Using resources — AQA GCSE Study Guide
Exam Board: AQA | Level: GCSE
This topic covers how we sustainably use Earth's resources, from making water safe to drink, to extracting metals without traditional mining, to evaluating the environmental impact of products. It's heavily tested in exams because it links core chemistry to real-world environmental challenges.

## Overview
Welcome to **Using Resources** (Topic 4.10). This section is fundamentally about sustainability: how we meet the needs of the current generation without compromising the ability of future generations to meet their own needs. In Chemistry, this means understanding the difference between finite and renewable resources, and finding ways to reduce our reliance on finite ones.
This topic is crucial because examiners love to test your ability to apply chemical principles to real-world scenarios. You won't just be balancing equations; you'll be evaluating the environmental impact of different processes, comparing metal extraction methods, and understanding the compromises made in industrial processes like the Haber process. It connects deeply to your knowledge of bonding, rates of reaction, and quantitative chemistry.
Expect a mix of short recall questions (e.g., "State the conditions for the Haber process") and longer, extended-response questions (e.g., "Evaluate the use of phytomining compared to traditional smelting").
## Key Concepts
### Concept 1: Potable Water vs. Pure Water
A fundamental distinction that examiners test every year is the difference between **potable water** and **pure water**.
**Pure water** contains only $H_2O$ molecules and absolutely nothing else. It boils at exactly $100^{\circ}C$.
**Potable water** is water that is safe to drink. It contains dissolved salts and minerals, but at levels low enough that they do not cause harm, and it is free from harmful microbes.
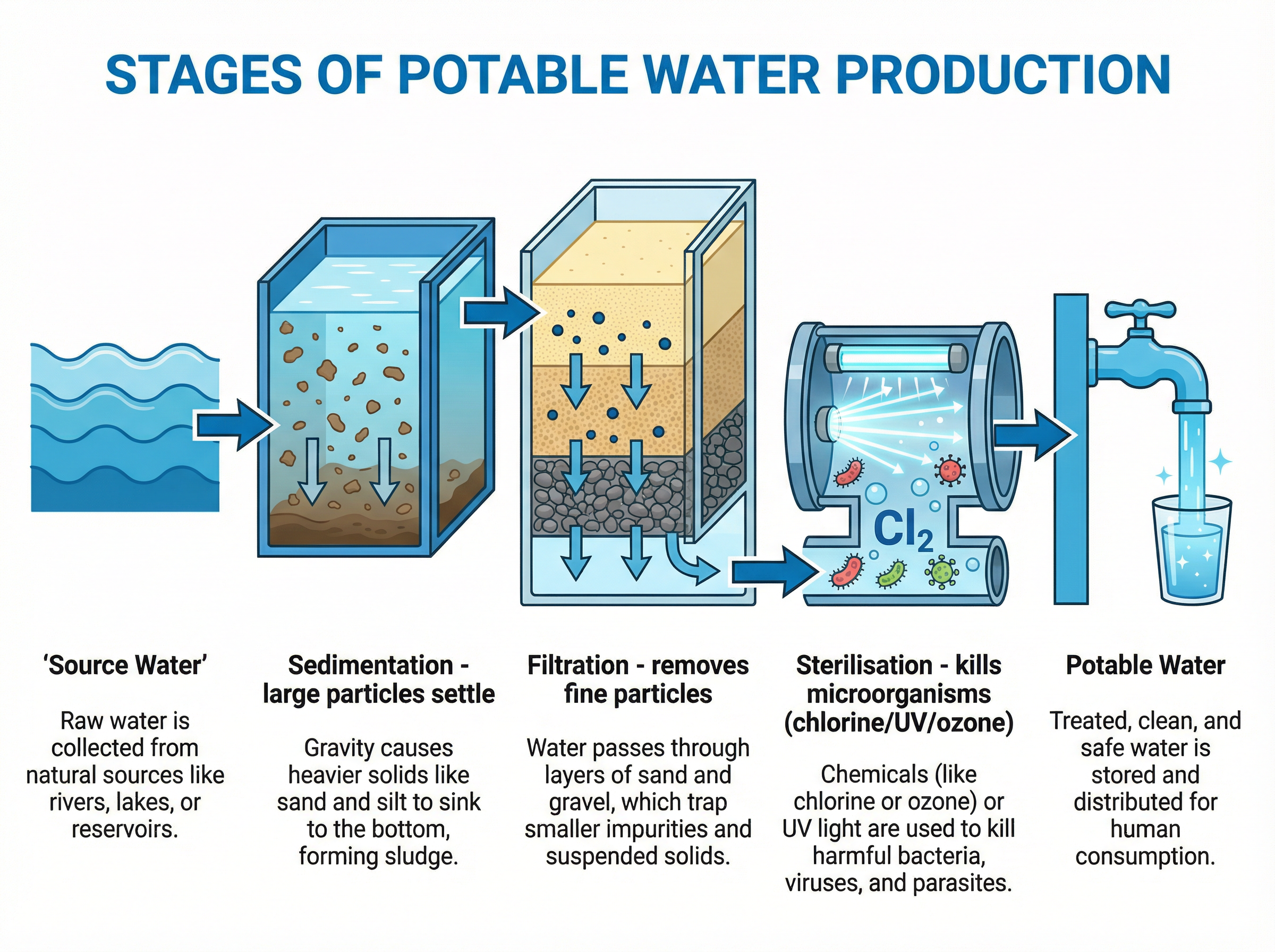
To produce potable water from fresh water sources (like lakes or rivers), three main stages are used:
1. **Sedimentation**: The water is stored in a large reservoir. Gravity causes larger solid particles (like grit and sand) to settle at the bottom.
2. **Filtration**: The water is passed through filter beds made of sand and gravel. This removes smaller, suspended solid particles.
3. **Sterilisation**: The water is treated to kill harmful microorganisms (bacteria and viruses). This is usually done by bubbling chlorine gas through the water, but ozone or ultraviolet (UV) light can also be used.
If fresh water is scarce, potable water can be produced from sea water by **desalination**. This can be done via distillation (boiling the water and condensing the steam) or reverse osmosis (forcing water through a membrane that traps the salt). Both methods require enormous amounts of energy, making them very expensive.
### Concept 2: Alternative Metal Extraction
The Earth's supply of metal ores (especially copper) is limited. Traditional extraction involves mining high-grade ores and smelting them (heating with carbon), which requires huge amounts of energy and damages the landscape. To be more sustainable, we now use biological methods to extract metals from **low-grade ores** (ores containing only a small percentage of the metal).
**Phytomining** uses plants to absorb metal compounds from the soil. The plants are harvested and burned, producing an ash that contains a high concentration of the metal compounds.
**Bioleaching** uses bacteria to break down low-grade ores. The bacteria produce a solution called a **leachate**, which contains the metal ions.
In both cases, the metal must still be extracted from the compound or leachate. For copper, this is usually done by displacement using scrap iron (since iron is more reactive than copper) or by electrolysis.
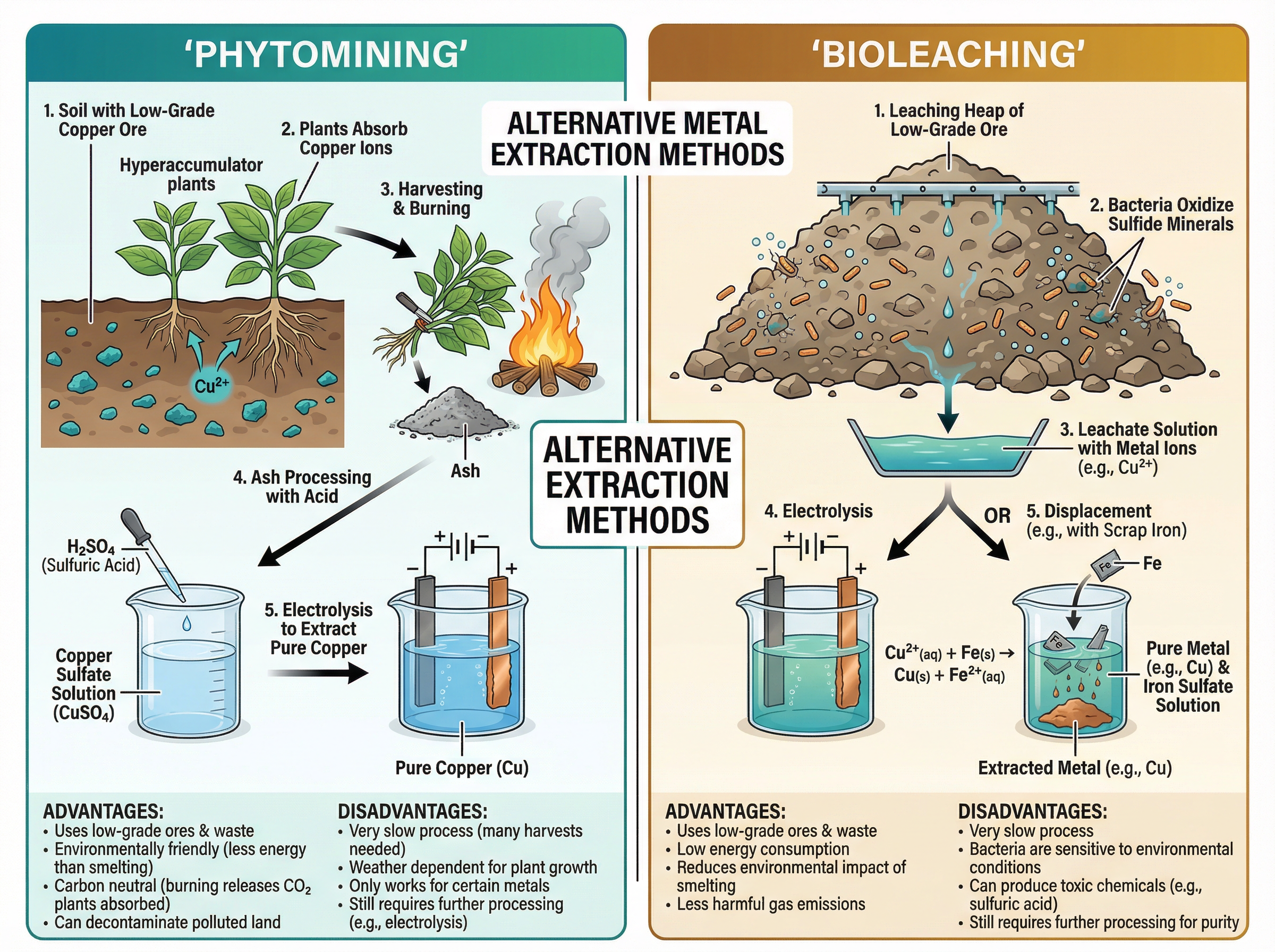
**Why this works**: These methods are much slower than smelting, but they allow us to economically extract metals from waste rock or low-grade ores, reducing the need to mine fresh ore and saving vast amounts of energy.
### Concept 3: Life Cycle Assessments (LCAs)
A Life Cycle Assessment (LCA) evaluates the environmental impact of a product across its entire lifespan. The four stages are:
1. **Extracting and processing raw materials**: e.g., mining ores, drilling for oil. This uses energy and damages habitats.
2. **Manufacturing and packaging**: e.g., energy used in factories, chemical waste produced.
3. **Use and operation during its lifetime**: e.g., emissions from driving a car, water used by a washing machine.
4. **Disposal at the end of its useful life**: e.g., space taken up in landfill, energy used to transport waste, pollution from incineration.
**Examiner Insight**: Examiners frequently ask you to compare the LCAs of two products (like plastic vs. paper bags). The key is to recognise that LCAs are not entirely objective. While we can easily measure the energy used or water consumed, assigning a value to the visual impact of a mine or the environmental damage of plastic waste involves subjective **value judgements**.
### Concept 4: Corrosion and its Prevention
Corrosion is the destruction of materials by chemical reactions with substances in the environment. The most common example is **rusting**, which specifically refers to the corrosion of iron (and its alloy, steel).
For iron to rust, **both oxygen and water must be present**. The chemical equation is:
$Iron + Oxygen + Water \rightarrow Hydrated Iron(III) Oxide$
We can prevent corrosion by applying a coating that acts as a physical barrier, such as greasing, painting, or electroplating.
Another method is **sacrificial protection**. A more reactive metal (like zinc or magnesium) is attached to the iron. Because it is more reactive, the water and oxygen react with the sacrificial metal instead of the iron. **Galvanising** is a specific type of sacrificial protection where iron is coated in a layer of zinc.
### Concept 5: The Haber Process
The Haber process is used to manufacture ammonia ($NH_3$), which is essential for producing nitrogen-based fertilisers.
The raw materials are:
* **Nitrogen**: Extracted from the air.
* **Hydrogen**: Obtained from natural gas (methane).
The reaction is reversible:
$N_2(g) + 3H_2(g) \rightleftharpoons 2NH_3(g)$ (The forward reaction is exothermic)
The conditions used are:
* **Temperature**: $450^{\circ}C$
* **Pressure**: 200 atmospheres
* **Catalyst**: Iron
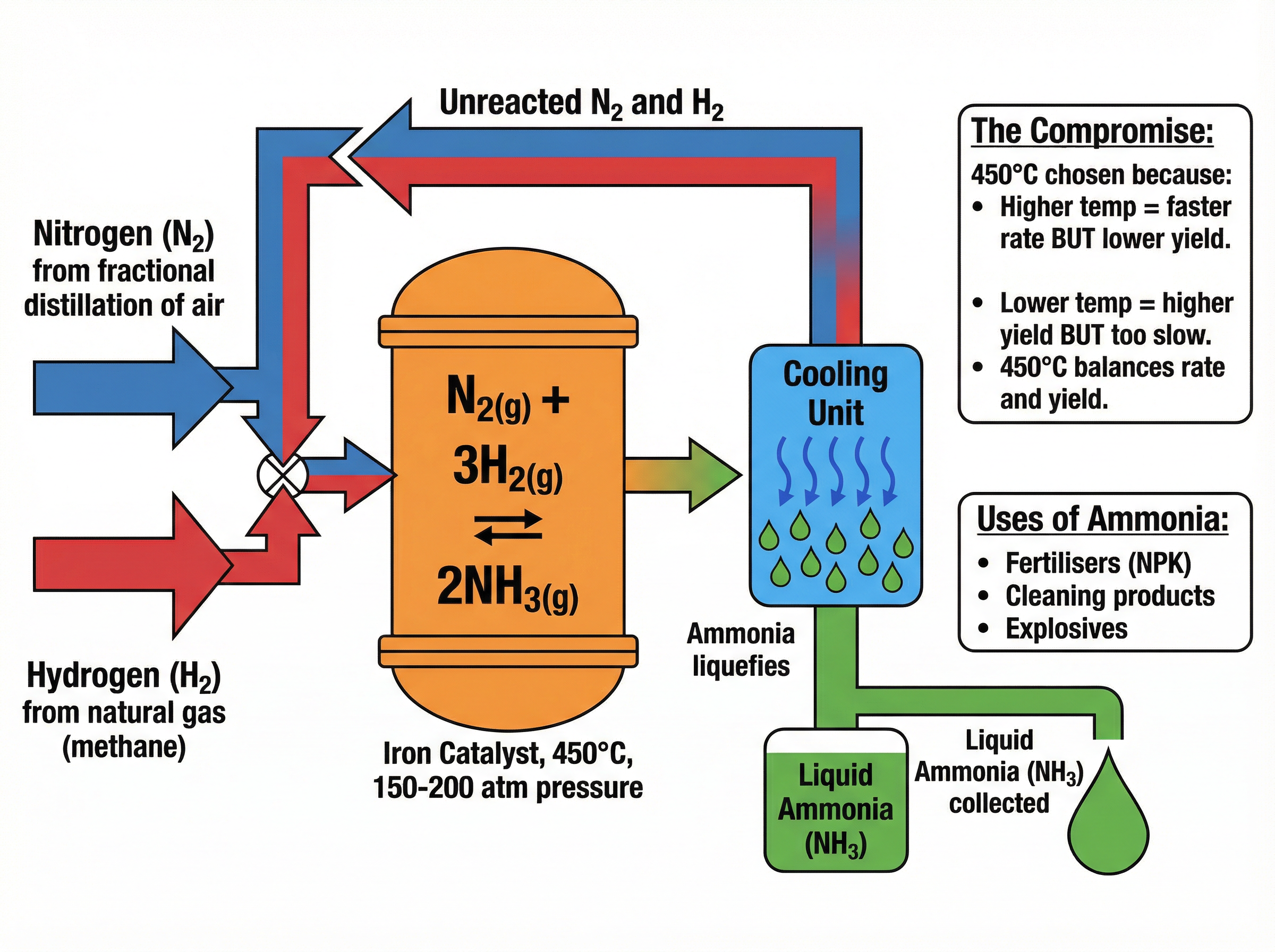
**The Compromise Temperature**: This is a classic exam question. Because the forward reaction is exothermic, a *lower* temperature would shift the equilibrium to the right, giving a *higher yield* of ammonia. However, a lower temperature means the reaction would be too slow. $450^{\circ}C$ is a compromise that provides a reasonable yield at a fast enough rate.
**The Pressure**: A higher pressure favours the forward reaction (as there are 4 moles of gas on the left and 2 on the right), increasing the yield. However, extremely high pressures are dangerous and expensive to maintain, so 200 atmospheres is used as an economic compromise.
## Mathematical/Scientific Relationships
* **Haber Process Equation**: $N_2(g) + 3H_2(g) \rightleftharpoons 2NH_3(g)$ (Must memorise)
* **Rusting Equation**: $Iron + Oxygen + Water \rightarrow Hydrated Iron(III) Oxide$ (Must memorise)
* **Displacement of Copper**: $Cu^{2+}(aq) + Fe(s) \rightarrow Cu(s) + Fe^{2+}(aq)$ (Must memorise)
## Practical Applications
* **Fertilisers (NPK)**: Formulated fertilisers contain Nitrogen (N), Phosphorus (P), and Potassium (K) to improve agricultural productivity. Ammonia from the Haber process is reacted with acids to make ammonium salts (like ammonium nitrate) for these fertilisers.
* **Recycling**: Metals, glass, building materials, clay ceramics, and most plastics can be recycled. This reduces the use of finite resources, energy consumption, and environmental impacts compared to extracting new materials.
## Audio Revision
Listen to our 10-minute podcast episode covering all the key concepts, common mistakes, and exam tips for this topic.
