The rate and extent of chemical change — AQA GCSE Study Guide
Exam Board: AQA | Level: GCSE
Master the rate and extent of chemical change, from calculating reaction speeds to predicting equilibrium shifts. This core GCSE Chemistry topic teaches you how to control chemical reactions—a vital skill for both your exams and the chemical industry.
## Overview

This topic is all about the speed of chemical reactions and how far they go before stopping. In the chemical industry, time is money. Understanding how to make reactions happen faster (the **rate** of reaction) and how to maximise the amount of product formed (the **extent** of reaction) is crucial for manufacturing everything from fertilisers to pharmaceuticals.
In this topic, you will learn how to measure the rate of a reaction, how to interpret rate graphs, and how factors like temperature, concentration, pressure, and surface area affect reaction speed. We will explain these effects using **Collision Theory**. You will also explore the role of catalysts in providing alternative reaction pathways. For Higher Tier candidates, the second half of this topic introduces reversible reactions, dynamic equilibrium, and **Le Chatelier's Principle**, which allows you to predict how changing conditions will shift the position of equilibrium.
Listen to our comprehensive audio guide for this topic below:

## Key Concepts
### Concept 1: Measuring Rate of Reaction
The rate of a chemical reaction tells us how fast the reactants are turned into products. We can measure this in two main ways: by measuring how quickly a reactant is used up, or how quickly a product is formed. For example, if a reaction produces a gas, we can measure the volume of gas produced over time using a gas syringe, or the loss in mass as the gas escapes.
**Example**: If 50 cm³ of hydrogen gas is produced in 20 seconds, the mean rate of reaction is 50 ÷ 20 = 2.5 cm³/s.
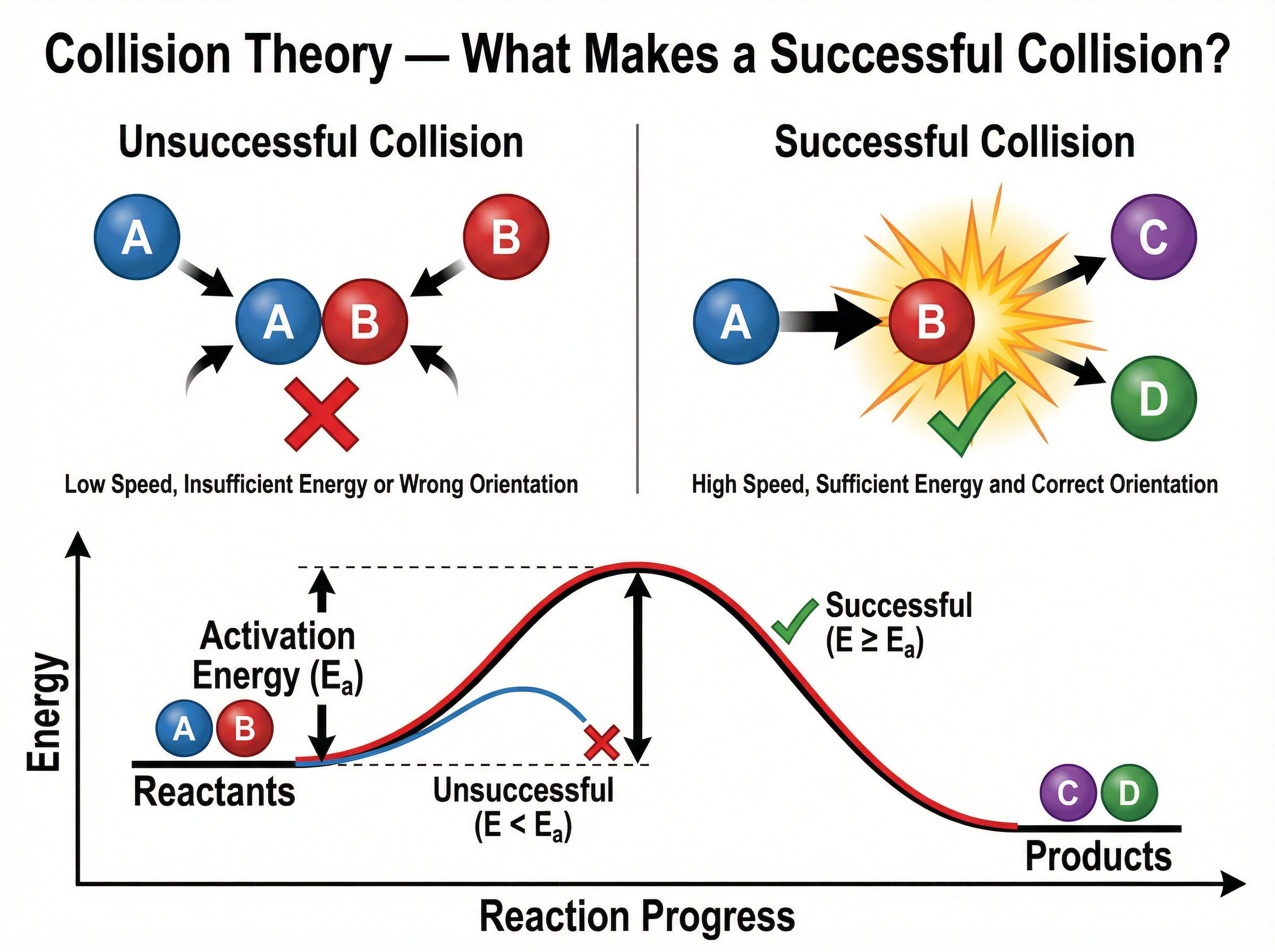
### Concept 2: Collision Theory and Activation Energy
For a chemical reaction to occur, reactant particles must collide with each other. However, not all collisions result in a reaction. A successful collision requires two things:
1. The particles must collide with the correct orientation.
2. The particles must have enough energy to break existing bonds.
The minimum amount of energy that particles must have to react when they collide is called the **activation energy**. If particles collide with less energy than the activation energy, they simply bounce off each other without reacting.
### Concept 3: Factors Affecting Rate of Reaction
Four main factors affect the rate of a chemical reaction. When explaining these in an exam, you must always refer to the **frequency of successful collisions**.
- **Temperature**: Increasing the temperature gives the particles more kinetic energy. They move faster, resulting in a higher frequency of collisions. More importantly, a higher proportion of the particles have energy greater than or equal to the activation energy, so a higher proportion of collisions are successful.
- **Concentration/Pressure**: Increasing the concentration of a solution, or the pressure of a reacting gas, means there are more particles in the same volume. This leads to a higher frequency of collisions.
- **Surface Area**: Breaking a solid reactant into smaller pieces increases its surface area to volume ratio. This means more particles are exposed at the surface and available to react, leading to a higher frequency of collisions.
- **Catalysts**: A catalyst is a substance that increases the rate of a chemical reaction without being used up itself. It does this by providing an alternative reaction pathway that has a lower activation energy. Because the activation energy is lower, a higher proportion of the reactant particles have enough energy to react when they collide.
### Concept 4: Reversible Reactions and Dynamic Equilibrium (HT)
Some chemical reactions are reversible, meaning the products can react to produce the original reactants. This is represented by a double-headed arrow (⇌). If a reversible reaction is carried out in a closed system (where no reactants or products can escape), it will eventually reach **dynamic equilibrium**.
At dynamic equilibrium, the forward and reverse reactions are occurring at exactly the same rate. As a result, the concentrations of the reactants and products remain constant.
### Concept 5: Le Chatelier's Principle (HT)
Le Chatelier's Principle states that if a change is made to the conditions of a system at equilibrium, the system will respond to counteract that change. We can use this to predict how the position of equilibrium will shift.
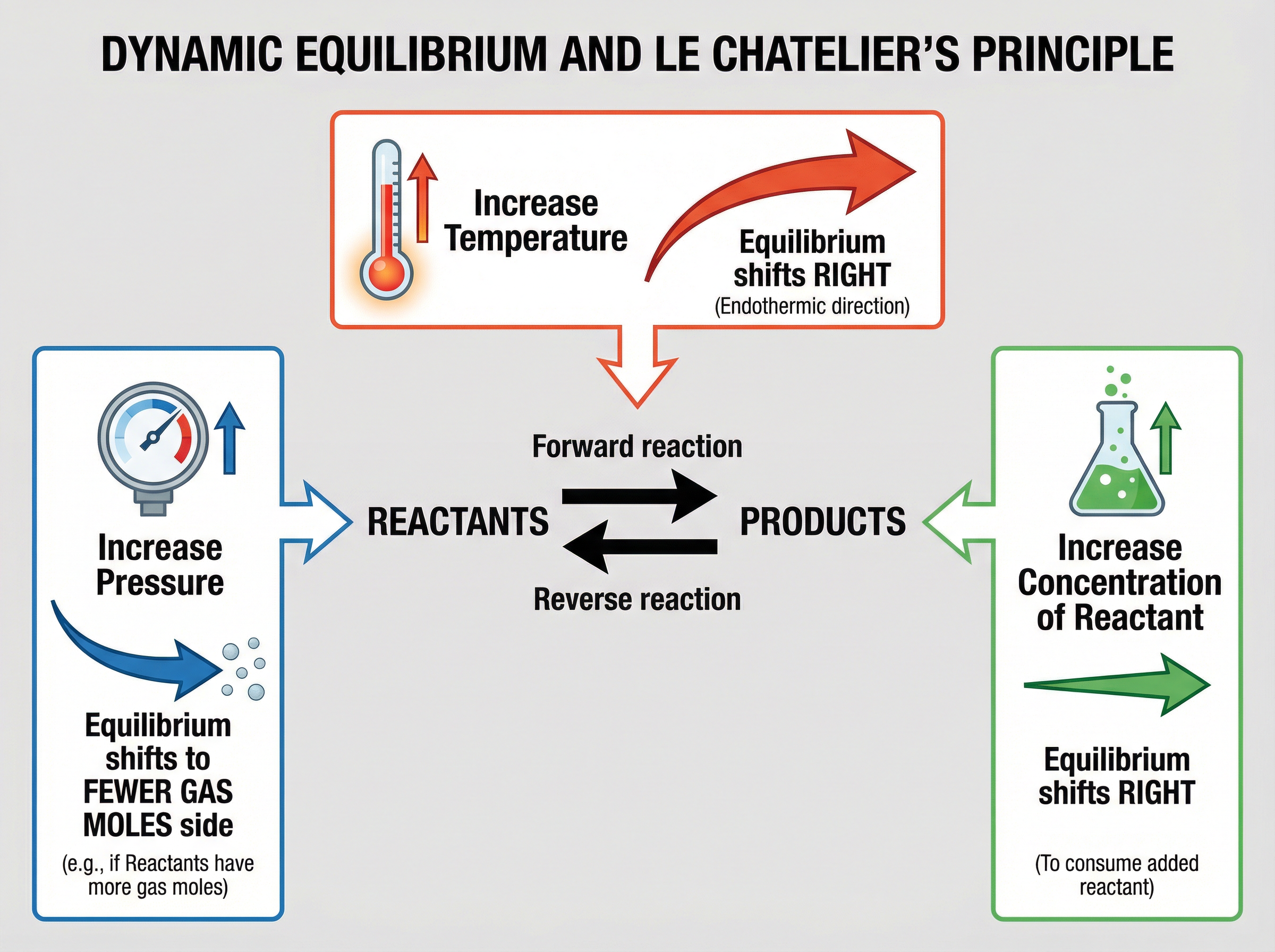
- **Changing Concentration**: If you increase the concentration of a reactant, the system will try to decrease it by making more product (equilibrium shifts to the right).
- **Changing Temperature**: If you increase the temperature, the system will try to decrease it by favouring the endothermic reaction. If you decrease the temperature, the system favours the exothermic reaction.
- **Changing Pressure (for gases)**: If you increase the pressure, the system will try to decrease it by shifting to the side of the equation with the fewest moles of gas molecules.
## Mathematical/Scientific Relationships
**Mean Rate of Reaction**
- Formula: `Mean rate of reaction = quantity of reactant used ÷ time taken`
- Formula: `Mean rate of reaction = quantity of product formed ÷ time taken`
- Units: g/s (mass), cm³/s (volume), or mol/s (moles - HT only)
- *Must memorise*
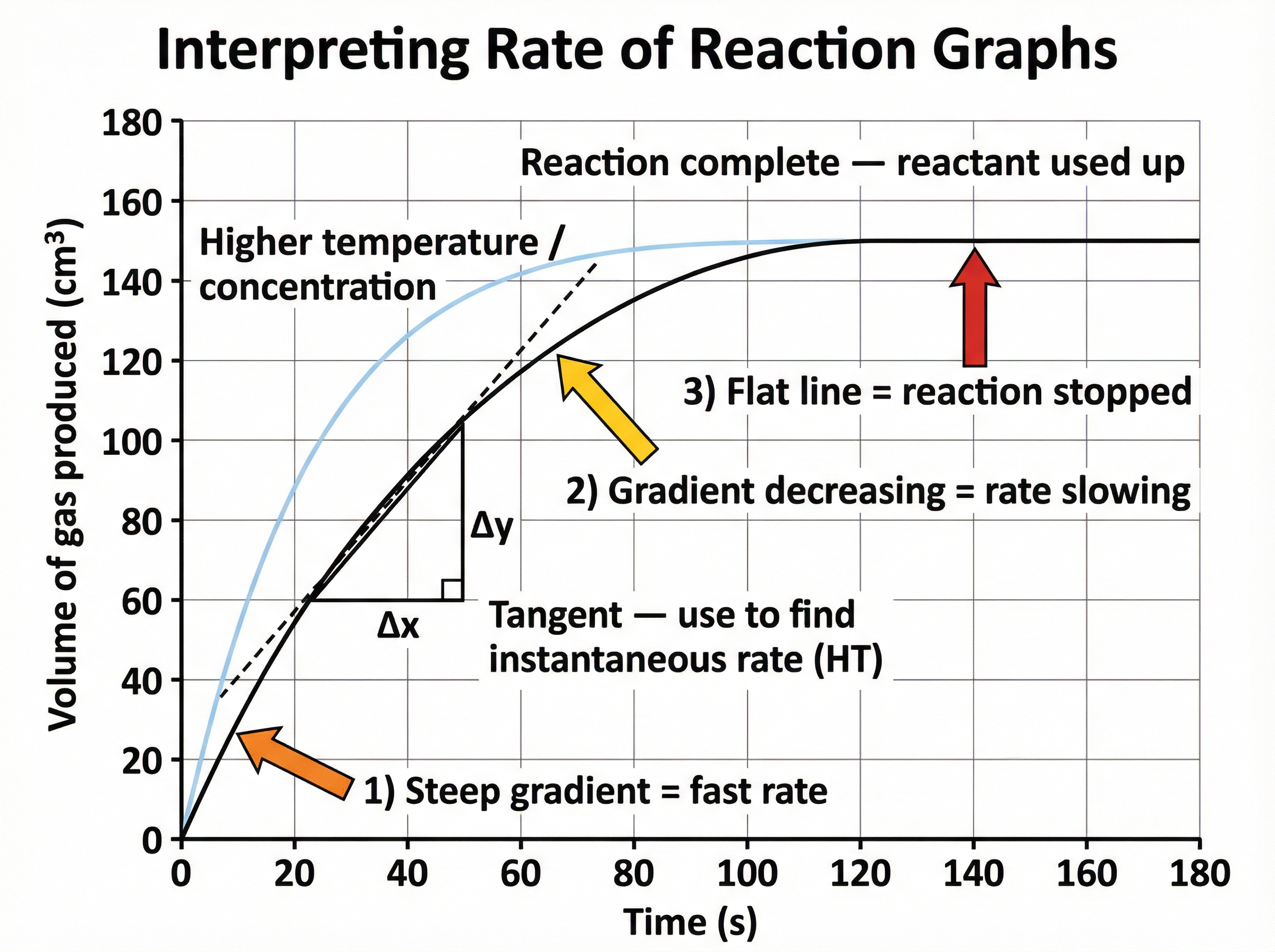
**Calculating Instantaneous Rate from a Graph (HT)**
To find the rate at a specific time, you must draw a tangent to the curve at that time and calculate its gradient.
- Formula: `Gradient = change in y ÷ change in x`
## Practical Applications
**Required Practical: Investigating the effect of concentration on rate of reaction**
There are two common methods to investigate this:
1. **Measuring the decreasing light transmission (Disappearing Cross)**:
- *Method*: React sodium thiosulfate with hydrochloric acid in a conical flask placed over a black cross. The reaction produces solid sulfur, making the solution turn cloudy.
- *Measurement*: Time how long it takes for the cross to disappear.
- *Common Error*: Judging exactly when the cross disappears is subjective and varies between individuals.
2. **Measuring the volume of gas produced**:
- *Method*: React magnesium ribbon with hydrochloric acid in a conical flask attached to a gas syringe (or inverted measuring cylinder in a water trough).
- *Measurement*: Record the volume of hydrogen gas produced at regular time intervals.
- *Common Error*: Gas escaping before the bung is placed on the flask.
In industry, the **Haber Process** is used to manufacture ammonia from nitrogen and hydrogen. This is a reversible reaction: N₂(g) + 3H₂(g) ⇌ 2NH₃(g). The forward reaction is exothermic. Industrial chemists must carefully choose the temperature and pressure to balance the rate of reaction with the yield of ammonia (the extent of reaction) while keeping costs down.