Key ideas — AQA GCSE Study Guide
Exam Board: AQA | Level: GCSE
Master the fundamental principles that underpin every chemistry topic. This guide covers the essential ideas—from atomic structure and bonding to energy conservation—that examiners test across all papers.
## Overview
Welcome to the foundational core of your chemistry course. The 'Key Ideas in Chemistry' aren't just an isolated topic; they are the universal rules that govern how matter behaves. Whether you are studying organic chemistry, electrolysis, or rates of reaction, these principles apply.
Examiners love to test these concepts in synoptic questions, requiring you to link atomic structure to macroscopic properties. By mastering these key ideas, you build a mental framework that makes learning every other topic significantly easier.
Listen to our comprehensive audio guide for a deeper dive:

## Key Concepts
### Concept 1: Atomic Structure and the Periodic Table
All matter is composed of tiny particles called atoms. There are approximately 100 naturally occurring elements, each consisting of just one type of atom. The defining feature of an element is its atomic number—the number of protons in its nucleus.
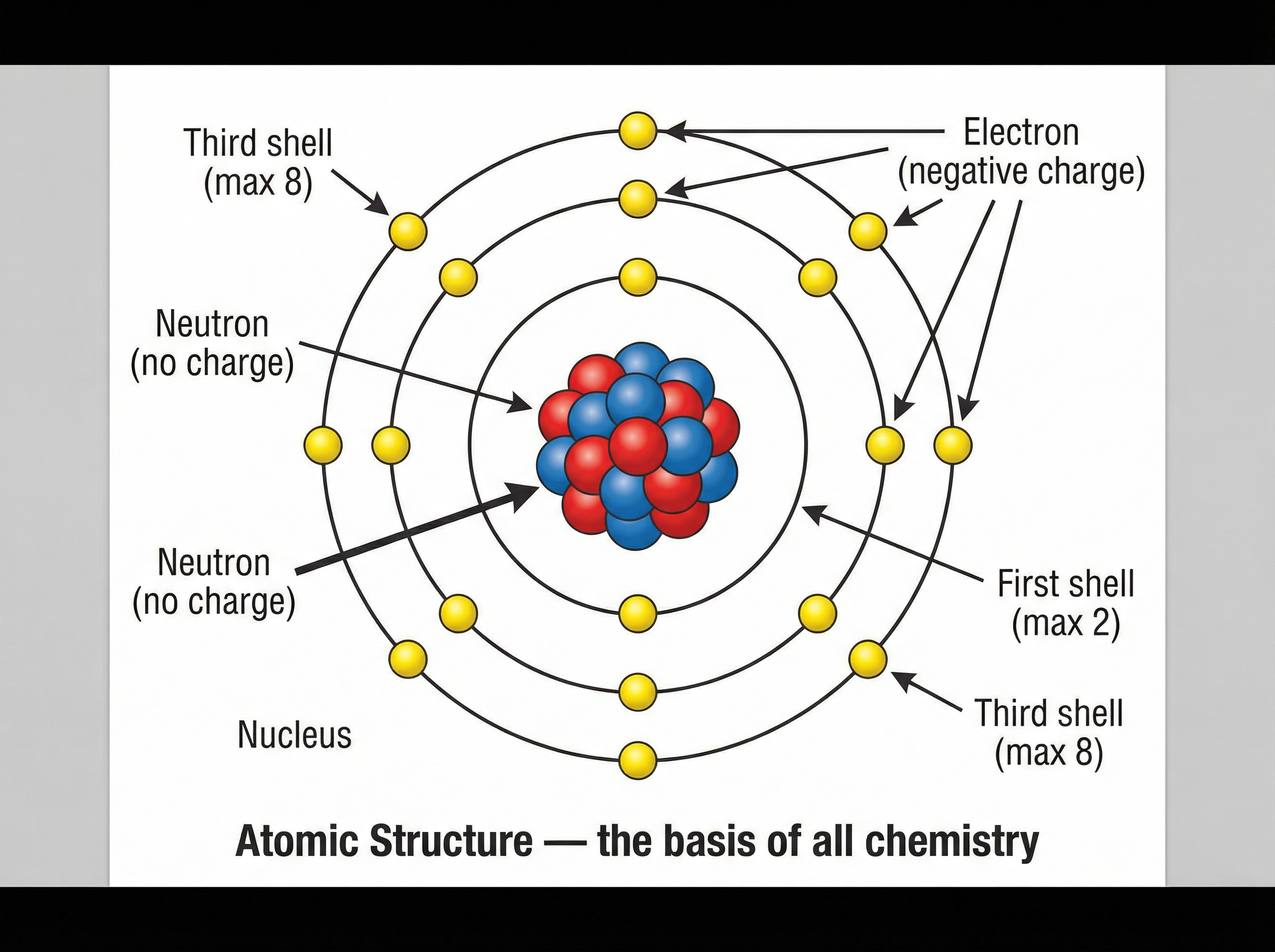
The periodic table is a masterpiece of scientific organisation. It arranges elements in order of increasing atomic number, but its true power lies in its columns (groups). Elements in the same group have the same number of electrons in their outer shell. This identical outer electron configuration is exactly why elements in the same group exhibit similar chemical properties.
**Example**: Group 1 alkali metals (Li, Na, K) all have one electron in their outer shell. Consequently, they all react vigorously with water to form alkaline solutions and hydrogen gas, and they all form 1+ ions in ionic compounds.
### Concept 2: Chemical Bonding and Structure
Atoms rarely exist in isolation. They bond to achieve a full outer electron shell, leading to more stable arrangements. There are three primary types of strong chemical bonds:
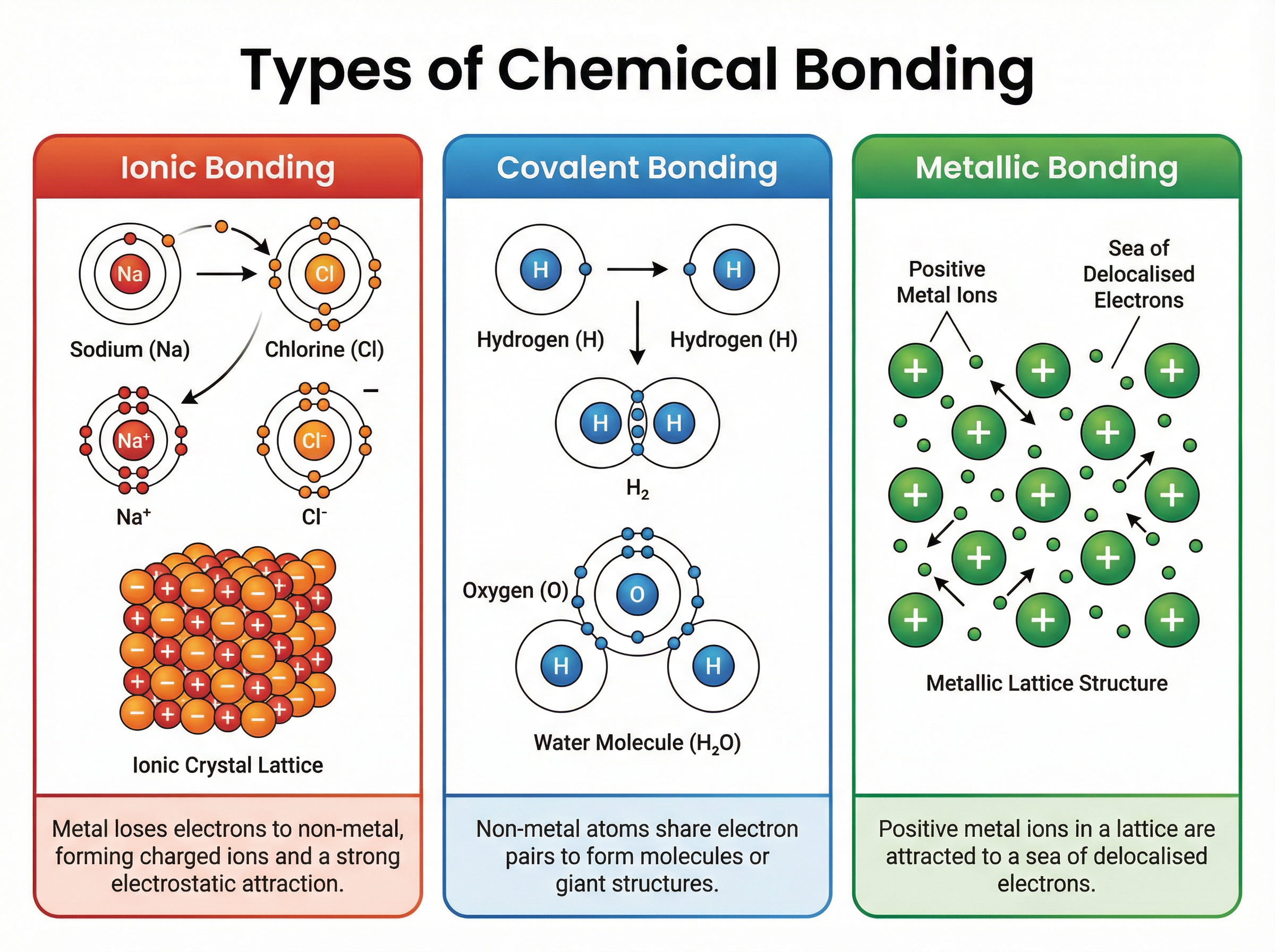
1. **Ionic Bonding**: Occurs between metals and non-metals. Electrons are transferred from the metal to the non-metal, creating oppositely charged ions that attract each other strongly in a giant ionic lattice.
2. **Covalent Bonding**: Occurs between non-metals. Atoms share pairs of electrons to achieve full outer shells. This can form simple discrete molecules (like $H_2O$) or giant covalent structures (like diamond).
3. **Metallic Bonding**: Occurs in metals. Positive metal ions form a lattice surrounded by a 'sea' of delocalised electrons, which are free to move and carry electrical charge or thermal energy.
The type of bonding dictates the macroscopic properties of the substance, such as melting point and electrical conductivity.
### Concept 3: Rates of Reaction and Collision Theory
Chemical reactions do not happen spontaneously just because reactants are mixed. For a reaction to occur, reactant particles must collide with each other, and these collisions must have sufficient energy to break existing bonds. This minimum energy threshold is called the **activation energy** ($E_a$).
Examiners will expect you to explain how changing conditions affects the rate of reaction using collision theory. Increasing temperature, concentration, or surface area leads to more frequent successful collisions. Using a catalyst provides an alternative reaction pathway with a lower activation energy.
### Concept 4: Energy Conservation in Reactions
One of the most profound laws in science is that energy cannot be created or destroyed. In chemistry, this means the total energy of the universe remains constant during a reaction.
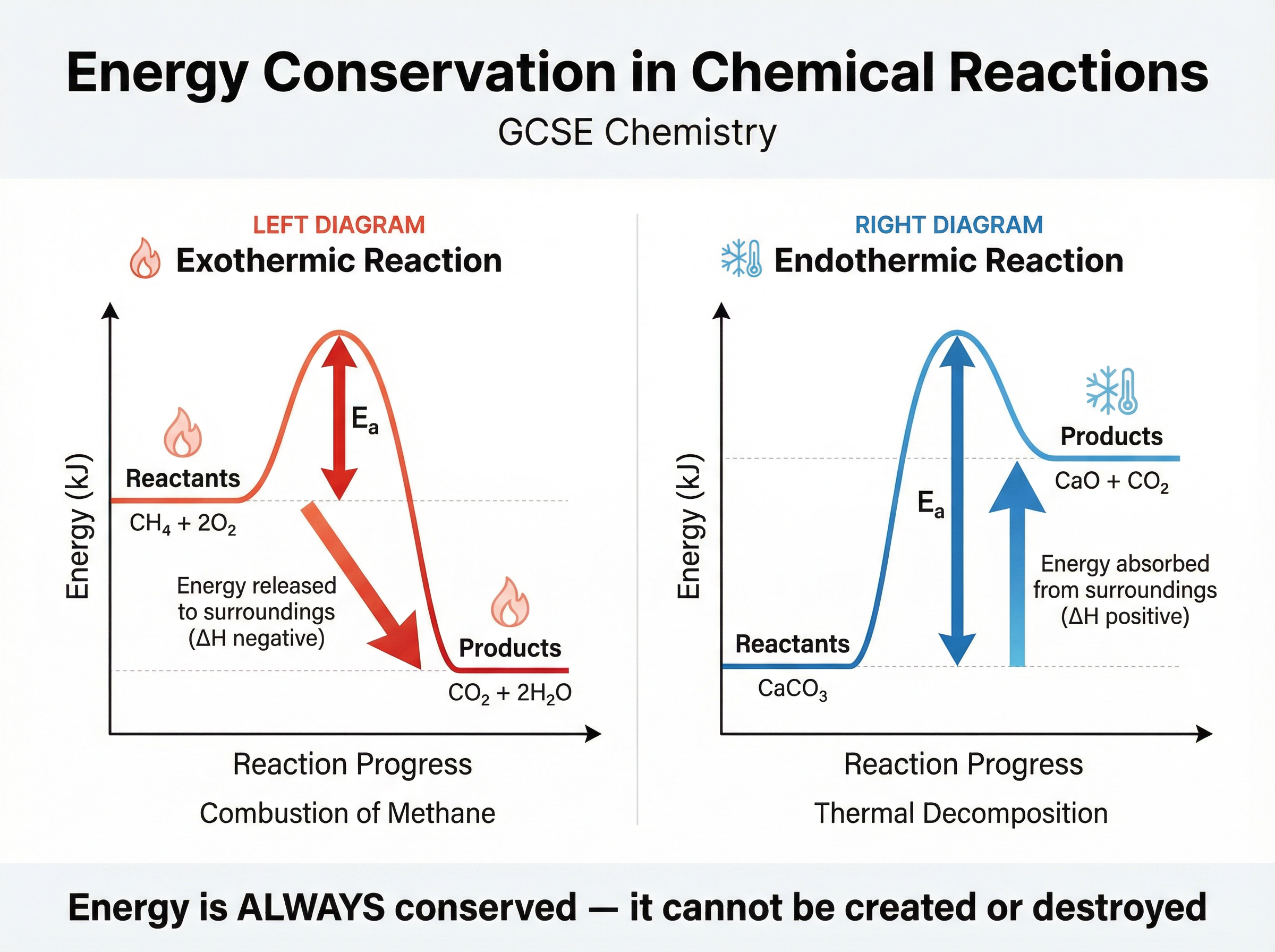
- **Exothermic reactions**: Energy is transferred to the surroundings (usually as heat). The products have less energy than the reactants. The enthalpy change ($\Delta H$) is negative.
- **Endothermic reactions**: Energy is taken in from the surroundings. The products have more energy than the reactants. The enthalpy change ($\Delta H$) is positive.
## Mathematical/Scientific Relationships
- **Atomic Number** = Number of protons (which equals the number of electrons in a neutral atom).
- **Mass Number** = Number of protons + Number of neutrons.
- **Number of Neutrons** = Mass Number - Atomic Number.
## Practical Applications
Understanding these key ideas is crucial for industrial applications. For instance, the Haber process for manufacturing ammonia relies heavily on manipulating temperature, pressure, and catalysts (reaction rates) while managing the exothermic nature of the reaction (energy conservation) to maximise yield efficiently.