Fuels and Earth science — Edexcel GCSE Study Guide
Exam Board: Edexcel | Level: GCSE
Master the chemistry of crude oil, fractional distillation, and combustion. This topic is essential for understanding both industrial chemistry and the environmental challenges of modern fuels.
## Overview
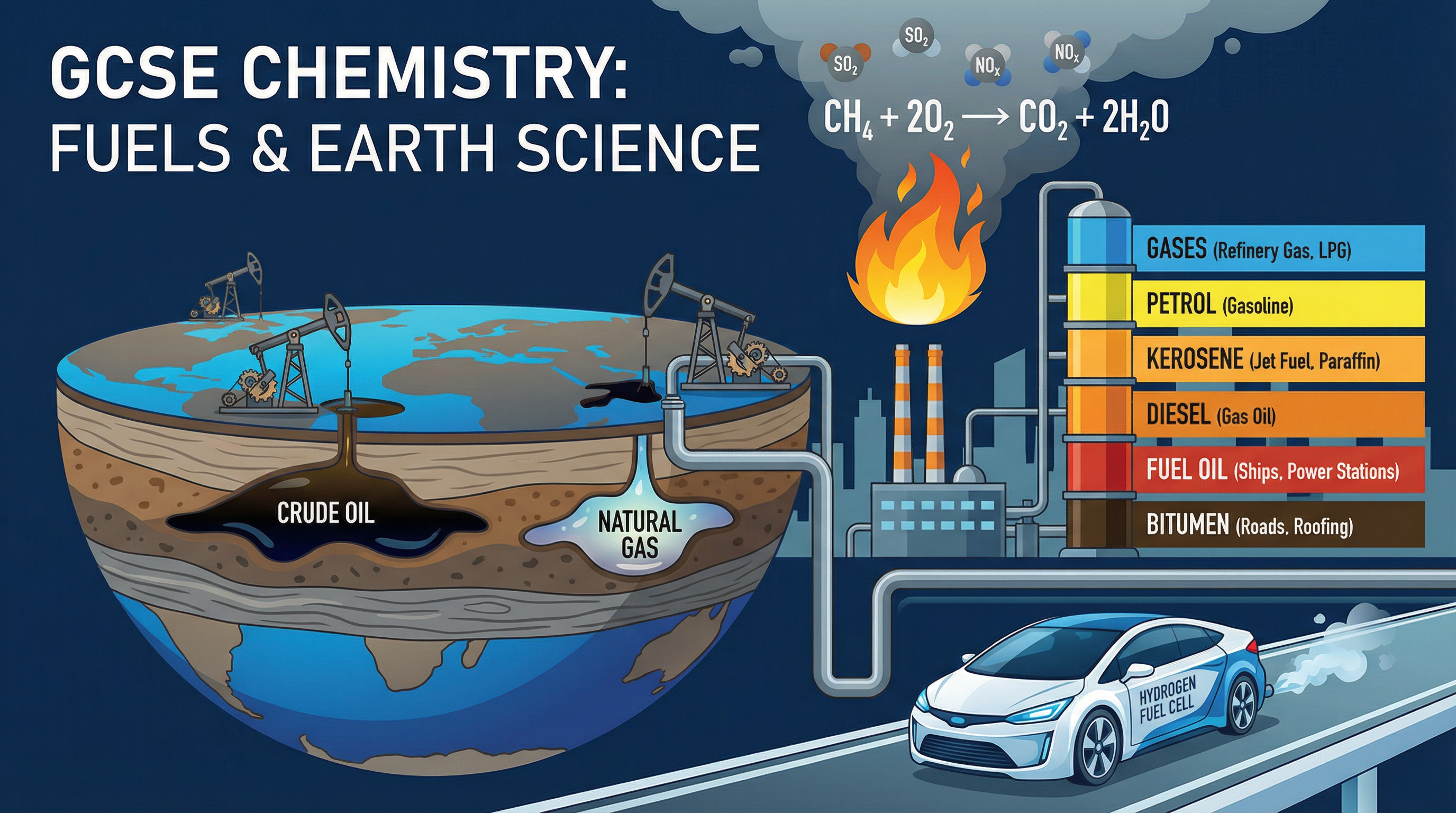
Fuels and Earth Science is a cornerstone topic in GCSE Chemistry that bridges the gap between theoretical organic chemistry and the real-world challenges of energy and the environment. This topic covers the origins of crude oil, how we separate it into useful fractions, and the chemical reactions that power our world. It's incredibly important because it directly addresses the environmental impacts of burning fossil fuels—such as acid rain and global warming—and introduces the vital industrial process of cracking.
Examiners frequently test this area through extended response questions (6-markers) on fractional distillation, balanced equations for combustion, and evaluative questions comparing traditional fuels with alternatives like hydrogen. A strong grasp of the relationship between molecular size and physical properties is crucial for securing top marks.

## Key Concepts
### Concept 1: Crude Oil and Hydrocarbons
Crude oil is a **finite resource** found in rocks. It was formed over millions of years from the remains of tiny, ancient sea animals and plants (like plankton) that were buried in mud. Over time, high pressure and temperature turned these remains into crude oil.
Crucially, crude oil is a mixture of a very large number of compounds, most of which are **hydrocarbons**. A hydrocarbon is a compound that contains **only** carbon and hydrogen atoms. Examiners will penalise you if you forget the word "only". The majority of hydrocarbons in crude oil are alkanes.
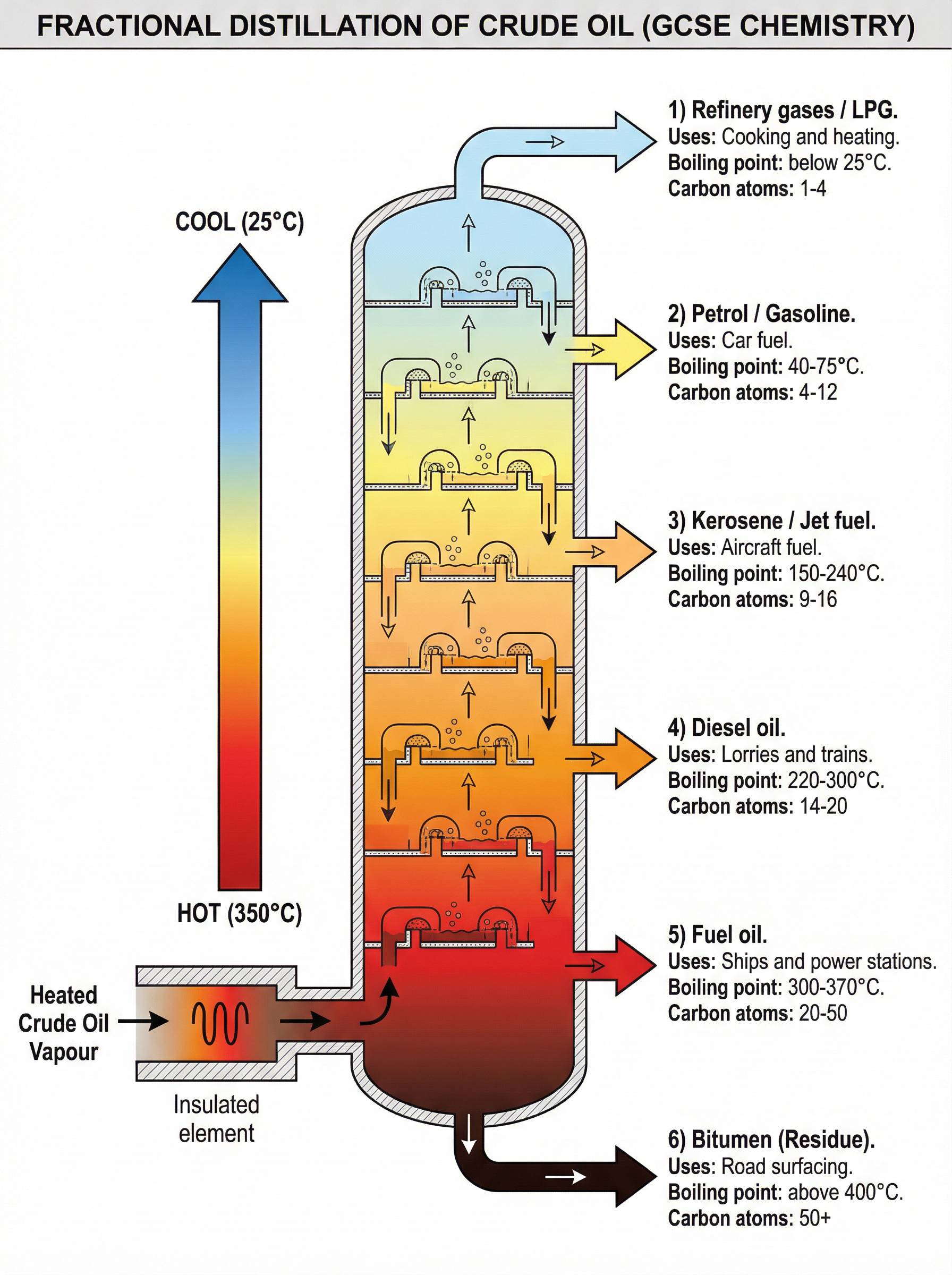
### Concept 2: Fractional Distillation
Because crude oil is a mixture, it must be separated to be useful. This is done via **fractional distillation**. The process relies on the fact that different hydrocarbons have different boiling points depending on their chain length.
1. **Vaporisation**: The crude oil is heated strongly until most of it turns into a gas (vapour).
2. **Temperature Gradient**: The vapour enters a fractionating column which is hot at the bottom and cooler at the top.
3. **Condensation**: The vapours rise up the column. When they reach a region where the temperature is lower than their boiling point, they condense back into a liquid.
**Trends in Properties:**
As the hydrocarbon chain length increases (going down the column):
- **Boiling point increases** (stronger intermolecular forces require more energy to overcome).
- **Viscosity increases** (the liquids become thicker and flow less easily).
- **Flammability decreases** (they are harder to ignite).
### Concept 3: Combustion
When hydrocarbons burn in plenty of oxygen, **complete combustion** occurs. The carbon and hydrogen are completely oxidised to produce carbon dioxide and water. This reaction releases a large amount of energy.
If oxygen is restricted, **incomplete combustion** occurs. Instead of carbon dioxide, this produces **carbon monoxide** (a toxic, colourless, odourless gas) and sometimes solid carbon particles (soot), which cause global dimming and respiratory problems.
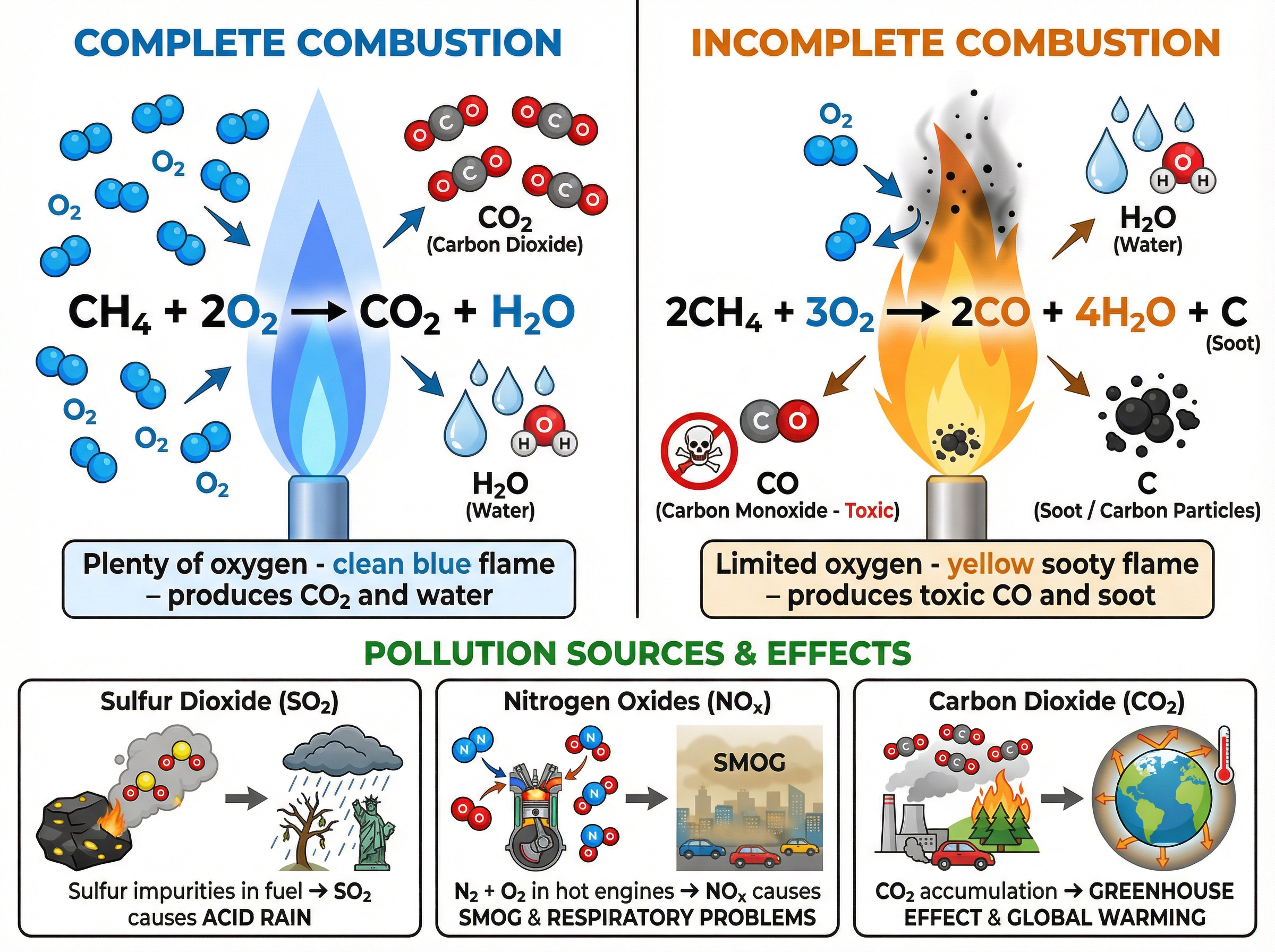
### Concept 4: Atmospheric Pollutants
Burning fuels releases several pollutants:
- **Sulfur Dioxide ($SO_2$)**: Crude oil contains sulfur impurities. When burned, the sulfur reacts with oxygen to form sulfur dioxide, which causes acid rain.
- **Nitrogen Oxides ($NO_x$)**: At the high temperatures inside car engines, nitrogen and oxygen from the air react together. These cause respiratory issues and contribute to acid rain and smog.
### Concept 5: Cracking
There is a high demand for short-chain hydrocarbons (like petrol) but a surplus of long-chain hydrocarbons. **Cracking** solves this by breaking down large, less useful alkane molecules into smaller, more useful alkanes and alkenes.
Cracking is a thermal decomposition reaction. It requires:
- High temperature ($500-600^\circ C$)
- A catalyst (such as aluminium oxide) OR steam
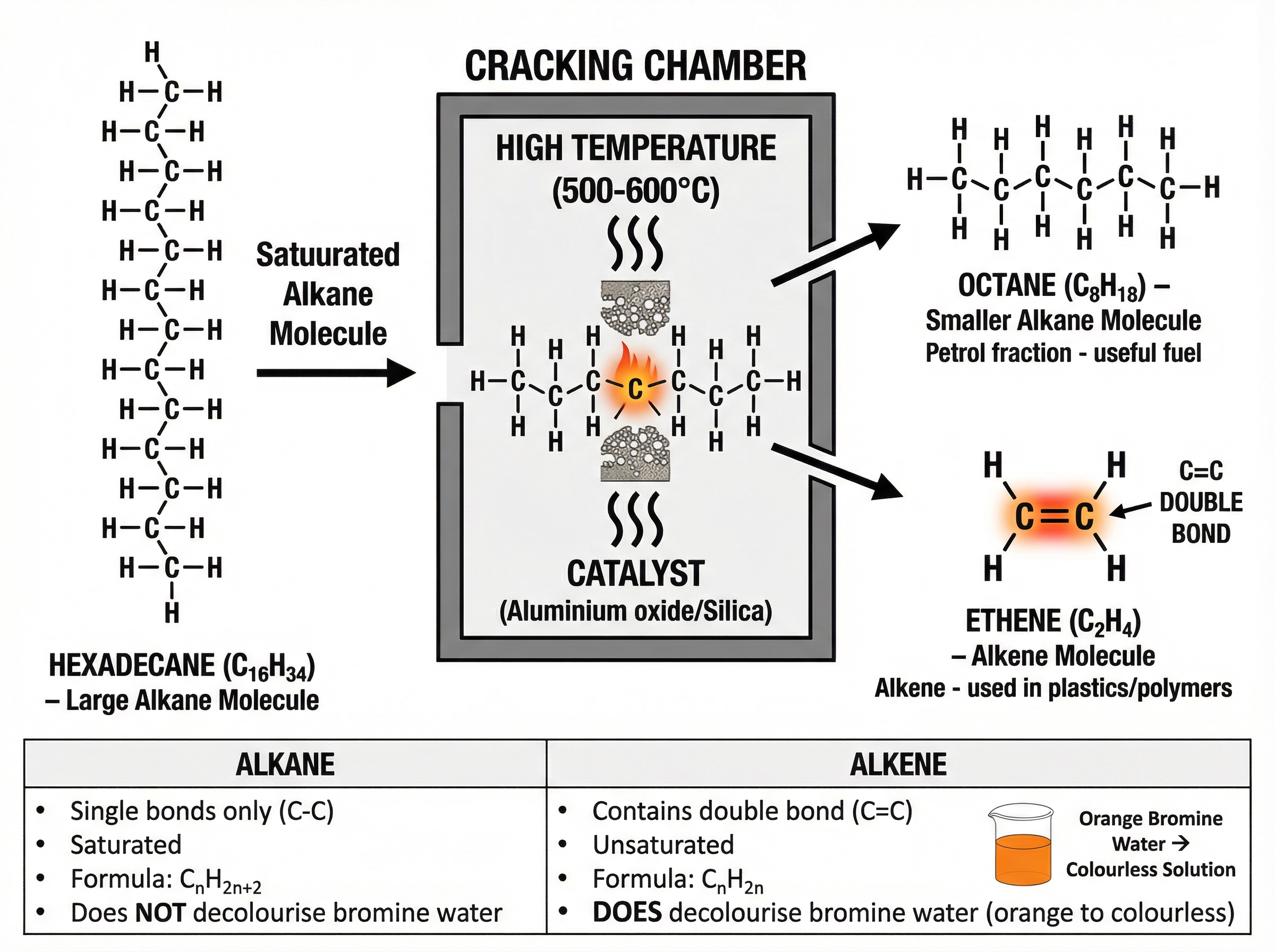
Cracking always produces an **alkene**. Alkenes are unsaturated (they contain a carbon-carbon double bond) and are used to make polymers. You can test for alkenes using bromine water: it turns from orange to colourless.
## Mathematical/Scientific Relationships
**General Formula for Alkanes**: $C_n H_{2n+2}$
- Where $n$ is the number of carbon atoms. If an alkane has 5 carbons, it has $(2 \times 5) + 2 = 12$ hydrogens ($C_5H_{12}$).
**General Formula for Alkenes**: $C_n H_{2n}$
- Alkenes have exactly twice as many hydrogen atoms as carbon atoms.
**Balanced Equations for Complete Combustion**:
Always balance in the order C, then H, then O.
Example for Propane ($C_3H_8$):
$C_3H_8 + 5O_2 \rightarrow 3CO_2 + 4H_2O$
## Practical Applications
Understanding these concepts is vital for the petrochemical industry. Refineries process millions of barrels of oil daily to produce the fuels that power global transport and the chemical feedstocks (alkenes) needed for the plastics industry. Furthermore, understanding combustion and pollutants drives the development of catalytic converters in cars and the push towards alternative fuels like hydrogen fuel cells.