Fuels and Earth science — Edexcel GCSE Study Guide
Exam Board: Edexcel | Level: GCSE
Master the evolution of the Earth's atmosphere, the greenhouse effect, and climate change. This topic is heavily tested every year, especially on linking physical and biological processes to atmospheric changes and distinguishing between the natural greenhouse effect and human-driven climate change.

## Overview
Welcome to Fuels and Earth Science! This topic explores the incredible 4.6-billion-year journey of our planet's atmosphere. It takes you from a toxic, volcanic early Earth to the oxygen-rich environment that supports life today. Understanding this evolution is crucial for GCSE Chemistry, as examiners frequently ask candidates to explain the physical and biological processes that drove these changes.
Furthermore, this topic connects directly to one of the most pressing issues of our time: climate change. You will need to understand the mechanics of the greenhouse effect, how human activities are enhancing it, and the resulting consequences for our planet. Typical exam questions range from simple 1-mark recall of early atmospheric gases to complex 6-mark extended responses evaluating the impacts of global warming.

## Key Concepts
### Concept 1: The Early Atmosphere (4.6 Billion Years Ago)
For the first billion years of Earth's existence, intense volcanic activity shaped the atmosphere. Think of early Earth as a giant, venting pressure cooker. Volcanoes released massive amounts of **carbon dioxide ($CO_2$)**, **water vapour ($H_2O$)**, and **nitrogen ($N_2$)**, along with small amounts of methane ($CH_4$) and ammonia ($NH_3$).
Crucially, there was **little or no oxygen ($O_2$)**. If an examiner asks you to describe the early atmosphere, your key points must be: high $CO_2$, high water vapour, and no oxygen.
### Concept 2: Formation of the Oceans and Decreasing $CO_2$
As the Earth gradually cooled, the water vapour in the atmosphere condensed to form the oceans. This physical change had a massive impact on the atmosphere. Carbon dioxide is a soluble gas, and huge quantities of it dissolved into the newly formed oceans.
Once dissolved, the $CO_2$ reacted to form carbonate precipitates, which eventually formed sedimentary rocks (like limestone) on the seabed. This process significantly reduced the amount of $CO_2$ in the atmosphere.
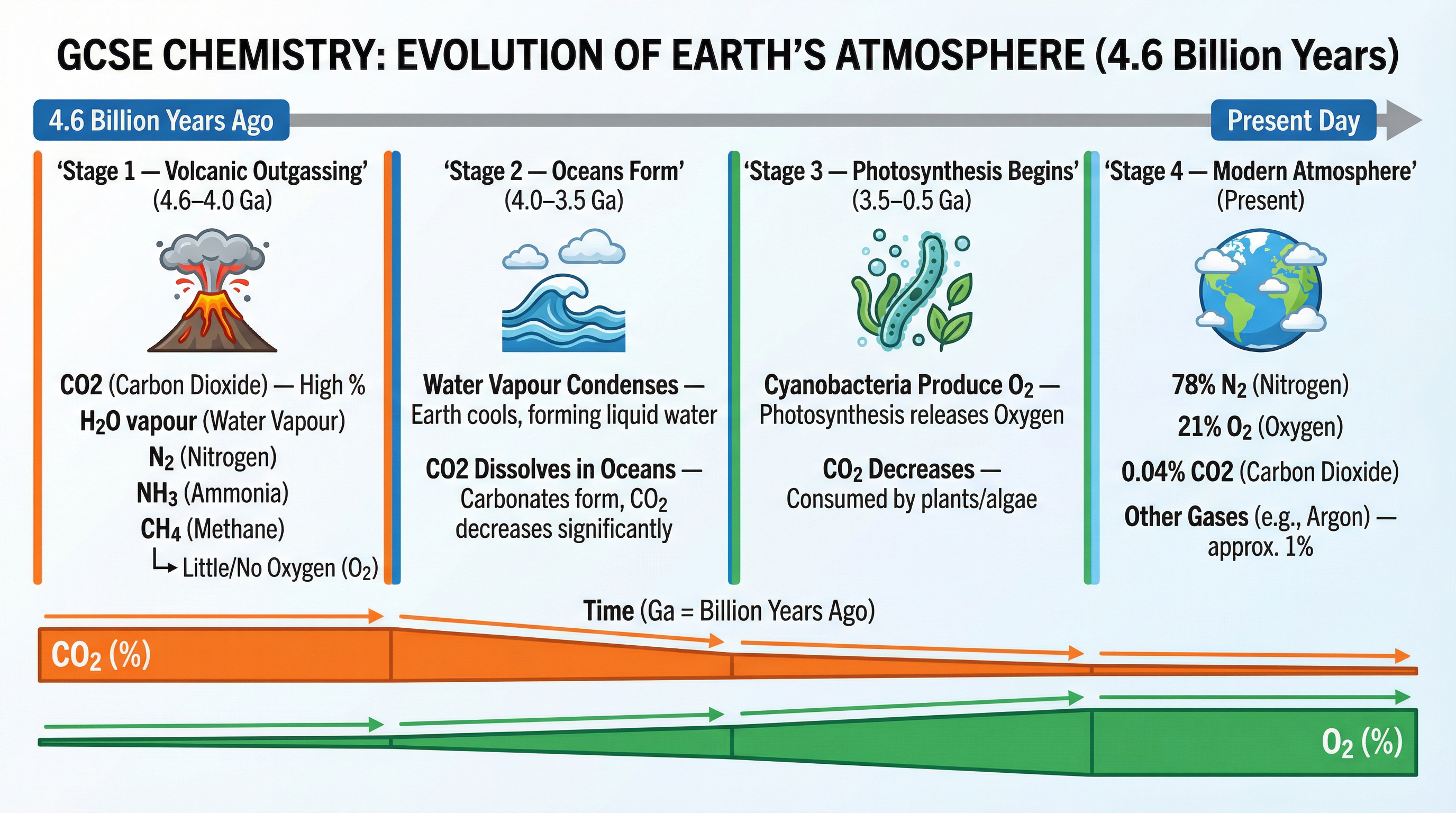
### Concept 3: The Rise of Oxygen via Photosynthesis
Around 2.7 billion years ago, a biological revolution occurred: the evolution of simple photosynthetic organisms, such as cyanobacteria (often called blue-green algae). These organisms used sunlight to convert carbon dioxide and water into glucose and oxygen.
$$6CO_2 + 6H_2O \rightarrow C_6H_{12}O_6 + 6O_2$$
Over hundreds of millions of years, photosynthesis steadily consumed $CO_2$ and released $O_2$. This biological process is entirely responsible for the oxygen-rich atmosphere we rely on today. Examiners will specifically look for you to link the increase in oxygen to the evolution of **photosynthetic organisms**.
### Concept 4: The Greenhouse Effect
The greenhouse effect is a natural and necessary process; without it, Earth would be too cold for life.
1. Short-wave solar radiation from the Sun passes through the atmosphere and is absorbed by the Earth's surface.
2. The Earth's surface warms up and re-emits this energy as long-wave infrared radiation (heat).
3. Greenhouse gases in the atmosphere (such as $CO_2$, methane, and water vapour) absorb this outgoing long-wave radiation.
4. These gases then re-radiate the heat in all directions, including back towards the Earth's surface, trapping the heat.
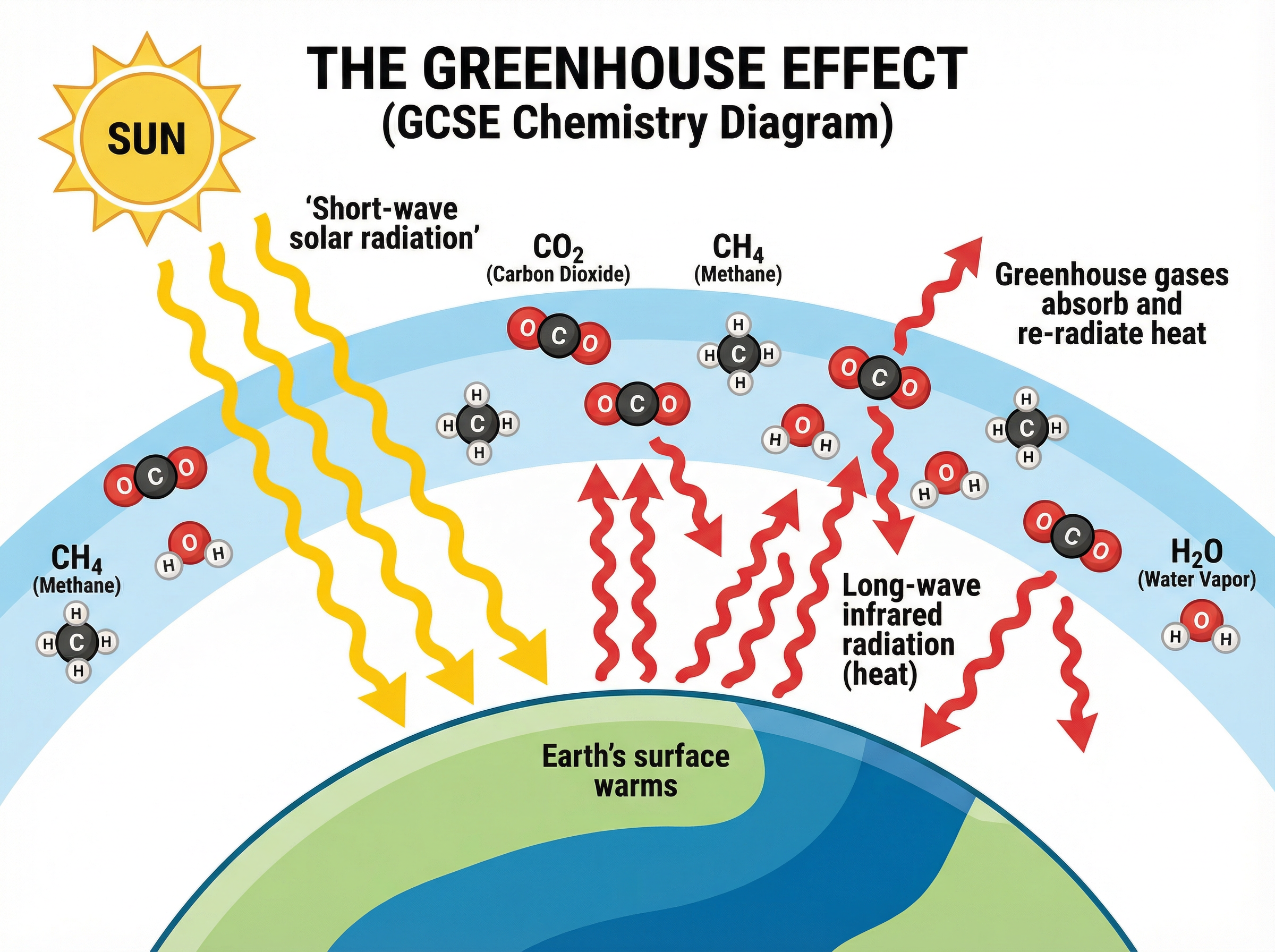
### Concept 5: Human Activity and Climate Change
While the natural greenhouse effect is beneficial, human activities are increasing the concentration of greenhouse gases, leading to an **enhanced greenhouse effect** and global warming.
- **Carbon Dioxide:** Increased by burning fossil fuels (coal, oil, gas) and deforestation (which removes trees that would otherwise absorb $CO_2$).
- **Methane:** Increased by livestock farming (cattle digestion), rice paddy fields, and the decomposition of organic waste in landfill sites.
The consequences of global warming include melting polar ice caps, rising sea levels, more extreme weather events, and changes in the distribution of wildlife species.
## Mathematical/Scientific Relationships
- **Photosynthesis Equation:** $6CO_2 + 6H_2O \rightarrow C_6H_{12}O_6 + 6O_2$
- **Proportions of the Modern Atmosphere:**
- Nitrogen ($N_2$): ~78%
- Oxygen ($O_2$): ~21%
- Argon ($Ar$): ~0.9%
- Carbon Dioxide ($CO_2$): ~0.04%
## Practical Applications: Testing for Gases
You must know the required practical chemical tests for common gases.
- **Test for Oxygen:** Insert a glowing splint into a test tube of the gas. If oxygen is present, the **splint relights**.
- **Test for Carbon Dioxide:** Bubble the gas through limewater (calcium hydroxide solution). If $CO_2$ is present, the limewater turns **cloudy/milky**.