Chemical changes — Edexcel GCSE Study Guide
Exam Board: Edexcel | Level: GCSE
Master the fundamentals of electrolysis, from identifying electrode products to writing balanced half-equations. This essential topic appears in almost every chemistry paper and is a prime opportunity to secure marks through systematic understanding rather than rote memorization.
## Overview

Electrolysis is the process of using direct electrical current (DC) to drive a non-spontaneous chemical reaction, specifically the decomposition of an electrolyte. It sits at the heart of the Chemical Changes topic because it beautifully demonstrates the relationship between electrical energy and chemical bonds.
This topic is critical because examiners use it to test multiple skills simultaneously: your understanding of ionic structures, your ability to predict chemical products, and your skill in writing balanced half-equations. It frequently appears as a 4-6 mark extended response question, making it a high-yield area for revision.
## Key Concepts
### Concept 1: The Electrolysis Cell
An electrolysis setup requires an electrolyte and two electrodes connected to a direct current (DC) power supply.
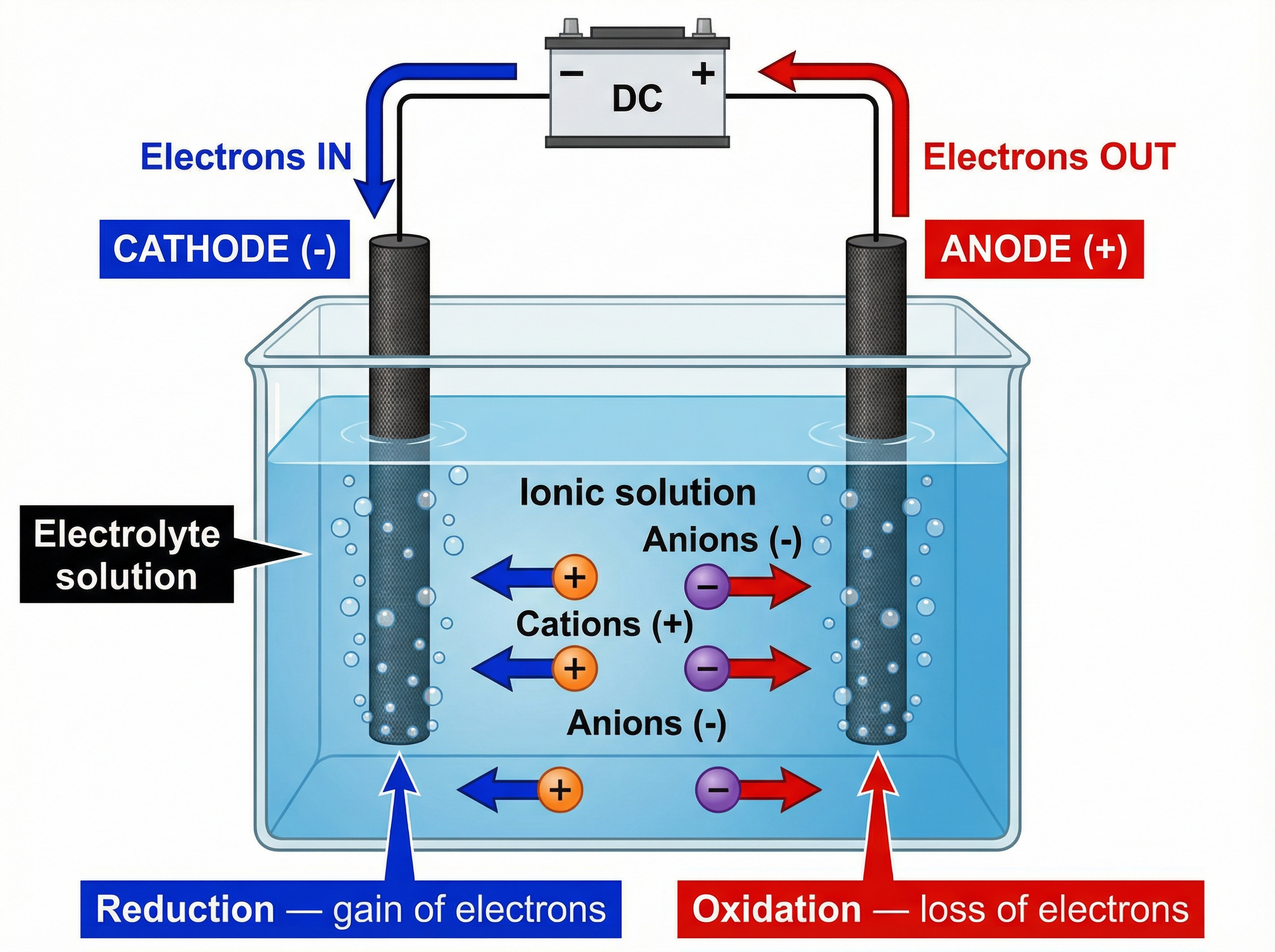
An **electrolyte** is an ionic compound that is either molten (melted) or dissolved in water (aqueous). In these states, the ions are free to move and carry charge. Solid ionic compounds cannot be electrolysed because their ions are locked in a rigid lattice.
The electrodes are conductive rods (often inert graphite or platinum) that dip into the electrolyte. The **cathode** is the negative electrode, and the **anode** is the positive electrode.
### Concept 2: Ion Migration and Discharge
When the current flows, the charged ions in the electrolyte migrate toward the oppositely charged electrodes:
- Positive ions (**cations**) move to the negative **cathode**.
- Negative ions (**anions**) move to the positive **anode**.
At the electrodes, the ions are discharged—they lose their charge to become neutral atoms or molecules. This happens through the transfer of electrons.
- At the cathode, cations *gain* electrons. This is **reduction**.
- At the anode, anions *lose* electrons. This is **oxidation**.
### Concept 3: Aqueous Solutions
Electrolysing aqueous solutions is more complex than molten compounds because water itself partially ionises into hydrogen ions (H⁺) and hydroxide ions (OH⁻). This creates competition at the electrodes.
**At the Cathode (-):**
Both the metal ion and the hydrogen ion are attracted here. The rule is: the *less reactive* element is discharged. If the metal is less reactive than hydrogen (e.g., copper, silver, gold), the metal is produced. If the metal is more reactive than hydrogen (e.g., sodium, magnesium), hydrogen gas is produced.
**At the Anode (+):**
Both the non-metal ion and the hydroxide ion are attracted here. The rule is: if a halide ion (Cl⁻, Br⁻, I⁻) is present, the halogen gas is produced. If no halide is present (e.g., sulfates, nitrates), oxygen gas is produced from the hydroxide ions.
## Mathematical/Scientific Relationships
### Half-Equations
Half-equations show the transfer of electrons at a specific electrode. Electrons are represented by 'e⁻'.
**Cathode (Reduction):** Electrons are gained, so they appear on the *left* side of the arrow.
Example: Cu²⁺(aq) + 2e⁻ → Cu(s)
**Anode (Oxidation):** Electrons are lost, so they appear on the *right* side of the arrow (or subtracted on the left).
Example: 2Cl⁻(aq) → Cl₂(g) + 2e⁻
*Examiner Tip: Always check that the total charge on the left equals the total charge on the right.*
## Practical Applications
### Required Practical: Electrolysis of Aqueous Solutions
Students must investigate what happens when aqueous solutions are electrolysed using inert electrodes. Common solutions tested are copper(II) chloride, copper(II) sulfate, sodium chloride, and sodium sulfate.
**Apparatus:** Beaker, two graphite electrodes, DC power supply, wires with crocodile clips, electrolyte solution.
**Method:**
1. Pour 50 cm³ of the electrolyte into the beaker.
2. Insert the graphite electrodes, ensuring they do not touch.
3. Connect the electrodes to the DC power supply.
4. Turn on the power to 4V and observe the electrodes.
5. Hold damp blue litmus paper near the anode to test for chlorine gas (it will bleach white).
### The Purification of Copper
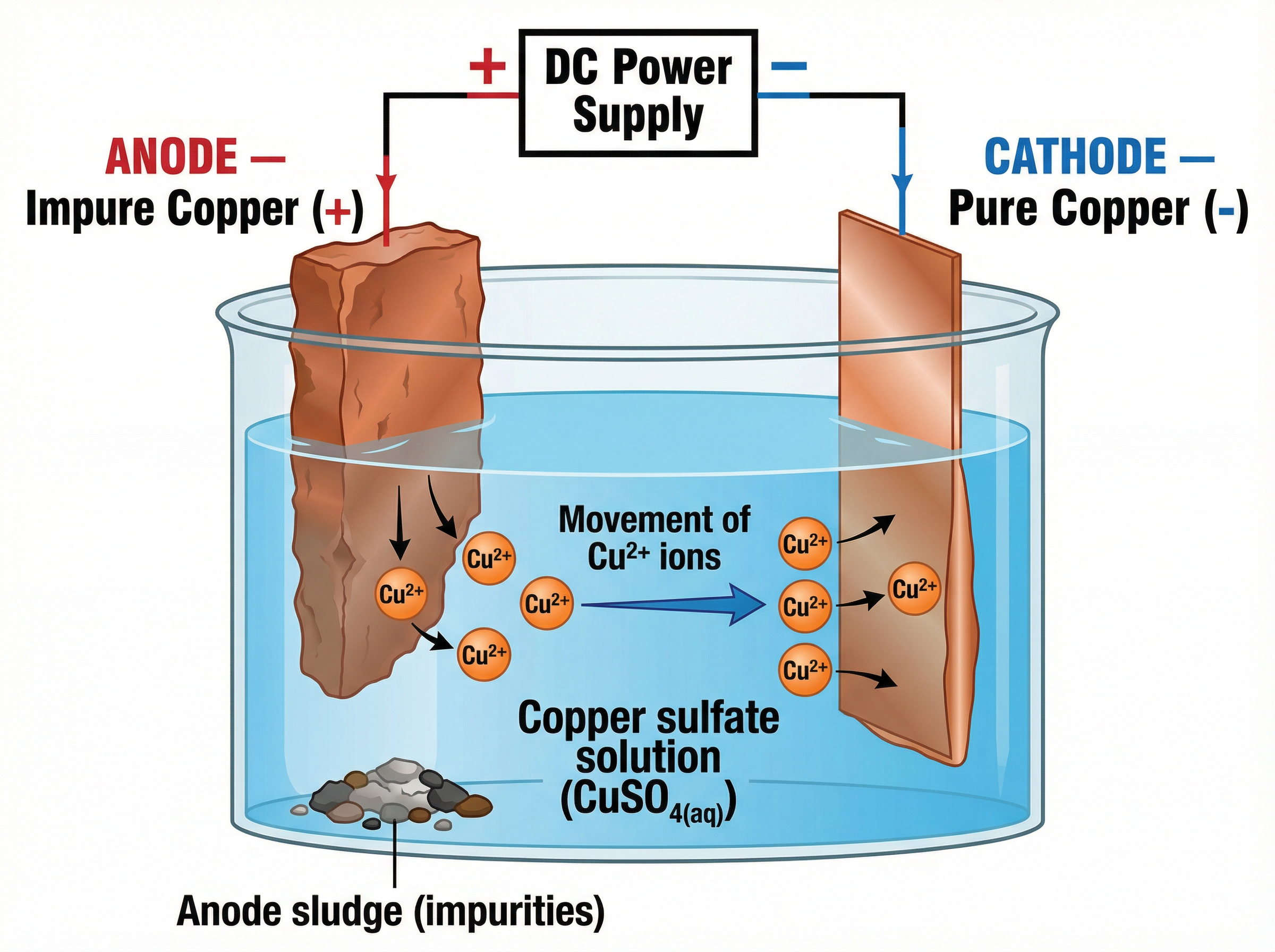
Electrolysis is used to purify copper for electrical wiring. The anode is made of impure copper, and the cathode is a thin sheet of pure copper. The electrolyte is copper(II) sulfate solution.
At the anode, copper atoms lose electrons and dissolve as Cu²⁺ ions. At the cathode, Cu²⁺ ions gain electrons and deposit as pure copper. The impurities fall to the bottom as 'anode sludge'. The concentration of the solution remains constant because the rate of copper dissolving equals the rate of copper depositing.
## Audio Revision
Listen to our comprehensive 10-minute podcast covering all these concepts, perfect for revising on the go:
