Extracting metals and equilibria — Edexcel GCSE Study Guide
Exam Board: Edexcel | Level: GCSE
Master the principles of reversible reactions, dynamic equilibrium, and the industrial Haber process. This topic is heavily tested across all exam boards and connects theoretical chemistry to real-world industrial applications, making it essential for securing top grades.

## Overview
Welcome to one of the most conceptually rich and frequently examined topics in GCSE Chemistry: Extracting Metals and Equilibria. This unit bridges the gap between theoretical chemistry and real-world industrial application. You will explore how reversible reactions operate, the conditions required to establish dynamic equilibrium, and how scientists manipulate these conditions to maximise product yield.
Understanding this topic is critical because it connects deeply with reaction kinetics, energetics, and industrial processes. Examiners frequently use this area to test higher-order thinking skills, asking candidates to evaluate the trade-offs between reaction rate, product yield, and economic viability. The classic example is the Haber process for manufacturing ammonia, which perfectly illustrates Le Chatelier's Principle in action.
By mastering these concepts, you will be well-equipped to tackle multi-mark extended response questions that demand a clear, logical explanation of how changing temperature, pressure, or concentration shifts the position of equilibrium.

## Key Concepts
### Concept 1: Reversible Reactions
Most chemical reactions you have studied so far go to completion; reactants turn into products, and the reaction stops. However, in a **reversible reaction**, the products can react together to reform the original reactants. This is denoted by a specific symbol: the double equilibrium arrow (⇌).
When a reversible reaction occurs in a **closed system** (where no reactants or products can escape), it can reach a state called **dynamic equilibrium**. At this point, the forward and backward reactions are still happening (hence 'dynamic'), but they occur at exactly the same rate. As a result, the concentrations of both reactants and products remain constant.
**Example**: The thermal decomposition of ammonium chloride.
NH₄Cl(s) ⇌ NH₃(g) + HCl(g)
Heating the solid ammonium chloride drives the forward endothermic reaction, producing ammonia and hydrogen chloride gases. When cooled, the gases react to reform the solid.
### Concept 2: Le Chatelier's Principle and Changing Conditions
Le Chatelier's Principle states that if a change is made to the conditions of a system at equilibrium, the system will shift to oppose that change. This principle allows chemists to predict how the position of equilibrium will shift when temperature, pressure, or concentration is altered.
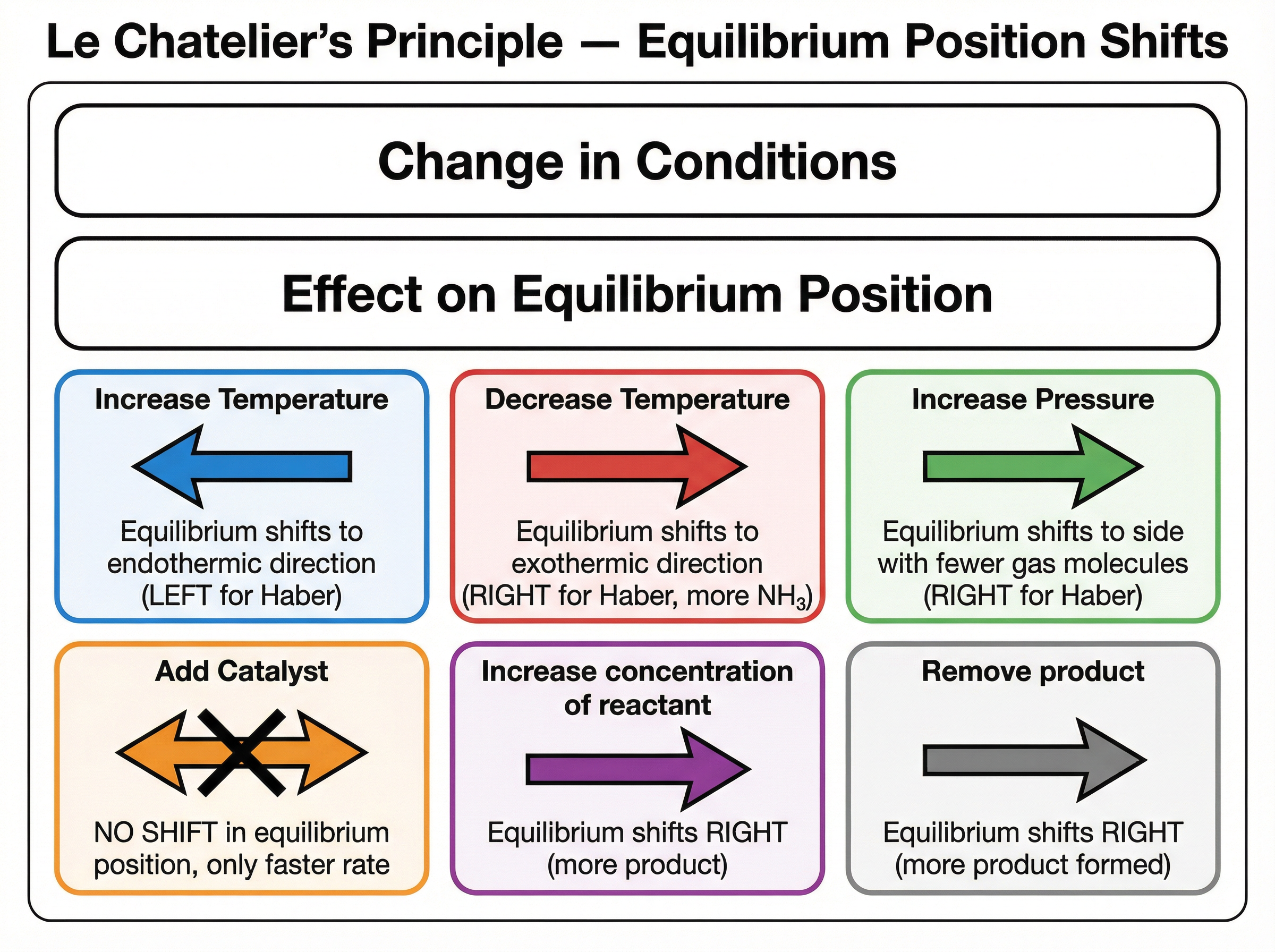
- **Temperature**: Increasing the temperature shifts the equilibrium in the direction of the endothermic reaction (to absorb the extra heat). Decreasing the temperature shifts it in the exothermic direction (to release heat).
- **Pressure**: For reactions involving gases, increasing the pressure shifts the equilibrium towards the side with fewer molecules of gas (to reduce the pressure). Decreasing pressure shifts it towards the side with more gas molecules.
- **Concentration**: Increasing the concentration of a reactant shifts the equilibrium to the right (towards the products) to use up the added reactant.
### Concept 3: The Role of a Catalyst
A catalyst speeds up the rate of a chemical reaction by providing an alternative reaction pathway with a lower activation energy. Crucially, in a reversible reaction, a catalyst speeds up BOTH the forward and backward reactions equally.
Therefore, a catalyst **does not change the position of equilibrium** or the final yield of the product. It only reduces the time taken for the system to reach equilibrium. This is a very common trap in exams; remember that catalysts affect *rate*, not *yield*.
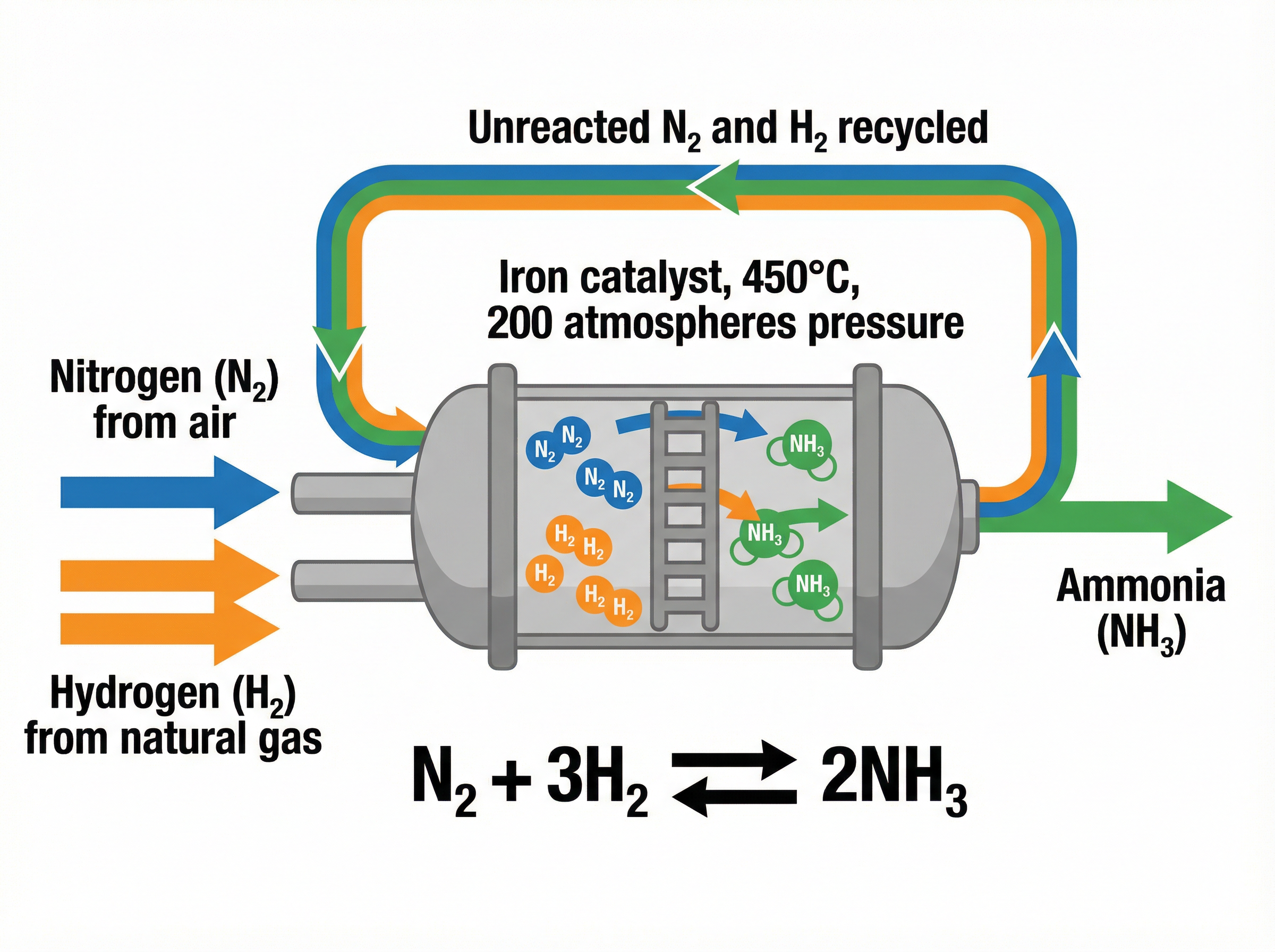
### Concept 4: The Haber Process
The Haber process is the industrial method used to manufacture ammonia (NH₃), which is essential for producing nitrogen-based fertilisers. The reaction involves nitrogen (extracted from the air) and hydrogen (obtained from natural gas).
N₂(g) + 3H₂(g) ⇌ 2NH₃(g)
The forward reaction is exothermic. To maximise the yield of ammonia, we need to carefully select the reaction conditions based on Le Chatelier's Principle, while also considering the rate of reaction and economic costs.
## Mathematical/Scientific Relationships
**The Equilibrium Constant (Kc)** (Higher Tier / A-Level transition):
While calculating Kc is generally reserved for A-Level, understanding the ratio of products to reactants helps conceptualise equilibrium position.
**Gas Molecule Counting**:
To predict pressure shifts, count the stoichiometric coefficients of gaseous substances in the balanced equation.
Example: N₂(g) + 3H₂(g) ⇌ 2NH₃(g)
Left side: 1 + 3 = 4 molecules of gas.
Right side: 2 molecules of gas.
## Practical Applications
The most significant practical application of this topic is the **Haber Process**. The conditions chosen are a classic example of an industrial compromise:
1. **Temperature (450 °C)**: Because the forward reaction is exothermic, a low temperature would give the highest yield of ammonia. However, a low temperature would result in a reaction rate that is too slow to be economically viable. 450 °C is a compromise that provides an acceptable yield at a reasonable rate.
2. **Pressure (200 atmospheres)**: The forward reaction produces fewer gas molecules (4 molecules react to form 2). Therefore, a high pressure shifts the equilibrium to the right, increasing the yield. However, generating and maintaining extremely high pressures is dangerous and requires expensive, heavily reinforced equipment. 200 atmospheres is a compromise between maximising yield and minimising cost and risk.
3. **Iron Catalyst**: An iron catalyst is used to speed up the rate at which equilibrium is reached, allowing the process to operate effectively at the compromise temperature of 450 °C.
4. **Recycling**: Unreacted nitrogen and hydrogen gases are recycled back into the reactor to minimise waste and improve overall efficiency.