Groups in the periodic table — Edexcel GCSE Study Guide
Exam Board: Edexcel | Level: GCSE
Group 1 (Alkali Metals) is one of the most frequently tested topics in GCSE Chemistry, focusing on dramatic reactions with water and the all-important trend in reactivity. Mastering how electronic structure explains these trends will unlock high-mark explanation questions.
## Overview
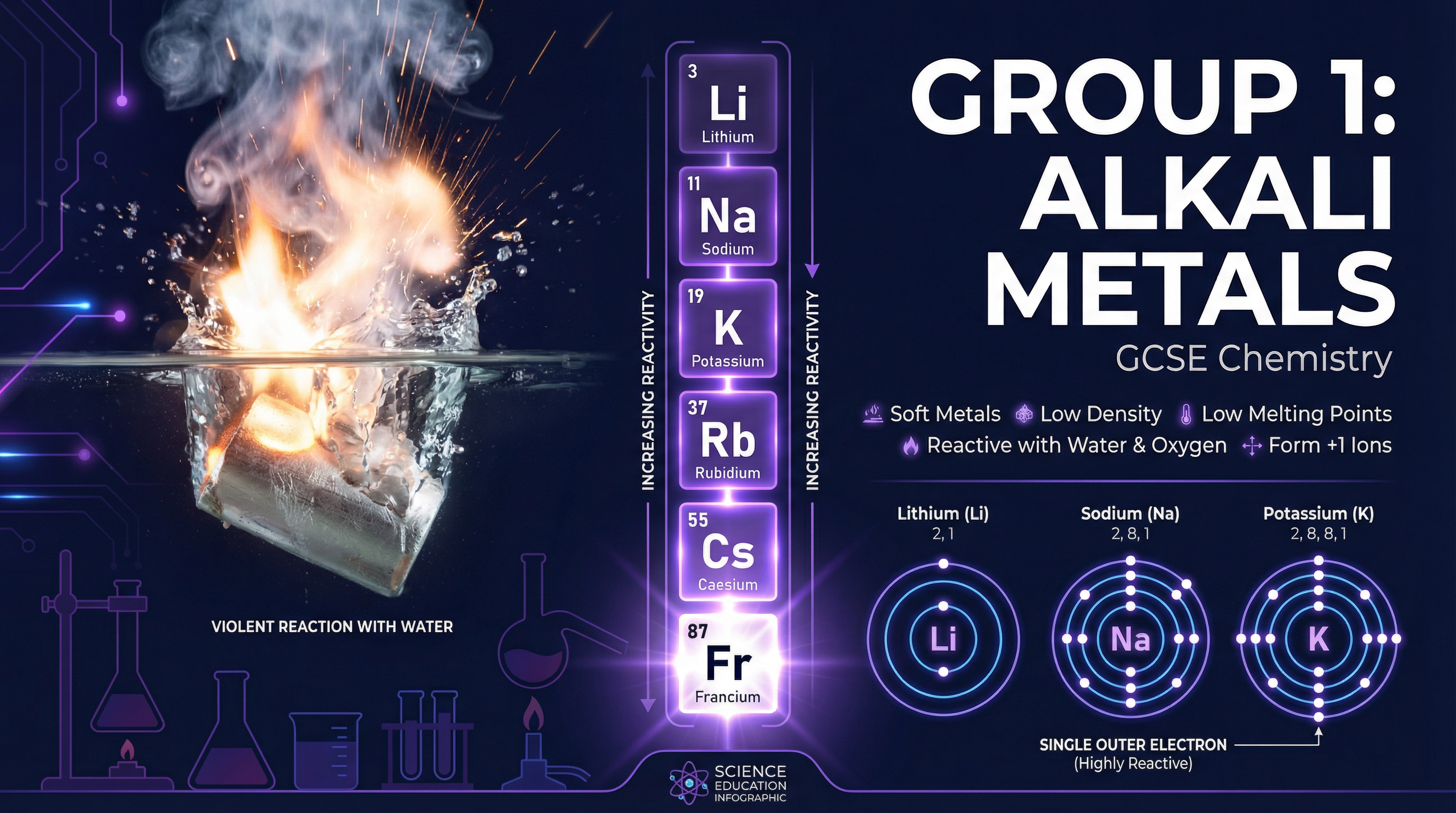
Group 1 of the periodic table contains the **alkali metals**: Lithium (Li), Sodium (Na), Potassium (K), Rubidium (Rb), Caesium (Cs), and Francium (Fr). This topic is fundamental to GCSE Chemistry because it perfectly illustrates how an element's position in the periodic table (and its electronic structure) dictates its chemical behaviour.
Examiners frequently use Group 1 to test your understanding of trends. You will need to describe their physical properties, write balanced equations for their reactions with water, and—crucially—explain *why* they become more reactive as you go down the group. This topic links heavily to atomic structure, ion formation, and chemical equations.

## Key Concepts
### Concept 1: Physical Properties of Alkali Metals
Unlike typical transition metals (like iron or copper), alkali metals have unusual physical properties. They are **soft** enough to be cut with a scalpel, exposing a shiny surface that rapidly tarnishes (oxidises) in the air. They have unusually **low melting and boiling points**, which decrease as you go down the group. They also have **low densities**; in fact, lithium, sodium, and potassium are less dense than water, which is why they float during reactions.
**Example**: Potassium has a melting point of just 63°C, meaning it would melt in a hot cup of tea!
### Concept 2: Reactions with Water
When alkali metals react with water, they produce a **metal hydroxide** (which is alkaline, hence the name) and **hydrogen gas**. The general word equation is:
`Alkali Metal + Water → Metal Hydroxide + Hydrogen`
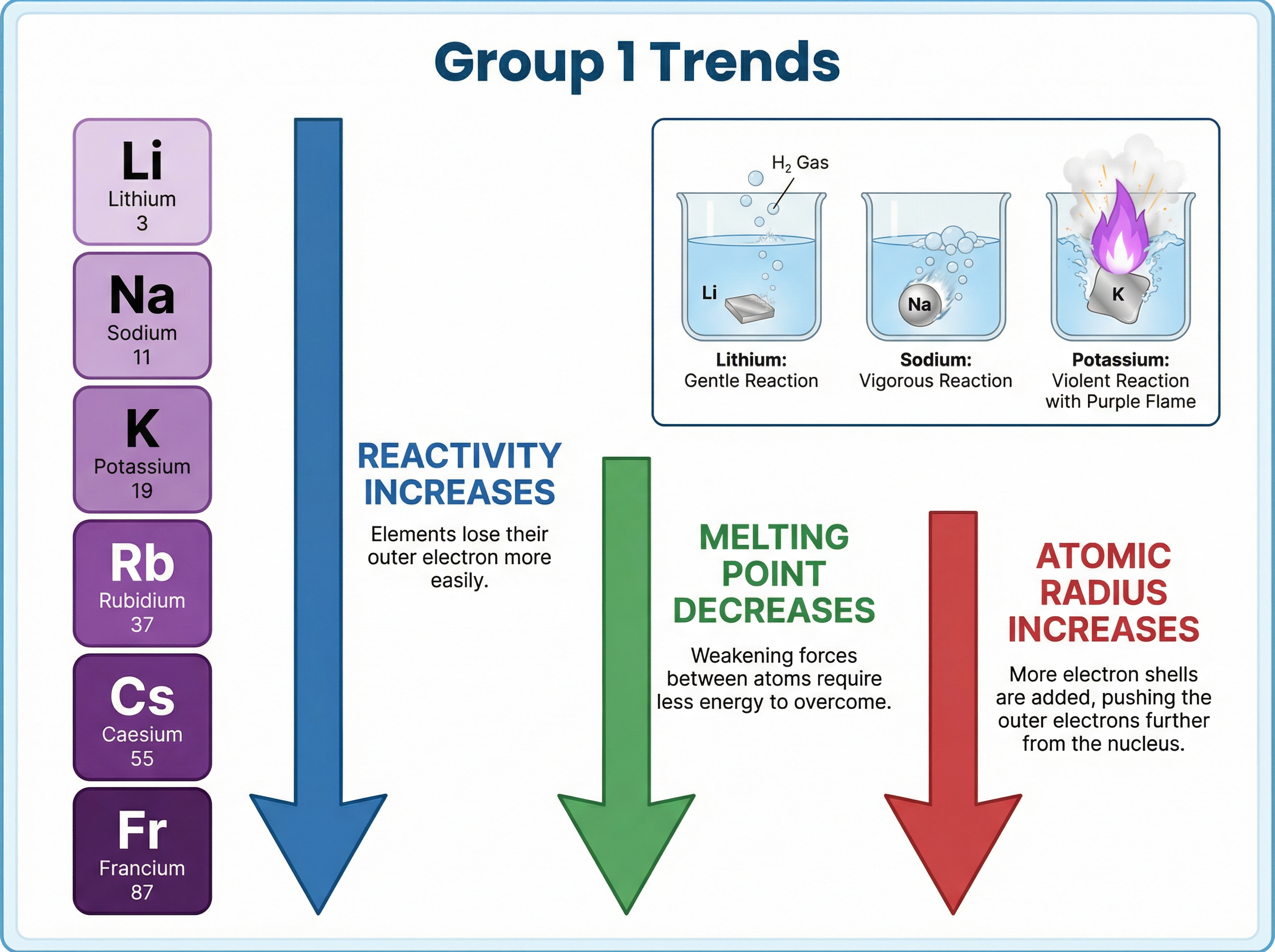
The reactions become increasingly vigorous down the group:
- **Lithium**: Fizzes steadily, floats, and gradually disappears.
- **Sodium**: Fizzes rapidly, melts into a sphere, and moves quickly across the surface.
- **Potassium**: Reacts violently, ignites instantly with a **lilac flame**, and moves very rapidly.
### Concept 3: The Trend in Reactivity (The 'Why')
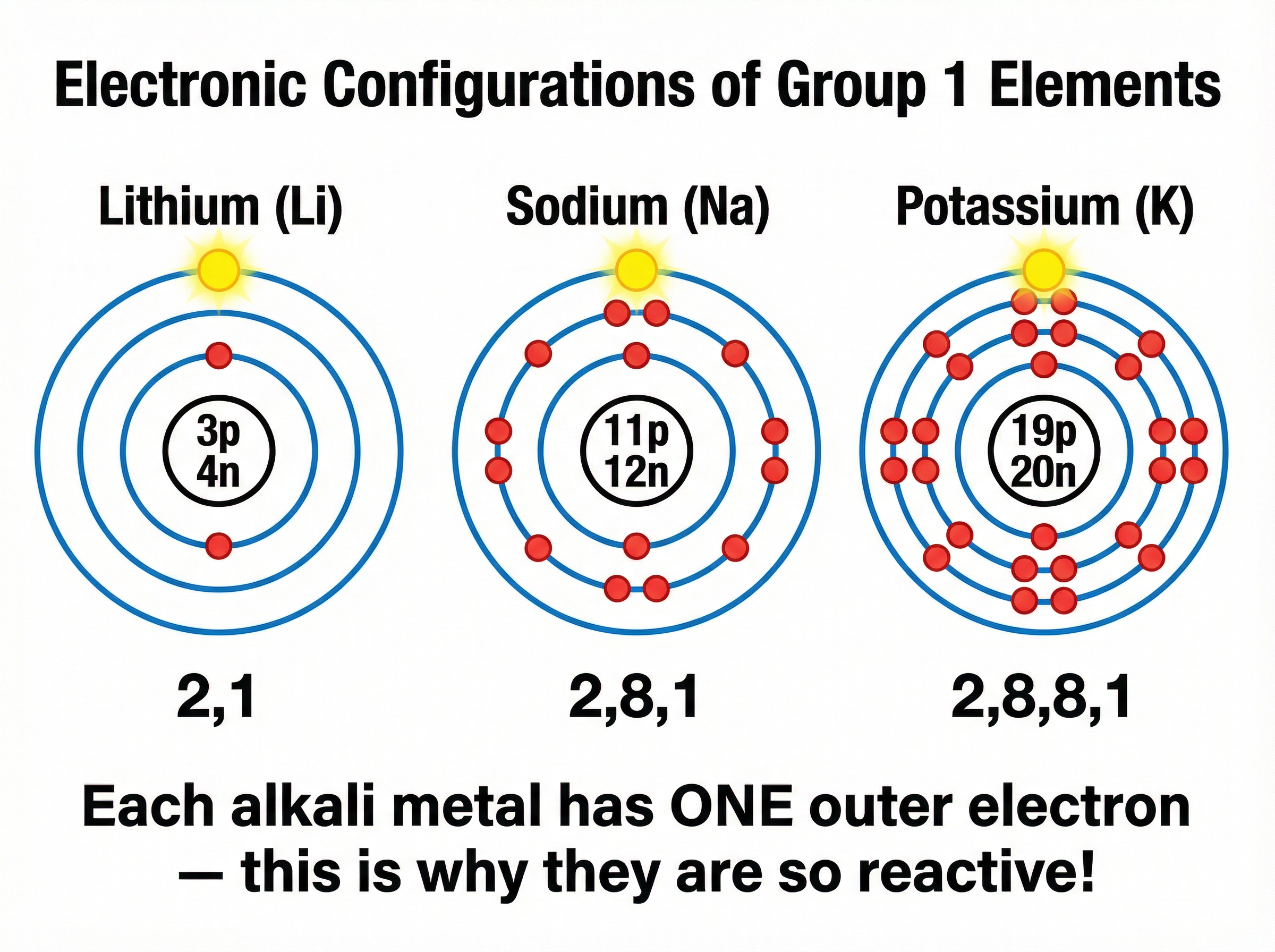
This is the most important concept for 4-6 mark explanation questions. All Group 1 metals have **one electron in their outer shell**. To react, they must lose this electron to form a 1+ ion.
As you go down the group (from Li to Fr):
1. The atoms get larger (more electron shells).
2. The outer electron is **further from the nucleus**.
3. There is more **shielding** from inner electron shells.
4. Therefore, the electrostatic attraction between the positive nucleus and the negative outer electron is **weaker**.
5. The outer electron is **lost more easily**, making the element **more reactive**.
## Mathematical/Scientific Relationships
**Balanced Chemical Equations:**
You must be able to write balanced symbol equations for these reactions. The ratio is always 2:2 → 2:1.
- Lithium: `2Li(s) + 2H₂O(l) → 2LiOH(aq) + H₂(g)`
- Sodium: `2Na(s) + 2H₂O(l) → 2NaOH(aq) + H₂(g)`
- Potassium: `2K(s) + 2H₂O(l) → 2KOH(aq) + H₂(g)`
*Note the state symbols: the metal is solid (s), water is liquid (l), the hydroxide dissolves to form an aqueous solution (aq), and hydrogen is a gas (g).*
## Practical Applications
While you won't do these highly reactive experiments yourself, teachers demonstrate them. The addition of Universal Indicator to the water before the reaction is a common exam context. Because a metal hydroxide is formed, the indicator will turn **purple/blue**, proving an alkaline solution (pH 10-14) has been created.