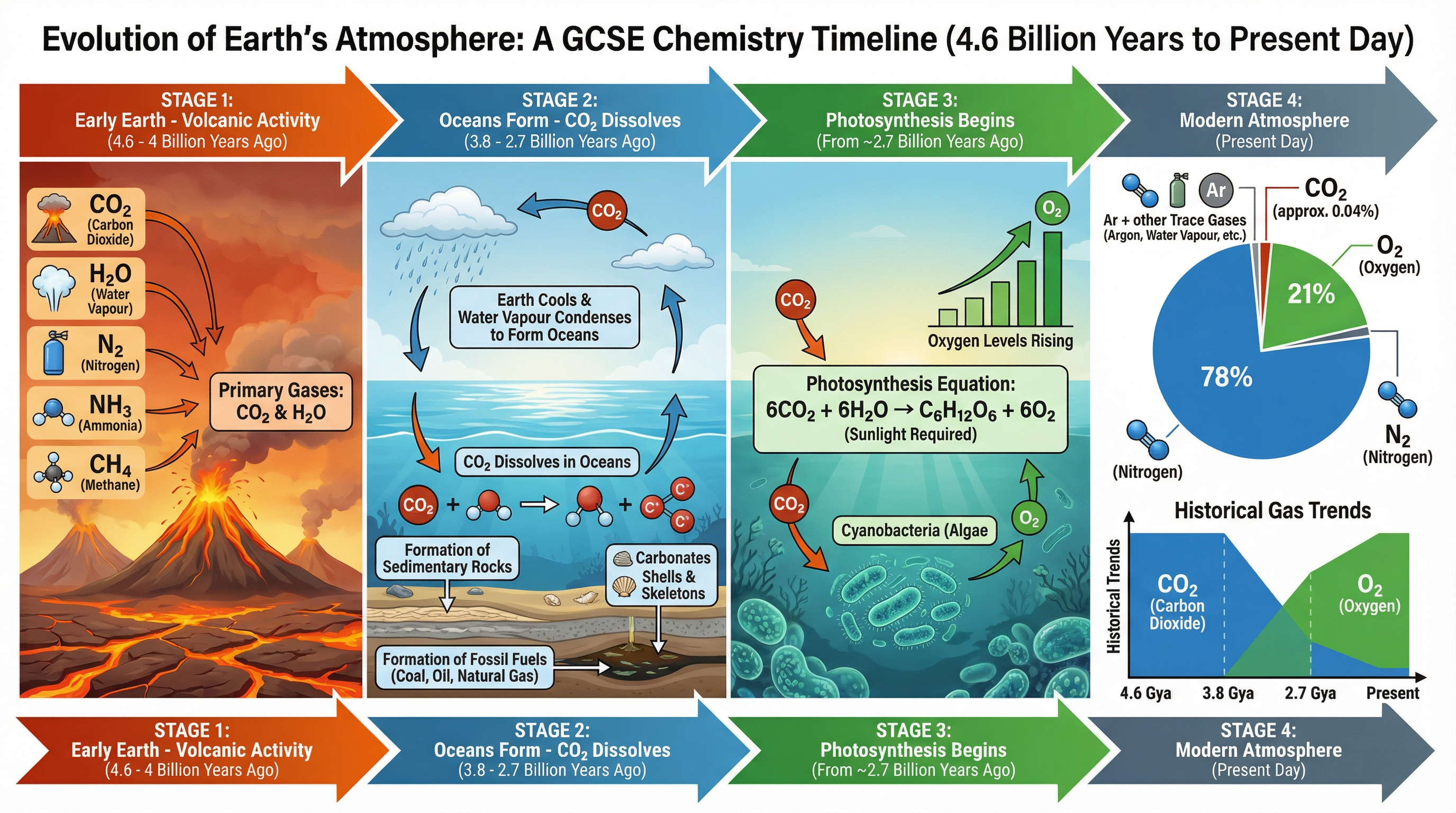
### Concept 2: How Carbon Dioxide Decreased
As the Earth gradually cooled, the water vapour in the atmosphere condensed to form the oceans. This was a pivotal moment because carbon dioxide is a soluble gas. Huge amounts of $CO_2$ dissolved in the newly formed oceans.
Once dissolved, the carbon dioxide underwent chemical reactions to form carbonate precipitates, which produced sediments on the seabed. Over millions of years, these sediments formed carbonate rocks like limestone. Furthermore, when marine organisms evolved, they used these carbonates to build their shells and skeletons. When they died, their remains were compressed over millions of years to form fossil fuels (coal, crude oil, and natural gas), locking away the carbon.
### Concept 3: How Oxygen Increased
About 2.7 billion years ago, algae first evolved. They were the first organisms to perform **photosynthesis**, a process that uses energy from sunlight to convert carbon dioxide and water into glucose and oxygen. Over the next billion years, plants evolved and the percentage of oxygen gradually increased to a level that enabled animals to evolve.
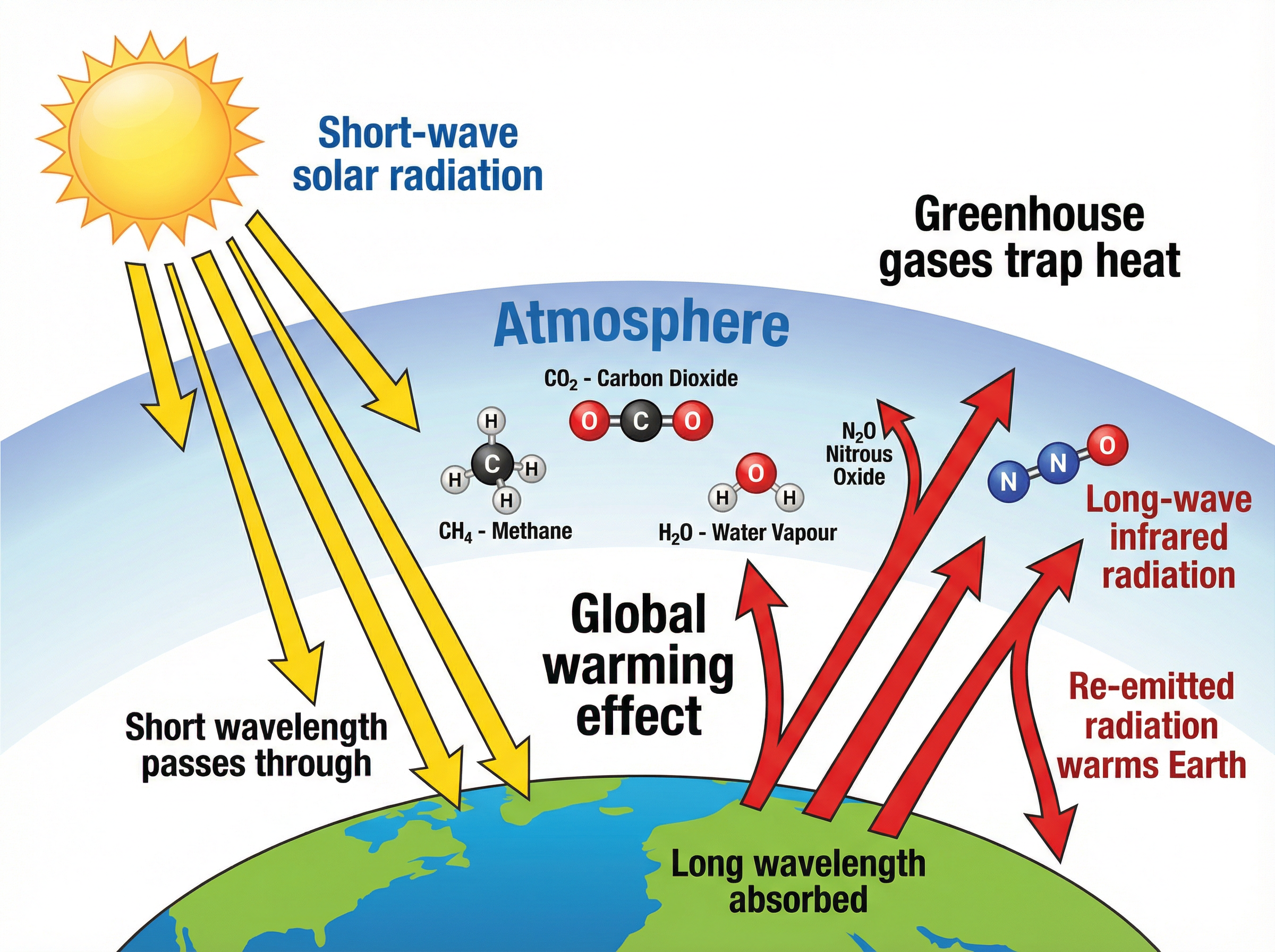
### Concept 4: The Greenhouse Effect
The greenhouse effect is a natural and necessary process that keeps the Earth warm enough to support life. However, human activities are causing an *enhanced* greenhouse effect.
Here is the exact mechanism examiners want to see:
1. The Sun emits **short-wavelength radiation** (e.g., visible light) which passes through the Earth's atmosphere.
2. The Earth's surface absorbs this radiation and warms up.
3. The Earth's surface then re-emits energy as **long-wavelength infrared radiation** (heat).
4. Greenhouse gases (like $CO_2$, $CH_4$, and $H_2O$) in the atmosphere absorb this long-wavelength radiation.
5. The greenhouse gases re-emit the radiation in all directions, including back towards the Earth, trapping the heat.
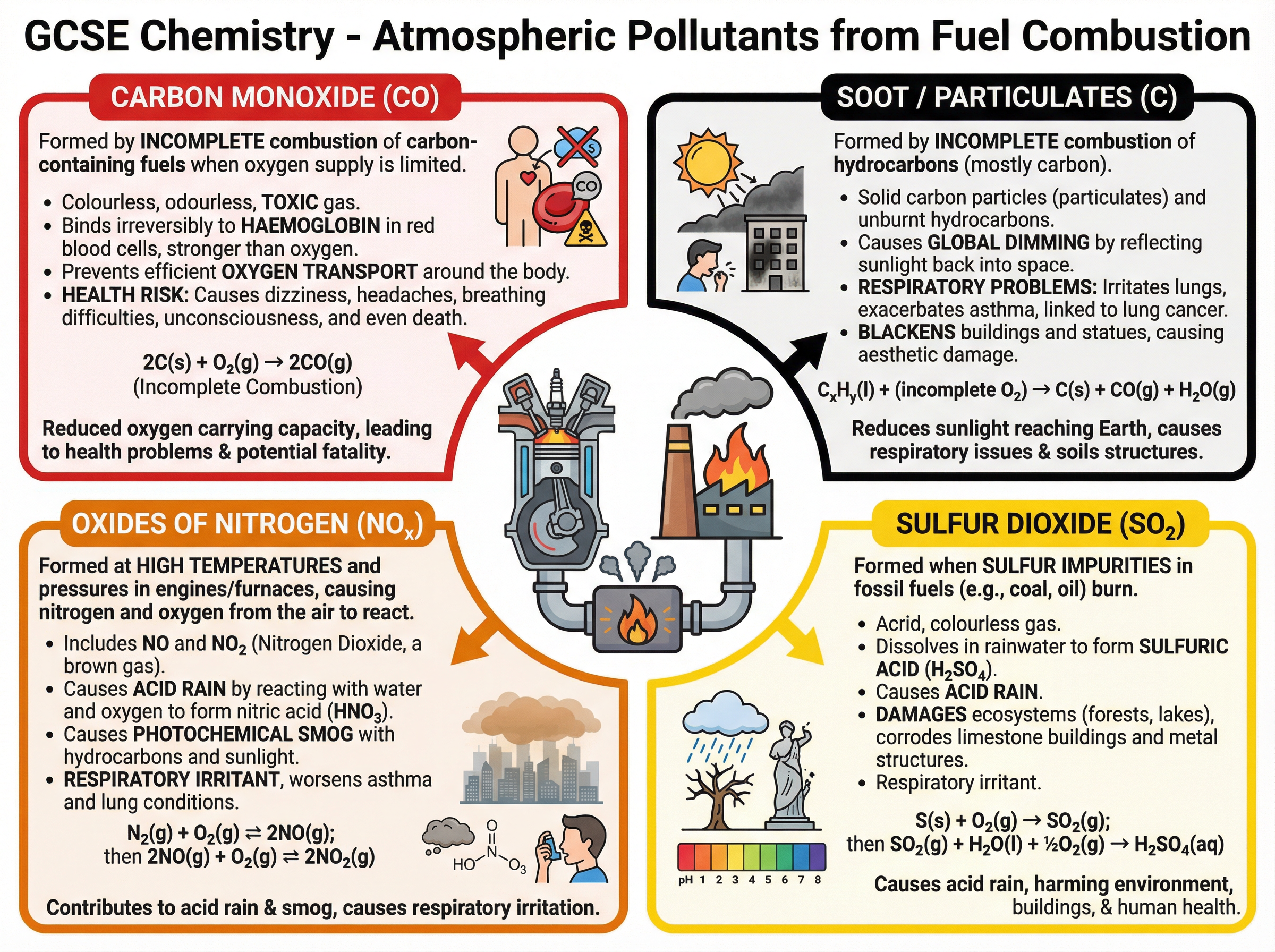
### Concept 5: Atmospheric Pollutants
When we burn fossil fuels, we release various pollutants into the atmosphere. The products depend on the fuel's composition and the conditions of combustion.
- **Carbon Monoxide ($CO$)**: Formed during *incomplete combustion* (when oxygen is limited). It is a toxic, colourless, and odourless gas that binds to haemoglobin in the blood, preventing oxygen transport.
- **Soot (Carbon Particulates)**: Also formed during incomplete combustion. It causes global dimming and respiratory problems.
- **Sulfur Dioxide ($SO_2$)**: Formed when sulfur impurities in fossil fuels (especially coal) react with oxygen. It causes acid rain and respiratory issues.
- **Oxides of Nitrogen ($NO_x$)**: Formed when nitrogen and oxygen from the air react at the high temperatures found in car engines. They cause acid rain and photochemical smog.
## Mathematical/Scientific Relationships
**Photosynthesis Equation:**
$6CO_2 + 6H_2O \rightarrow C_6H_{12}O_6 + 6O_2$
*(Carbon dioxide + Water $\rightarrow$ Glucose + Oxygen)*
**Complete Combustion of a Hydrocarbon (e.g., Methane):**
$CH_4 + 2O_2 \rightarrow CO_2 + 2H_2O$
**Incomplete Combustion (producing Carbon Monoxide):**
$2CH_4 + 3O_2 \rightarrow 2CO + 4H_2O$
**Formation of Sulfur Dioxide:**
$S + O_2 \rightarrow SO_2$
## Practical Applications
Understanding these gases is essential for modern environmental science. For example, catalytic converters in cars are designed specifically to reduce emissions of carbon monoxide and oxides of nitrogen. Environmental monitoring stations constantly measure the levels of particulates ($PM_{10}$ and $PM_{2.5}$) to issue health warnings for people with asthma.