Chemical changes — Edexcel GCSE Study Guide
Exam Board: Edexcel | Level: GCSE
Master the fundamental properties of acids, alkalis, and neutralisation reactions. This topic is heavily examined every year and forms the foundation for understanding complex chemical processes and quantitative analysis.
## Overview
Chemical changes involving acids, alkalis, and salts form the backbone of GCSE Chemistry. This topic explores the fundamental nature of substances at the particle level, specifically focusing on the behaviour of hydrogen ($H^+$) and hydroxide ($OH^-$) ions in aqueous solutions. Understanding these concepts is crucial because they connect deeply with quantitative chemistry, electrolysis, and chemical analysis. Examiners frequently test this area through required practicals on salt preparation and multi-step calculations involving concentration and pH. By mastering the core principles of dissociation, neutralisation, and the logarithmic nature of the pH scale, candidates can secure significant marks across both standard and extended response questions.

## Key Concepts
### Concept 1: Acids and Alkalis at the Particle Level
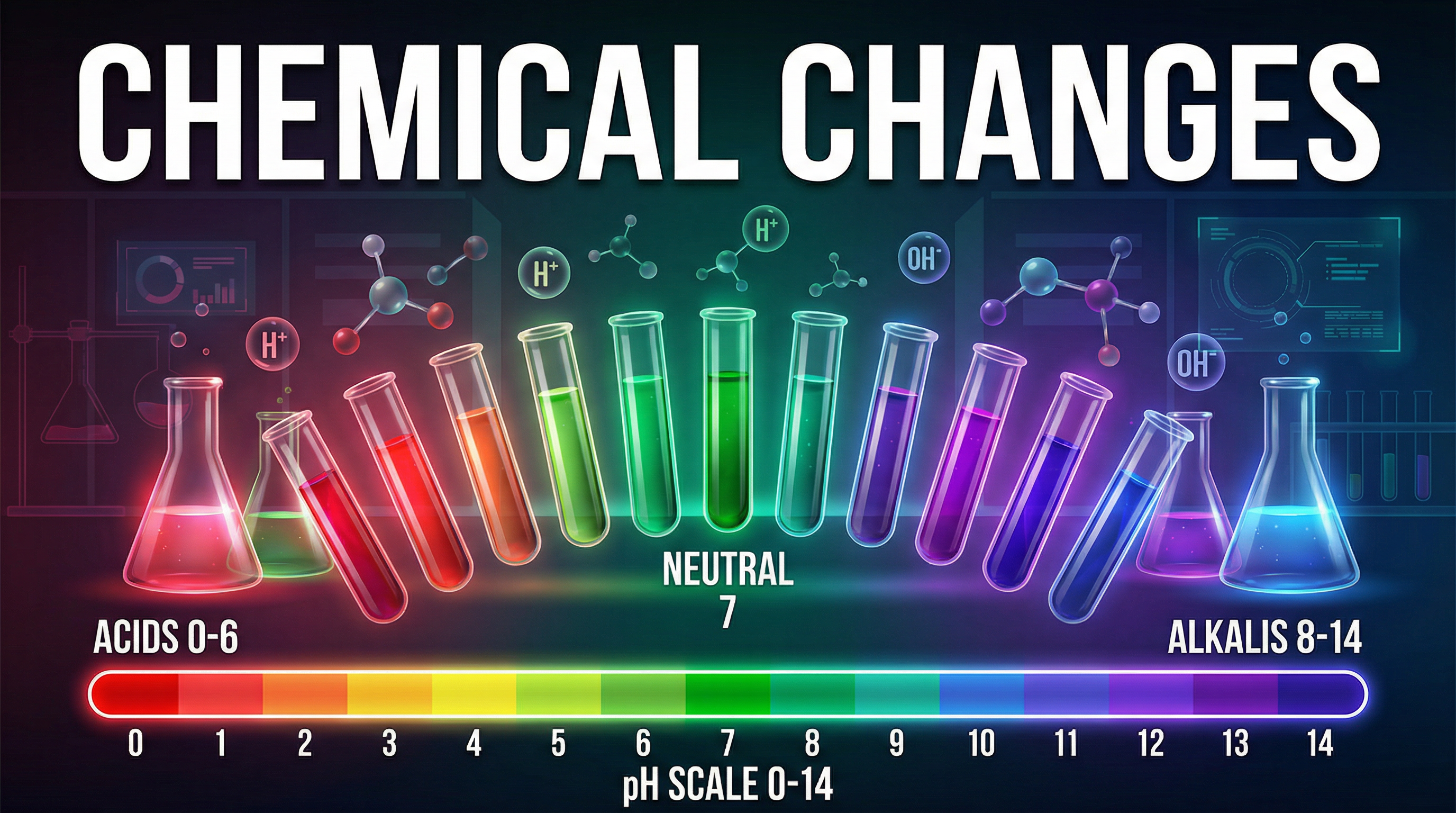
The defining characteristic of an acid in aqueous solution is its ability to produce hydrogen ions ($H^+$). Conversely, an alkali is a soluble base that produces hydroxide ions ($OH^-$) in water. The concentration of these ions determines the position of a substance on the pH scale.
**Example**: When hydrogen chloride gas dissolves in water, it dissociates to form hydrochloric acid: $HCl(g) \rightarrow H^+(aq) + Cl^-(aq)$.
### Concept 2: Strong vs Weak Acids
A critical distinction that candidates must master is the difference between acid strength and concentration. The strength of an acid refers to its degree of ionisation in water. Strong acids (like hydrochloric, sulfuric, and nitric acid) completely dissociate into ions. Weak acids (like ethanoic, citric, and carbonic acid) only partially dissociate, establishing an equilibrium between the intact molecules and the dissociated ions. Concentration, however, refers to the number of moles of acid per decimetre cubed ($dm^3$) of solution. It is entirely possible to have a dilute solution of a strong acid, or a concentrated solution of a weak acid.
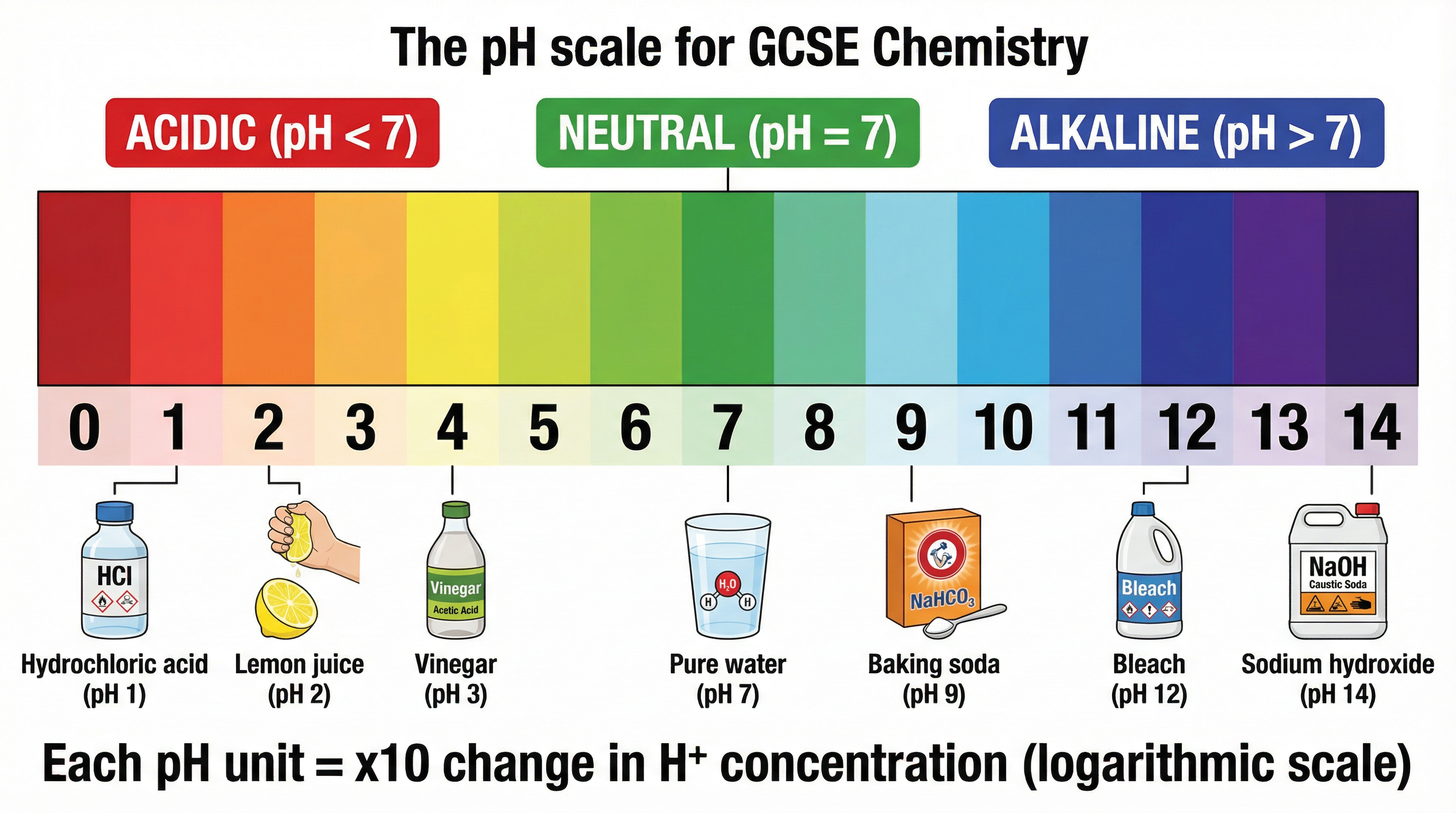
### Concept 3: The Logarithmic pH Scale
The pH scale is a measure of the hydrogen ion concentration in a solution. It ranges from 0 to 14, where 7 is neutral. Values below 7 indicate acidic solutions, while values above 7 indicate alkaline solutions. Crucially, the scale is logarithmic. This means that a decrease of one pH unit corresponds to a tenfold increase in the concentration of hydrogen ions.
**Example**: A solution with a pH of 3 has a hydrogen ion concentration that is 10 times greater than a solution with a pH of 4, and 100 times greater than a solution with a pH of 5.
### Concept 4: Neutralisation and Salt Formation
Neutralisation occurs when an acid reacts with a base (or alkali) to form a salt and water. The underlying ionic equation for all neutralisation reactions in aqueous solution is the combination of hydrogen and hydroxide ions to form water: $H^+(aq) + OH^-(aq) \rightarrow H_2O(l)$. The specific salt produced depends on the acid used (hydrochloric acid produces chlorides, sulfuric acid produces sulfates, nitric acid produces nitrates) and the positive ion from the base.
## Mathematical/Scientific Relationships
- **Ionic Equation for Neutralisation**: $H^+(aq) + OH^-(aq) \rightarrow H_2O(l)$
*This is the fundamental reaction occurring when any acid neutralises any alkali.*
- **Factor of $H^+$ Concentration Change**: $10^{-x}$ where $x$ is the change in pH.
*Used to calculate how much more or less concentrated $H^+$ ions are when pH changes.*
## Practical Applications
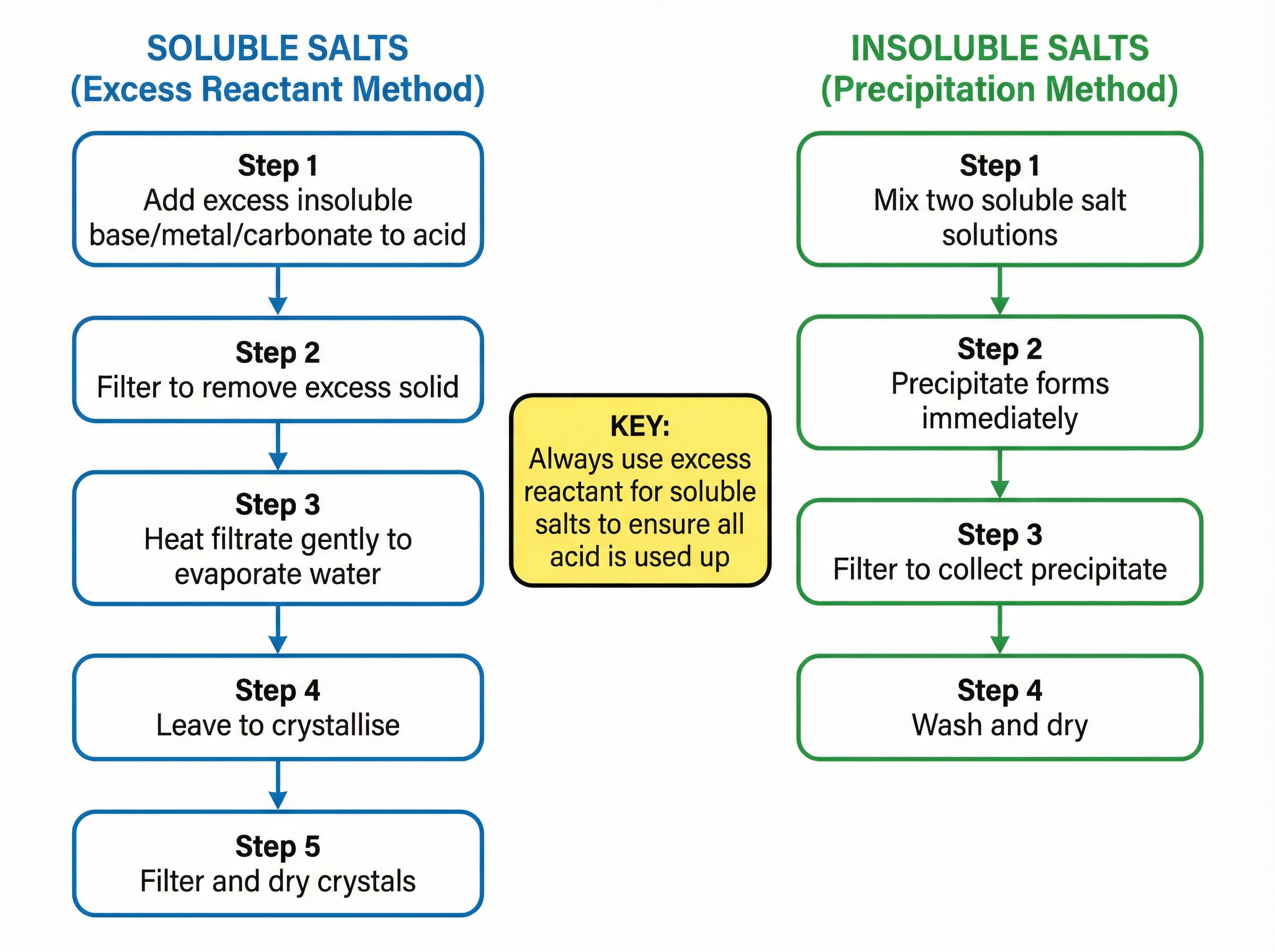
The preparation of pure, dry salts is a required practical that frequently appears in exam questions. For soluble salts from an insoluble base, the excess reactant method is used. This involves adding an excess of the solid base to the acid to ensure complete neutralisation, filtering the unreacted solid, and then gently heating the filtrate to crystallise the salt. For insoluble salts, the precipitation method is employed, where two soluble salt solutions are mixed to form an insoluble precipitate, which is then filtered, washed, and dried.