Energy changes — AQA GCSE Study Guide
Exam Board: AQA | Level: GCSE
Master the critical differences between exothermic and endothermic reactions, learn to interpret reaction profiles, and calculate bond energies. This topic is a guaranteed mark-earner if you understand the direction of energy transfer.
## Overview
Welcome to Energy Changes (Topic 4.5), a fundamental area of GCSE Chemistry. Every chemical reaction involves an energy transfer—energy is never created or destroyed, only moved between the reacting chemicals and their surroundings. Understanding whether a reaction releases heat (exothermic) or absorbs heat (endothermic) is crucial not just for passing your exams, but for understanding real-world applications like hand warmers, sports injury packs, and hydrogen fuel cells.
Examiners love this topic because it tests multiple skills: your ability to recall definitions, interpret graphical data (reaction profiles), and perform multi-step calculations (bond energies). It also links heavily to rates of reaction and equilibrium. Let's break down the core concepts so you can secure maximum marks.
## Key Concepts
### Concept 1: Exothermic and Endothermic Reactions
In chemistry, we divide the universe into two parts: the **system** (the reacting chemicals) and the **surroundings** (everything else, including the test tube, the air, and the thermometer).
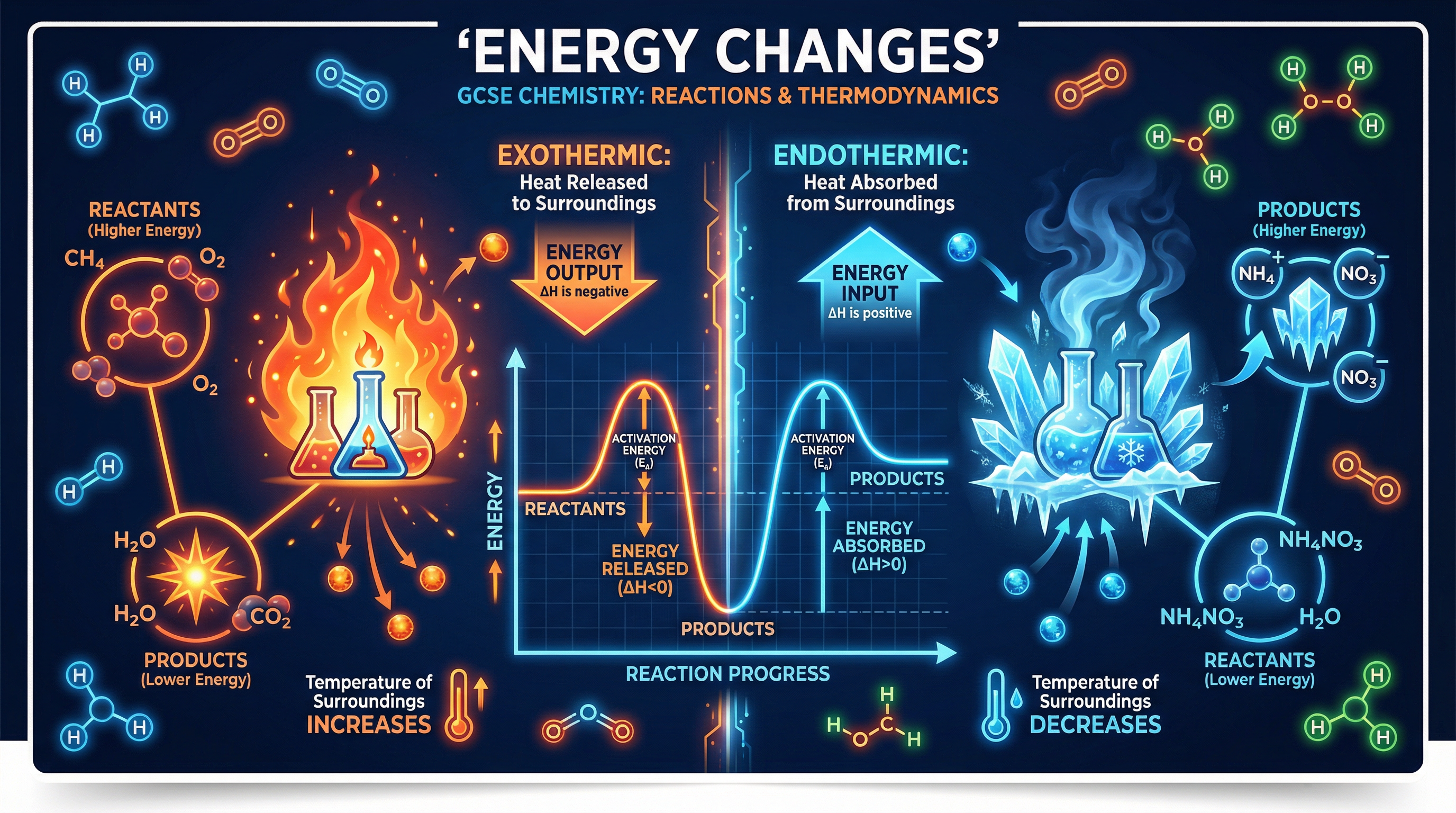
An **exothermic reaction** is one that transfers energy *to* the surroundings. Because energy is leaving the system and entering the surroundings, the temperature of the surroundings increases.
*Why does this happen?* During a chemical reaction, old bonds are broken and new bonds are formed. In an exothermic reaction, the energy released when new bonds are formed is *greater* than the energy required to break the old bonds. The 'spare' energy is released as heat.
**Example**: Combustion of methane (CH₄ + 2O₂ → CO₂ + 2H₂O). When you light a Bunsen burner, you are performing an exothermic reaction. The heat you feel is the energy being transferred to the surroundings.
An **endothermic reaction** is one that takes in energy *from* the surroundings. Because energy is entering the system from the surroundings, the temperature of the surroundings decreases.
*Why does this happen?* In an endothermic reaction, the energy required to break the old bonds is *greater* than the energy released when new bonds are formed. The system must pull in extra energy from its environment to make the reaction happen.
**Example**: The reaction between citric acid and sodium hydrogencarbonate. If you mix these in a test tube, the tube will feel freezing cold to the touch as it absorbs heat from your hand.
### Concept 2: Reaction Profiles
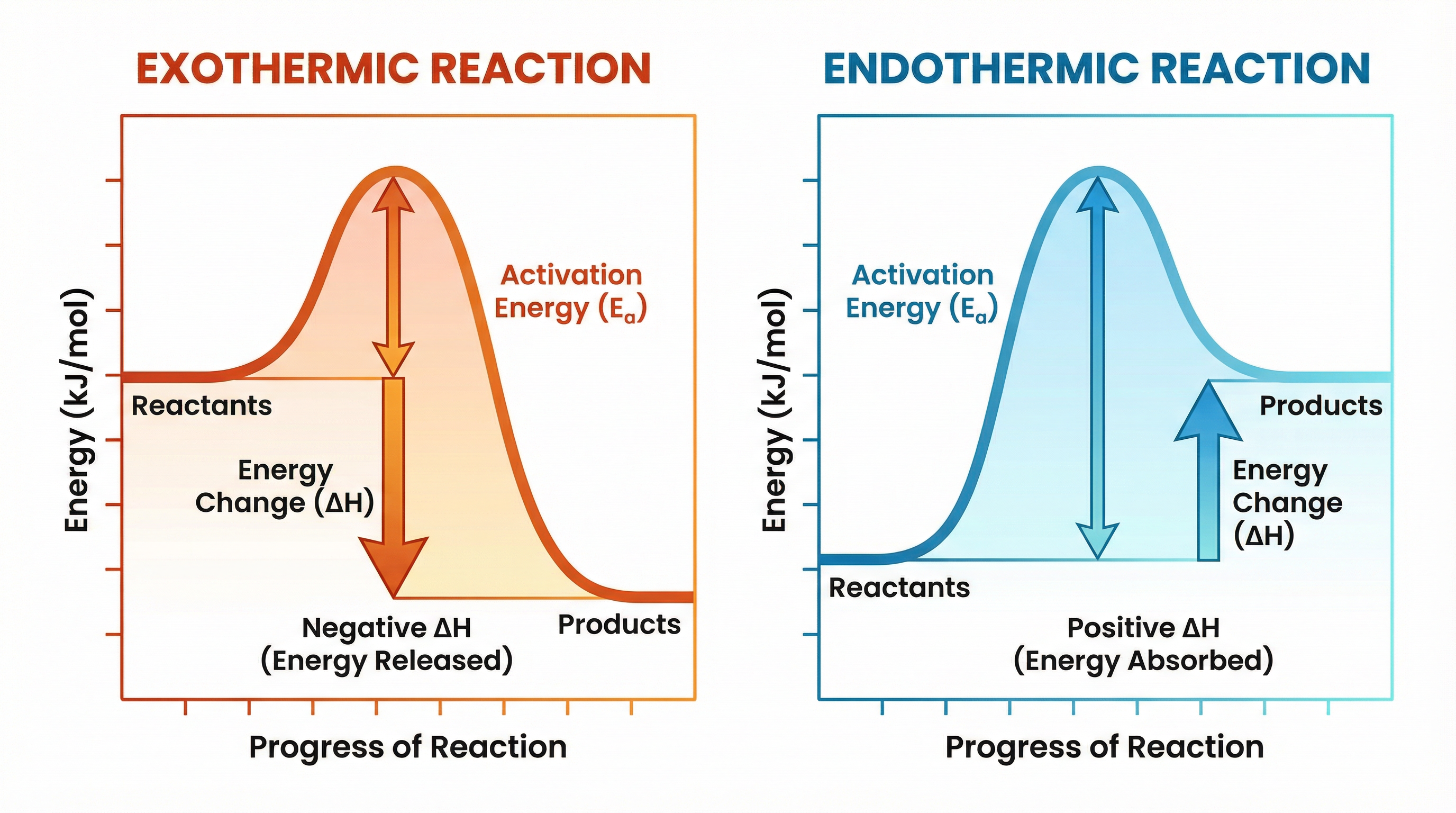
Reaction profiles (or energy level diagrams) are graphs that show the relative energies of reactants and products, the activation energy, and the overall energy change of a reaction.
1. **Reactants and Products**: The horizontal lines represent the energy of the chemicals.
2. **Activation Energy (Ea)**: This is the minimum amount of energy that particles must have to react when they collide. It is represented by the 'hump' or curve on the graph. **Crucial Examiner Tip**: The activation energy arrow MUST start from the reactants line and go straight up to the peak of the curve.
3. **Overall Energy Change (ΔH)**: This is the difference in energy between the reactants and the products.
For an **exothermic profile**, the products are at a *lower* energy level than the reactants. The overall energy change is negative (energy has been lost to the surroundings).
For an **endothermic profile**, the products are at a *higher* energy level than the reactants. The overall energy change is positive (energy has been gained from the surroundings).
### Concept 3: Bond Energy Calculations
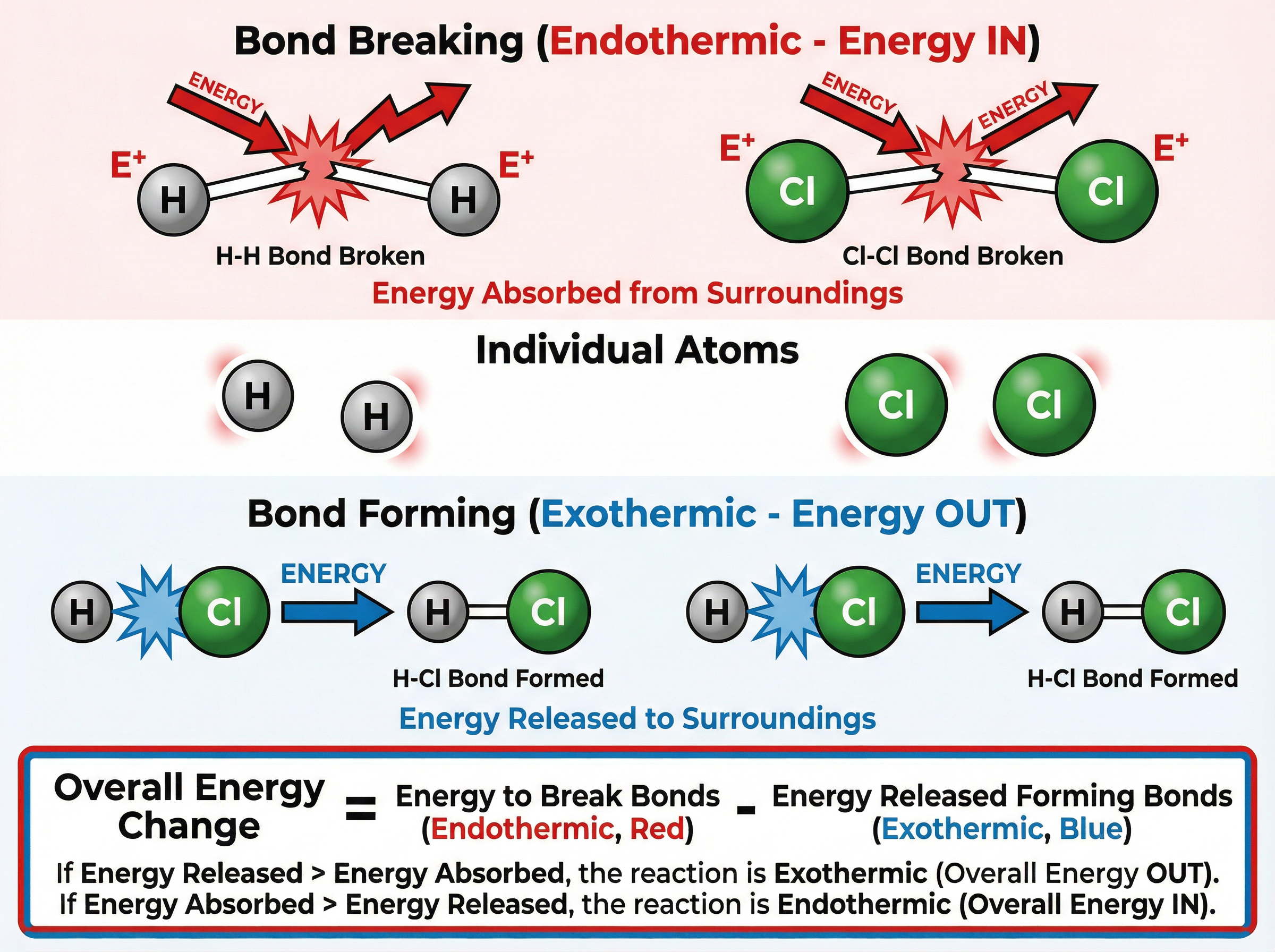
To calculate the overall energy change for a reaction, you need to use bond energies. A bond energy is the amount of energy required to break one mole of a particular covalent bond.
**The Golden Rule**:
* **B**reaking bonds is **E**ndothermic (requires energy IN).
* **M**aking bonds is **E**xothermic (releases energy OUT).
**The Formula**:
Overall Energy Change = Total Energy to Break Bonds - Total Energy Released Forming Bonds
If the final answer is negative, the reaction is exothermic. If positive, it is endothermic.
### Concept 4: Chemical Cells and Fuel Cells (Higher Tier / Chemistry Only)
Cells and batteries use chemical reactions to produce electricity. A simple cell can be made by placing two different metals (electrodes) into a liquid that conducts electricity (an electrolyte). The difference in reactivity between the two metals creates a voltage.
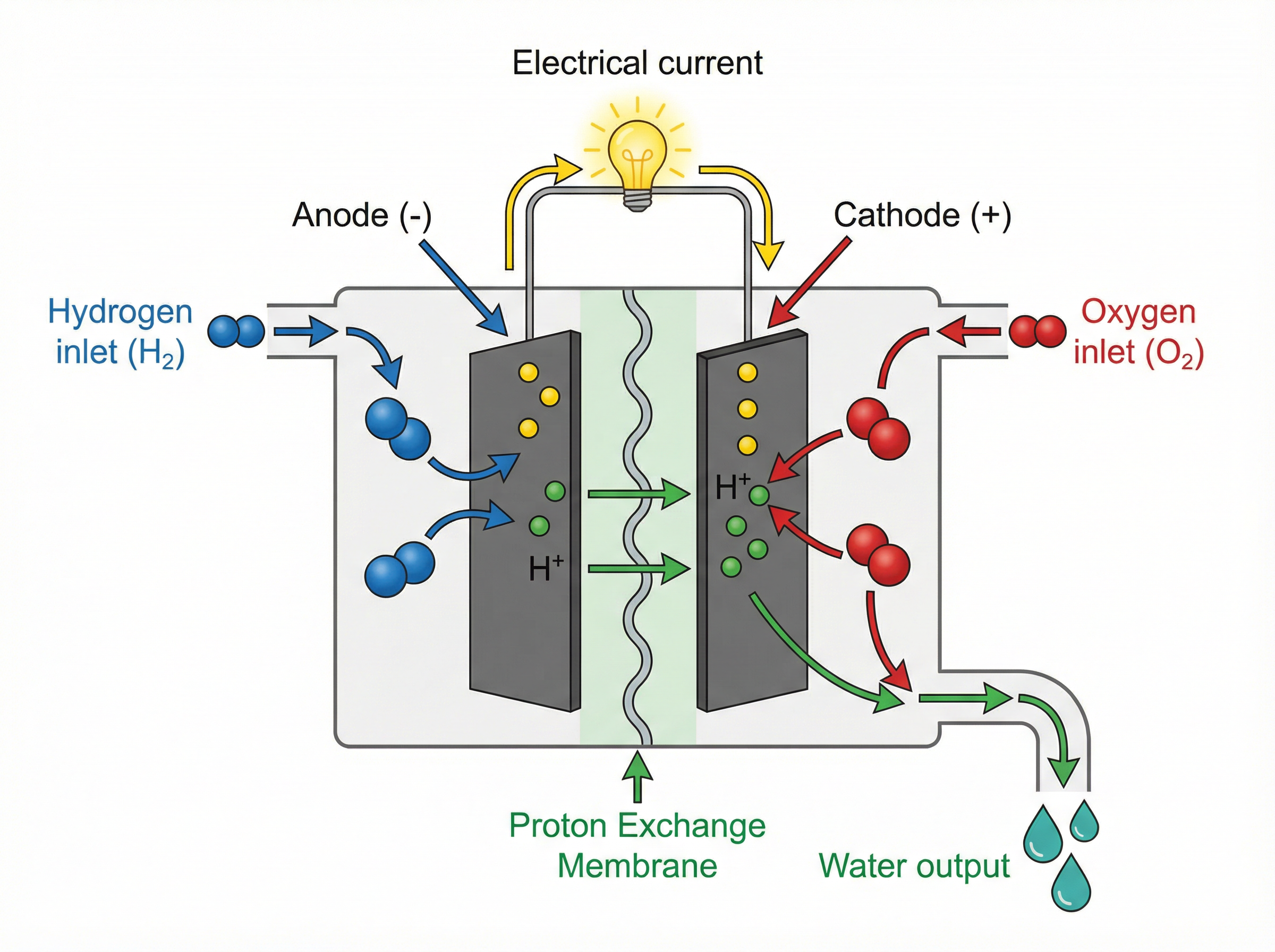
A **hydrogen fuel cell** is a special type of cell that uses hydrogen and oxygen to generate electricity.
* At the anode (negative electrode), hydrogen molecules are oxidised (lose electrons) to form hydrogen ions (H⁺).
* The electrons flow through the external circuit to the cathode, creating the electrical current.
* The H⁺ ions move through the electrolyte to the cathode.
* At the cathode (positive electrode), oxygen reacts with the H⁺ ions and the electrons to form water.
The overall reaction is simply: 2H₂ + O₂ → 2H₂O. The only waste product is water, making them an environmentally friendly alternative to fossil fuels.
---
## Listen to the Podcast
For a full audio review of this topic, listen to our 10-minute revision podcast:

---
## Mathematical/Scientific Relationships
**Overall Energy Change (ΔH) = Energy to Break Bonds - Energy Released Forming Bonds**
* **Energy to Break Bonds**: Sum of all bond energies for the reactants.
* **Energy Released Forming Bonds**: Sum of all bond energies for the products.
* **Units**: kJ/mol (kilojoules per mole).
* **Sign**: A negative (-) result means exothermic. A positive (+) result means endothermic.
## Practical Applications
**Required Practical: Temperature Changes**
**Aim**: To investigate the variables that affect temperature changes in reacting solutions (e.g., acid plus alkali).
**Method**:
1. Measure 30 cm³ of dilute hydrochloric acid and transfer it to a polystyrene cup.
2. Stand the cup inside a beaker. This provides insulation and stability.
3. Use a thermometer to measure the initial temperature of the acid.
4. Measure 5 cm³ of sodium hydroxide solution and pour it into the cup.
5. Fit a lid with a hole on the cup and gently stir the solution with the thermometer through the hole.
6. Record the highest temperature reached.
7. Repeat steps 4-6, adding a further 5 cm³ of sodium hydroxide each time, until a total of 40 cm³ has been added.
8. Repeat the whole experiment and calculate mean maximum temperatures.
**Examiner Focus**: Examiners frequently ask *why* a polystyrene cup and lid are used. The answer is to reduce heat loss to the surroundings, ensuring the temperature reading is as accurate as possible.