Groups in the periodic table — Edexcel GCSE Study Guide
Exam Board: Edexcel | Level: GCSE
Master the properties, trends, and reactions of Group 7 Halogens. This guide covers everything from physical states to complex displacement and redox reactions to secure top marks in your GCSE Chemistry exam.
## Overview
Welcome to your definitive guide on **Group 7: The Halogens**. This topic is a cornerstone of GCSE Chemistry, appearing consistently across all major exam boards (AQA, Edexcel, OCR, and Cambridge iGCSE). The halogens are highly reactive non-metals found on the right-hand side of the periodic table, just before the noble gases.
Understanding Group 7 is critical because it bridges fundamental atomic structure with observable chemical reactions. Examiners love to test your ability to link the macroscopic properties (like colour and physical state) to microscopic explanations (like electron shielding and intermolecular forces). This topic also introduces essential synoptic links to redox chemistry, requiring you to explain displacement reactions in terms of electron transfer.
Typical exam questions range from simple 1-mark recall of colours, to complex 4-6 mark explanations of reactivity trends and ionic equations. By mastering this guide, you will be fully prepared to tackle these questions with the precision and terminology examiners reward.
Listen to the companion podcast for a comprehensive audio review:

## Key Concepts
### Concept 1: Physical Properties and Trends
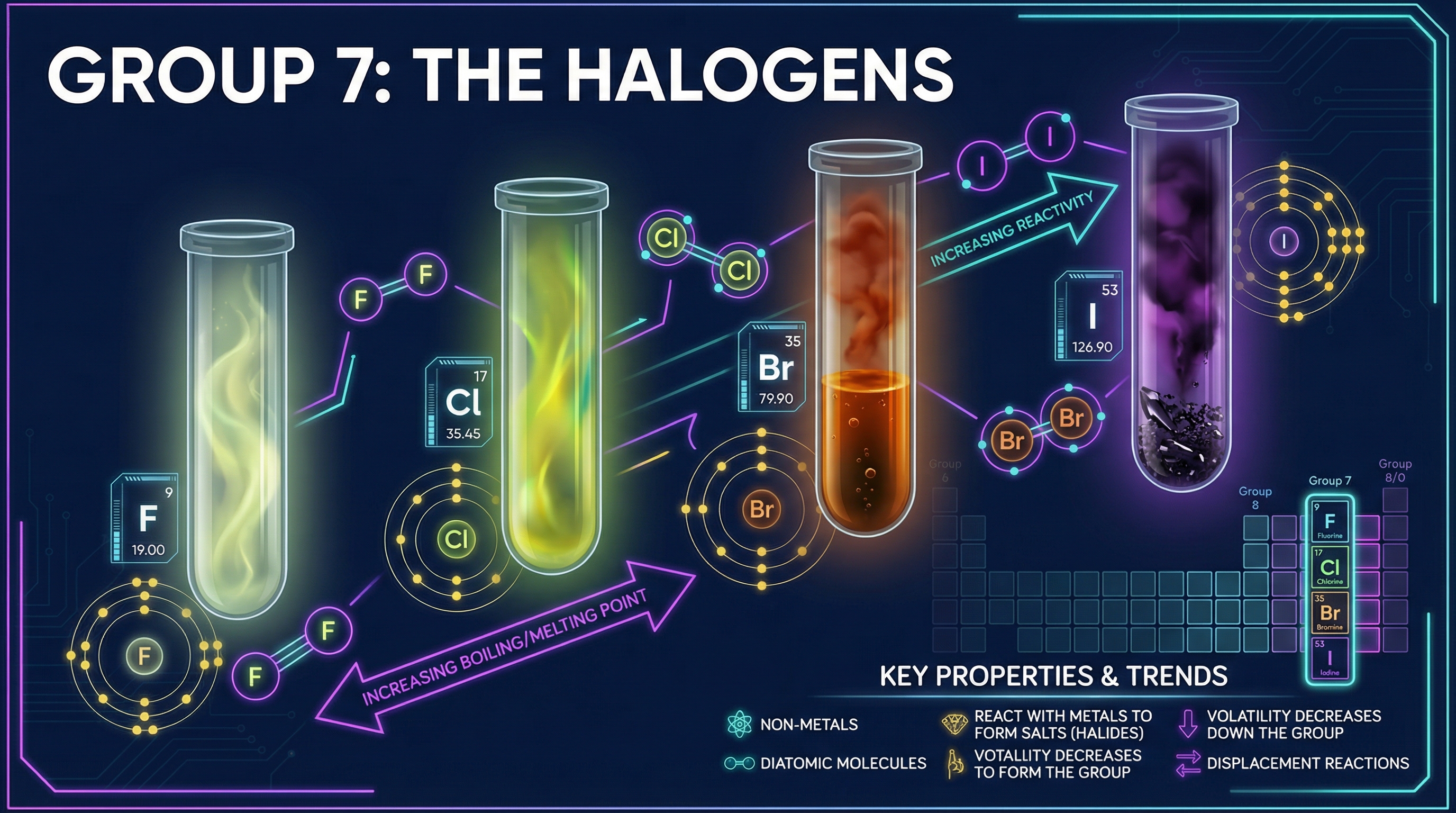
The halogens exist as diatomic molecules ($F_2$, $Cl_2$, $Br_2$, $I_2$), meaning they travel in pairs held together by a single covalent bond. At room temperature, they exhibit a clear trend in physical states and colours:
* **Fluorine ($F_2$)**: Pale yellow gas
* **Chlorine ($Cl_2$)**: Yellow-green gas
* **Bromine ($Br_2$)**: Orange-brown liquid (one of only two elements liquid at room temperature)
* **Iodine ($I_2$)**: Dark grey/purple-black solid (sublimes to a purple vapour)
**The Trend:** As you go down Group 7, the melting and boiling points **increase**.
**Why does this happen?** As the molecules get larger down the group, they have more electrons. This leads to stronger intermolecular forces (specifically London dispersion forces/induced dipoles) between the molecules. Consequently, more thermal energy is required to overcome these forces and separate the molecules to change state.
### Concept 2: The Trend in Reactivity
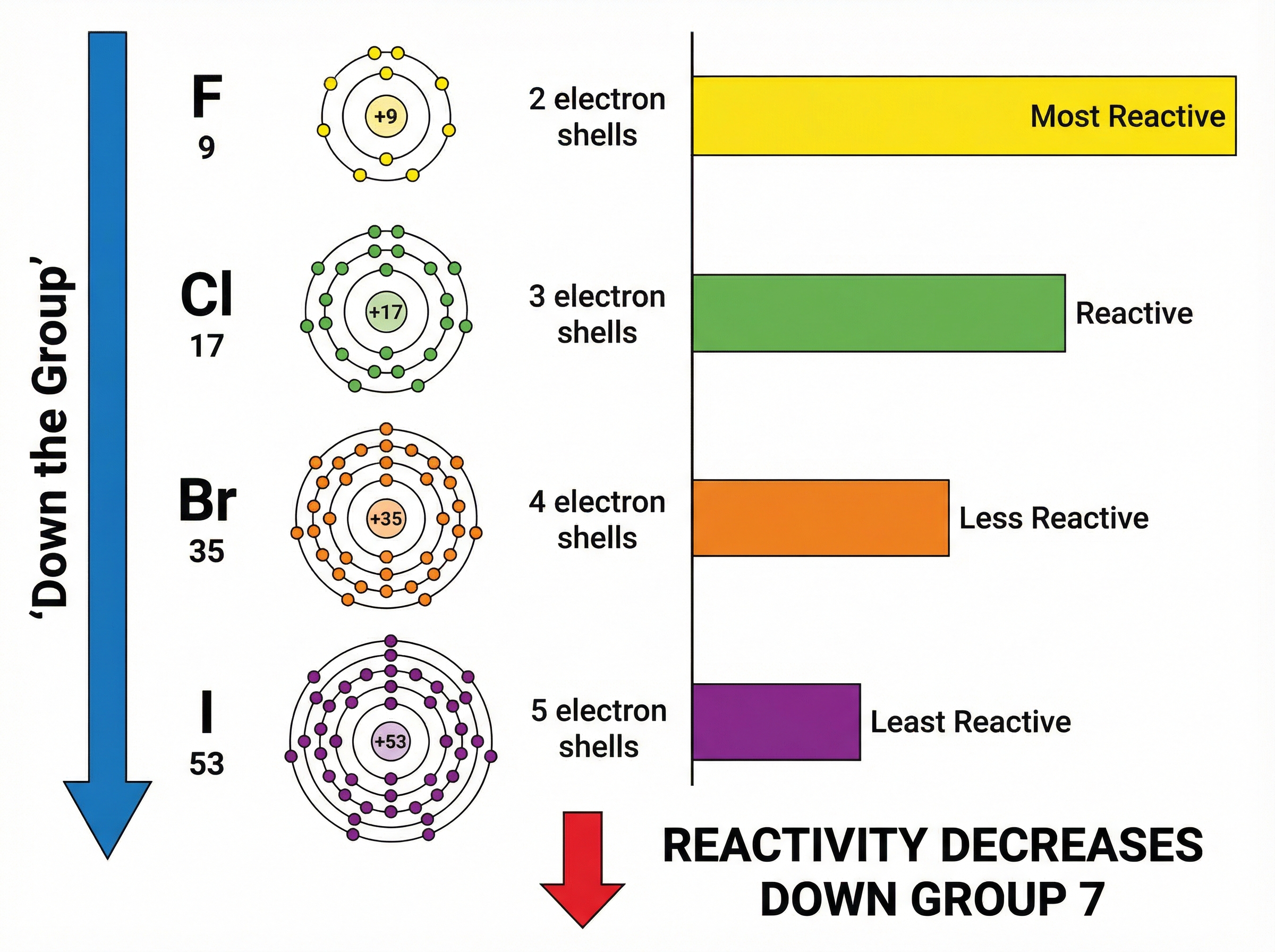
Unlike Group 1 metals, the reactivity of Group 7 halogens **decreases** as you go down the group. Fluorine is the most reactive, and iodine is the least reactive of the common halogens.
**Why does this happen?** All halogens have seven electrons in their outer shell and need to gain one electron to achieve a stable, full outer shell (a halide ion with a 1- charge). The reactivity depends on how easily they can attract this incoming electron.
As you go down the group:
1. The atoms have more electron shells.
2. The outer shell is further from the positive nucleus (increased atomic radius).
3. There is greater **shielding** from the inner electron shells.
4. Therefore, the electrostatic attraction from the nucleus to the incoming electron is weaker.
Because it is harder to attract and gain the extra electron, the element is less reactive. Examiners will look for the term "shielding" or "shielding effect" to award full marks in explanation questions.
### Concept 3: Displacement Reactions and Redox
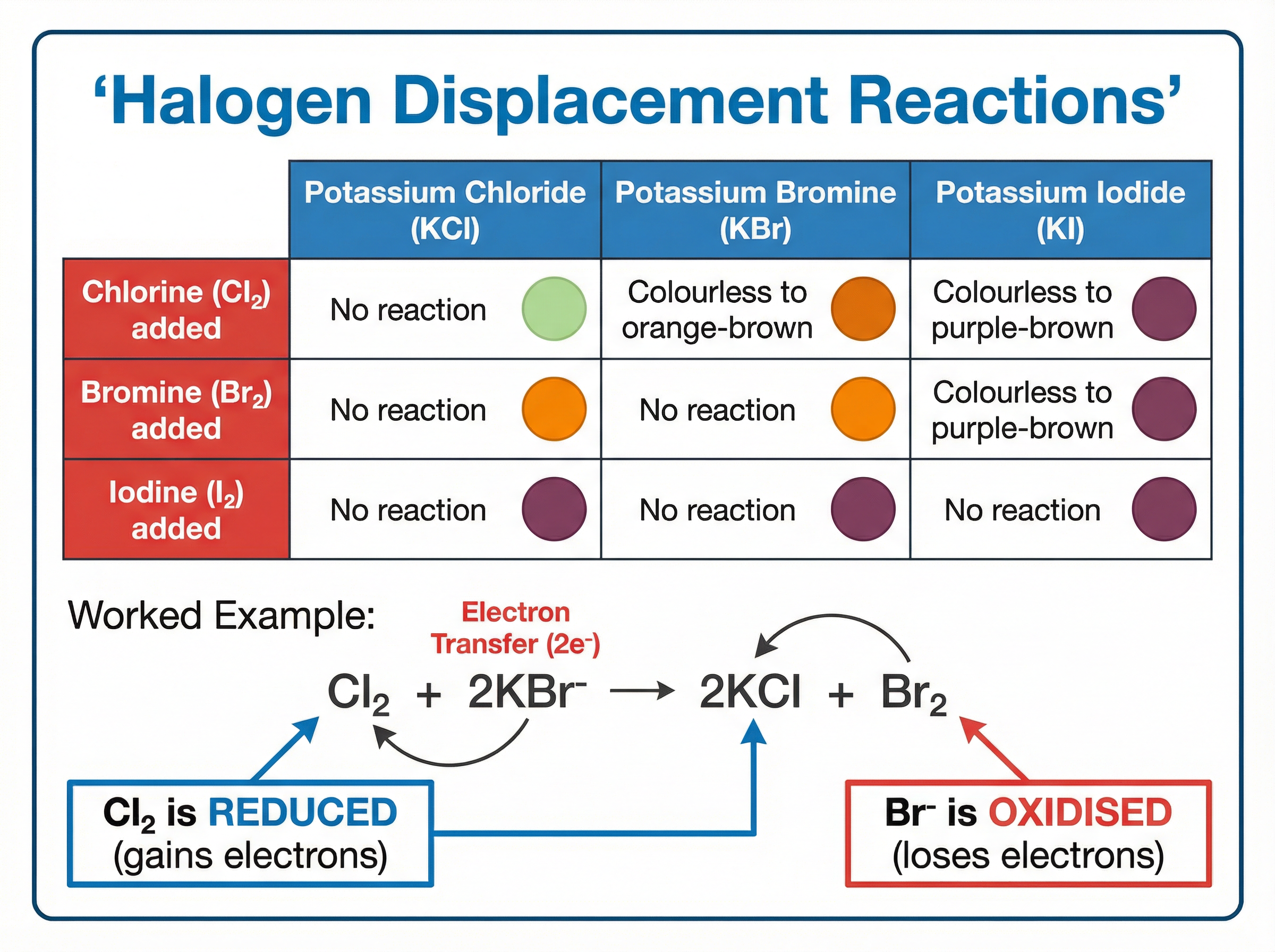
A more reactive halogen will displace a less reactive halogen from an aqueous solution of its halide salt.
For example, if you add chlorine water to potassium bromide solution, the solution turns orange-brown. The more reactive chlorine displaces the less reactive bromide ions, forming potassium chloride and bromine.
**Equation:** $Cl_{2(aq)} + 2KBr_{(aq)} \rightarrow 2KCl_{(aq)} + Br_{2(aq)}$
These are **redox reactions** (reduction-oxidation). Examiners require you to explain them in terms of electron transfer:
* **Reduction:** Gain of electrons. The halogen molecules gain electrons to form halide ions. (e.g., $Cl_2 + 2e^- \rightarrow 2Cl^-$)
* **Oxidation:** Loss of electrons. The halide ions lose electrons to form halogen molecules. (e.g., $2Br^- \rightarrow Br_2 + 2e^-$)
In the example above, chlorine is reduced (it is the oxidising agent), and bromide is oxidised (it is the reducing agent). The potassium ions are spectator ions and do not participate in the electron transfer.
### Concept 4: Reactions with Metals and Hydrogen
**Reactions with Metals:** Halogens react vigorously with metals to form ionic compounds called metal halides. For example, sodium reacts with chlorine gas to form sodium chloride (table salt).
$2Na_{(s)} + Cl_{2(g)} \rightarrow 2NaCl_{(s)}$
The vigour of the reaction decreases down the group, following the reactivity trend.
**Reactions with Hydrogen:** Halogens react with hydrogen gas to form hydrogen halides (e.g., hydrogen chloride, $HCl$).
$H_{2(g)} + Cl_{2(g)} \rightarrow 2HCl_{(g)}$
These hydrogen halides are gases at room temperature, but they are highly soluble in water. When they dissolve, they dissociate to form acidic solutions (e.g., hydrochloric acid).
### Concept 5: Testing for Chlorine
The chemical test for chlorine gas is a standard required recall point.
**Test:** Insert damp blue (or red) litmus paper into the gas.
**Result:** The litmus paper may briefly turn red (as chlorine dissolves to form an acidic solution) but will then be **bleached white**.
## Mathematical/Scientific Relationships
There are no specific mathematical formulas to memorise for this topic, but you must be proficient in writing balanced symbol equations and ionic equations, ensuring state symbols are correct.
* **Diatomic Molecules:** Always write halogens as $X_2$ (e.g., $Cl_2$).
* **Halide Ions:** Always write halide ions with a 1- charge (e.g., $Cl^-$).
* **Ionic Equations:** Exclude spectator ions. For displacement: $X_2 + 2Y^- \rightarrow 2X^- + Y_2$ (where X is more reactive than Y).
## Practical Applications
* **Chlorine in Water Treatment:** Chlorine is added to drinking water and swimming pools to kill bacteria and sterilise the water, preventing diseases like cholera.
* **Iodine as an Antiseptic:** Iodine solutions are used as antiseptics to clean wounds before surgery.
* **Fluoride in Toothpaste:** Fluoride ions are added to toothpaste and some water supplies to strengthen tooth enamel and prevent decay.