Extracting metals and equilibria — Edexcel GCSE Study Guide
Exam Board: Edexcel | Level: GCSE
Master the principles of extracting metals, from the reactivity series to electrolysis and biological methods. This topic is essential for understanding how chemistry applies to real-world industrial processes and sustainability, and frequently features in high-mark exam questions.
## Overview
Extracting metals is a fundamental topic in GCSE Chemistry that bridges theoretical concepts with crucial industrial applications. This topic explores how we obtain the metals we rely on daily—from the iron in our buildings to the aluminium in our devices.
At its core, this topic is about the **reactivity series**. A metal's position in this series dictates the method required to extract it from its ore. You will learn why some metals can simply be heated with carbon, while others require massive amounts of electrical energy through electrolysis. Examiners love this topic because it allows them to test multiple concepts simultaneously: redox reactions, displacement, environmental chemistry, and evaluating data through Life Cycle Assessments (LCAs).
Expect questions that ask you to explain extraction methods in terms of electron transfer, evaluate the environmental impact of different processes, and apply your knowledge to unfamiliar metals.
---
## Listen to the Podcast
For a complete audio overview of this topic, including exam tips and a quick-fire quiz, listen to our dedicated podcast episode:

---
## Key Concepts
### Concept 1: The Reactivity Series and Extraction Methods
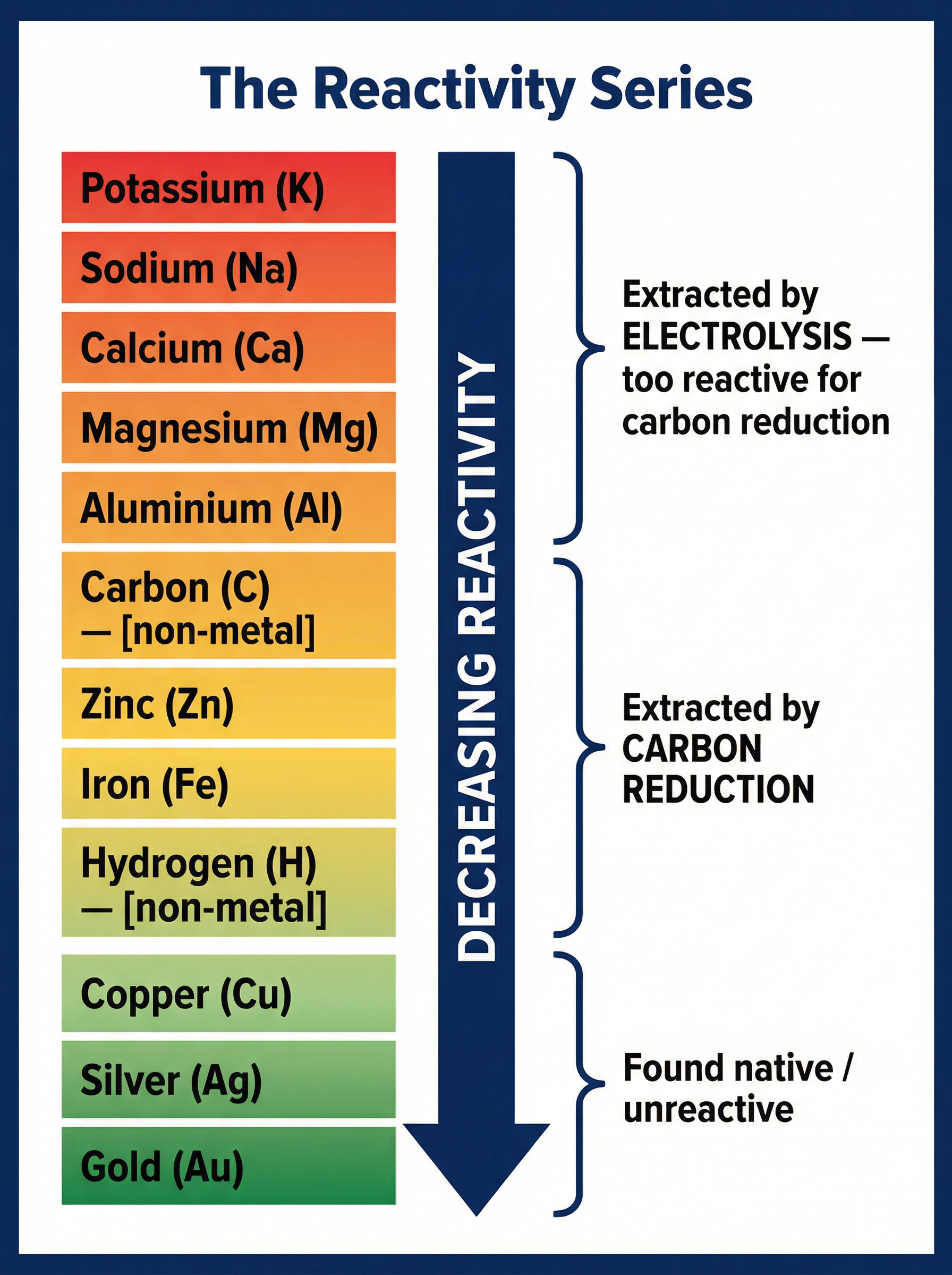
The reactivity series is a list of metals ordered by their reactivity, from most reactive at the top to least reactive at the bottom. It is the master key for understanding metal extraction.
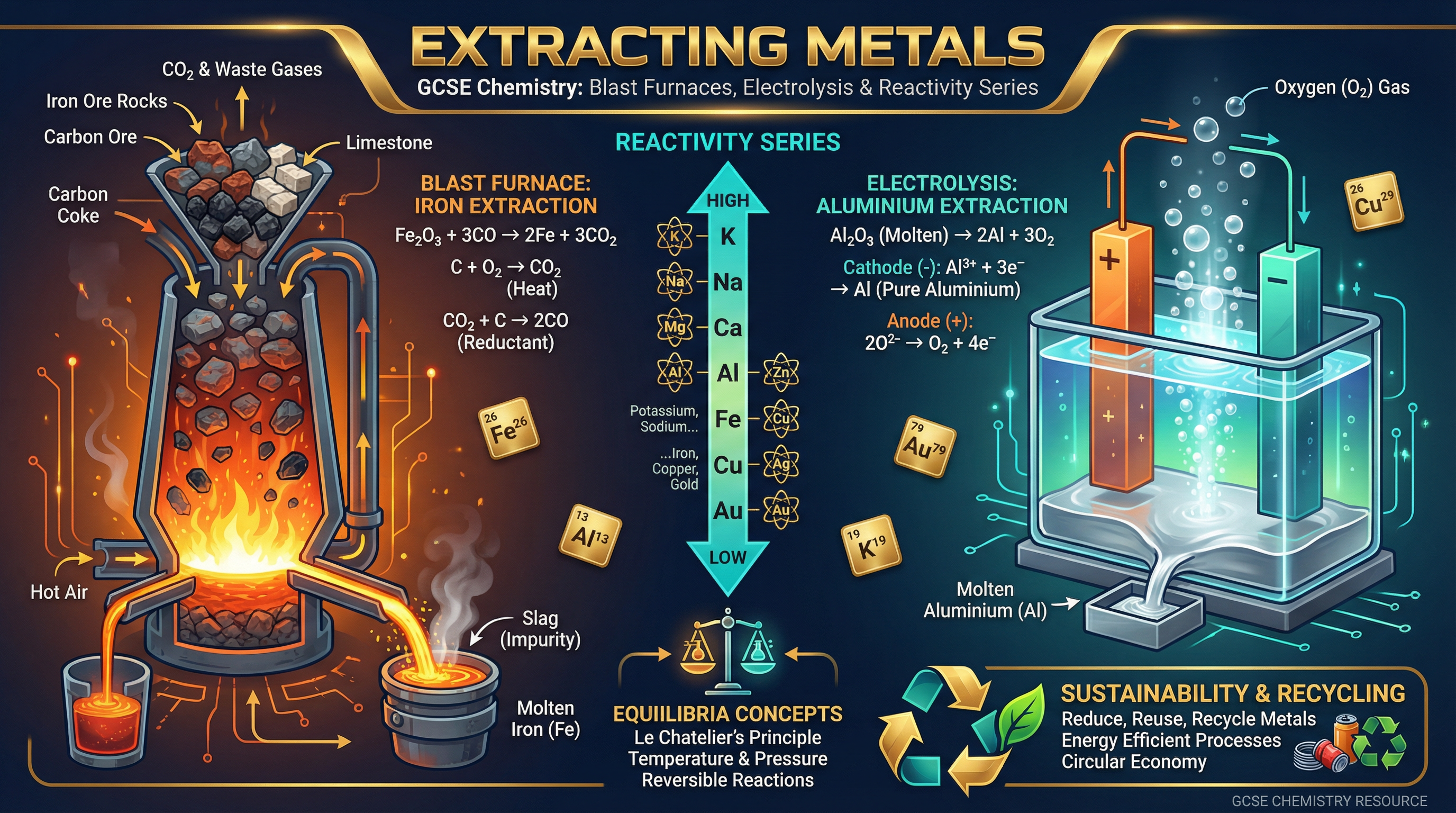
Metals are usually found in the Earth's crust combined with other elements, most commonly oxygen, as metal oxides (ores). To extract the pure metal, we must remove the oxygen. This is a **reduction** reaction. The method we use depends entirely on the metal's position in the reactivity series relative to carbon.
**Metals less reactive than carbon** (e.g., zinc, iron, copper) can be extracted by heating their oxides with carbon. Because carbon is more reactive, it displaces the metal from the oxide. It "steals" the oxygen, forming carbon dioxide and leaving the pure metal behind.
**Metals more reactive than carbon** (e.g., aluminium, magnesium, sodium) cannot be extracted this way because carbon is not reactive enough to displace them. Instead, these metals must be extracted using **electrolysis**—passing an electric current through the molten metal compound. This process is highly energy-intensive and therefore very expensive.
**Example**: Iron is extracted in a blast furnace. Iron(III) oxide reacts with carbon (or carbon monoxide) to produce iron and carbon dioxide: $2Fe_2O_3 + 3C \rightarrow 4Fe + 3CO_2$.
### Concept 2: Displacement Reactions and Redox
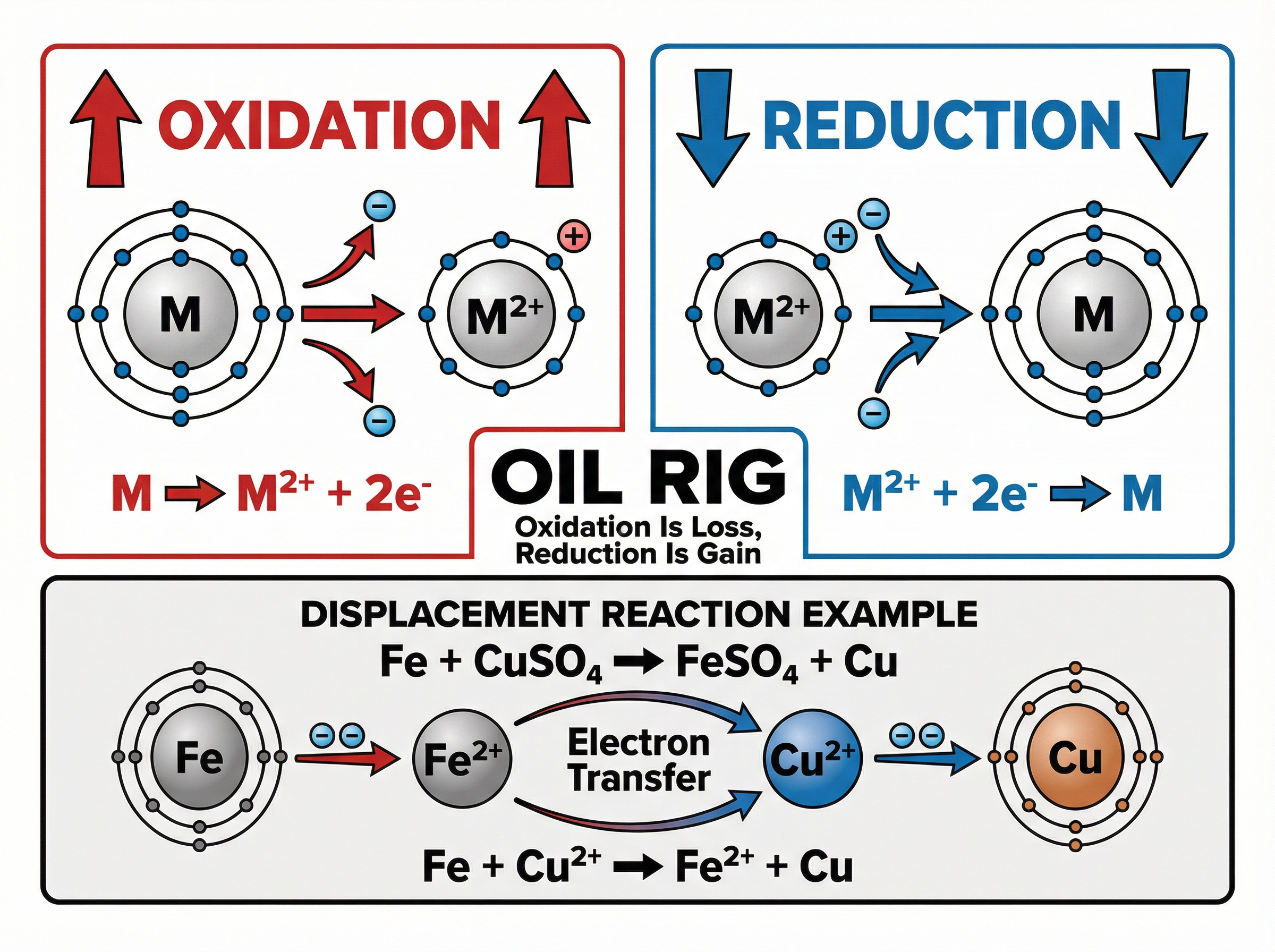
A displacement reaction occurs when a more reactive metal is added to a solution containing a compound of a less reactive metal. The more reactive metal "pushes out" the less reactive metal from its compound.
Examiners require you to understand these reactions in terms of **electron transfer**. This makes them **redox** reactions (Reduction-Oxidation).
When a more reactive metal (like magnesium) is placed in a solution of a less reactive metal's salt (like copper sulfate), the magnesium atoms lose electrons to become magnesium ions. **Oxidation is loss of electrons**. Simultaneously, the copper ions in the solution gain those electrons to become copper atoms. **Reduction is gain of electrons**.
**Example**: $Mg + CuSO_4 \rightarrow MgSO_4 + Cu$
In terms of ions: $Mg + Cu^{2+} \rightarrow Mg^{2+} + Cu$
Here, Mg is oxidised (loses 2 electrons) and $Cu^{2+}$ is reduced (gains 2 electrons).
### Concept 3: Biological Extraction Methods
As high-grade ores (ores containing a high percentage of metal) become scarce, scientists have developed alternative methods to extract metals from low-grade ores. These biological methods are more sustainable but much slower.
**Phytoextraction**: Plants are grown on soil containing low-grade metal ores. The plants absorb metal ions through their roots and concentrate them in their leaves. The plants are then harvested and burned. The resulting ash contains a high concentration of the metal compound, which can then be processed using standard methods (like displacement with scrap iron or electrolysis).
**Bioleaching**: Bacteria are mixed with low-grade ores. The bacteria carry out chemical reactions that break down the ore, producing an acidic solution called a **leachate**, which contains metal ions. The metal can then be extracted from the leachate.
### Concept 4: Life Cycle Assessments (LCAs) and Recycling
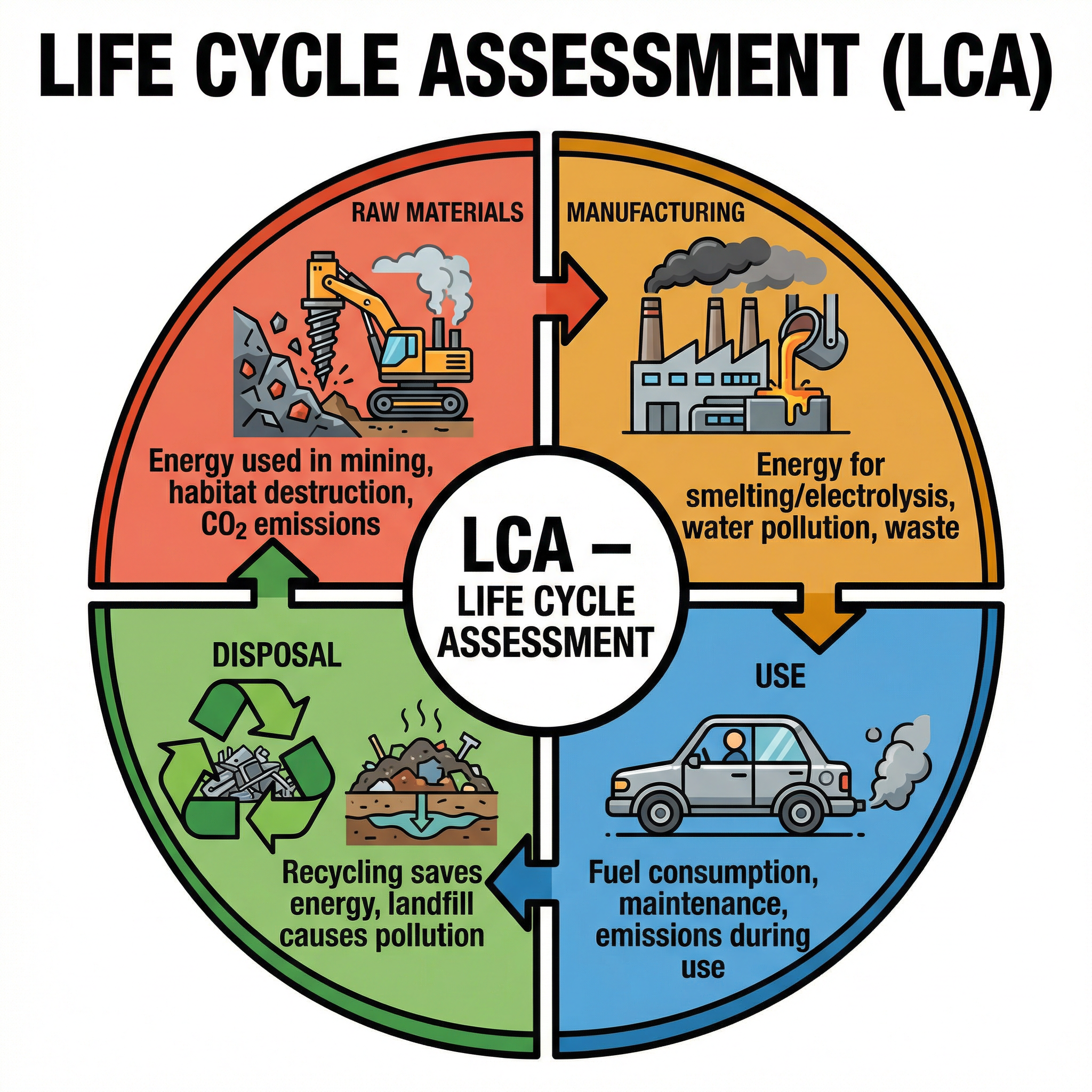
A Life Cycle Assessment (LCA) is an evaluation of the environmental impact of a product across its entire lifespan. Examiners often provide data tables and ask you to evaluate which product is more environmentally friendly.
An LCA must consider four stages:
1. **Extracting and processing raw materials**: Includes mining (habitat destruction, energy use) and processing (smelting, electrolysis).
2. **Manufacturing and packaging**: Includes energy use, water pollution, and waste gas emissions (like $CO_2$ or $SO_2$).
3. **Use and operation**: Includes energy/fuel required during its lifetime and maintenance.
4. **Disposal**: Includes landfill space, pollution, or the energy required to recycle it.
**Recycling** is crucial because it addresses issues across the LCA. It requires significantly less energy than extracting new metal from ore (e.g., recycling aluminium uses only 5% of the energy needed for electrolysis of bauxite). This makes it economically beneficial while also reducing the environmental impacts of mining and reducing waste sent to landfill.
---
## Mathematical/Scientific Relationships
While this topic is largely conceptual, you must be comfortable with writing and balancing chemical equations, particularly half-equations for redox processes.
**General Displacement Equation**:
$More\ Reactive\ Metal + Less\ Reactive\ Metal\ Compound \rightarrow More\ Reactive\ Metal\ Compound + Less\ Reactive\ Metal$
**Half-Equations (Higher Tier)**:
- **Oxidation (Loss of electrons)**: $M \rightarrow M^{n+} + ne^-$
- **Reduction (Gain of electrons)**: $M^{n+} + ne^- \rightarrow M$
---
## Practical Applications
Understanding metal extraction is essential for the transition to a sustainable economy. The shift towards electric vehicles requires massive amounts of lithium, cobalt, and copper. Traditional mining is environmentally destructive, making bioleaching and advanced recycling technologies critical areas of current chemical research.
When you put an aluminium can in the recycling bin, you are saving the massive electrical energy cost of electrolysis. That recycled aluminium can be back on a supermarket shelf as a new can in as little as 60 days.