Groups in the periodic table — Edexcel GCSE Study Guide
Exam Board: Edexcel | Level: GCSE
Group 0 elements, the noble gases, are the most unreactive elements on the periodic table due to their full outer electron shells. Understanding why they are inert and how their physical properties change down the group is essential for securing top marks in GCSE Chemistry.
## Overview
Group 0, located on the far right of the periodic table, contains the noble gases: Helium (He), Neon (Ne), Argon (Ar), Krypton (Kr), Xenon (Xe), and Radon (Rn). These elements are renowned for their chemical inertness—they almost never react with other elements to form compounds. This unique characteristic makes them incredibly useful in real-world applications where non-reactive environments are essential, from light bulbs to MRI scanners.
In your GCSE Chemistry exam, questions on Group 0 frequently focus on explaining *why* they are unreactive using electronic structure, describing the trends in their physical properties as you go down the group, and linking these properties to their specific uses. Examiners often use noble gases to test your understanding of fundamental chemical principles, such as atomic structure and intermolecular forces.

## Key Concepts
### Concept 1: Electronic Structure and Inertness
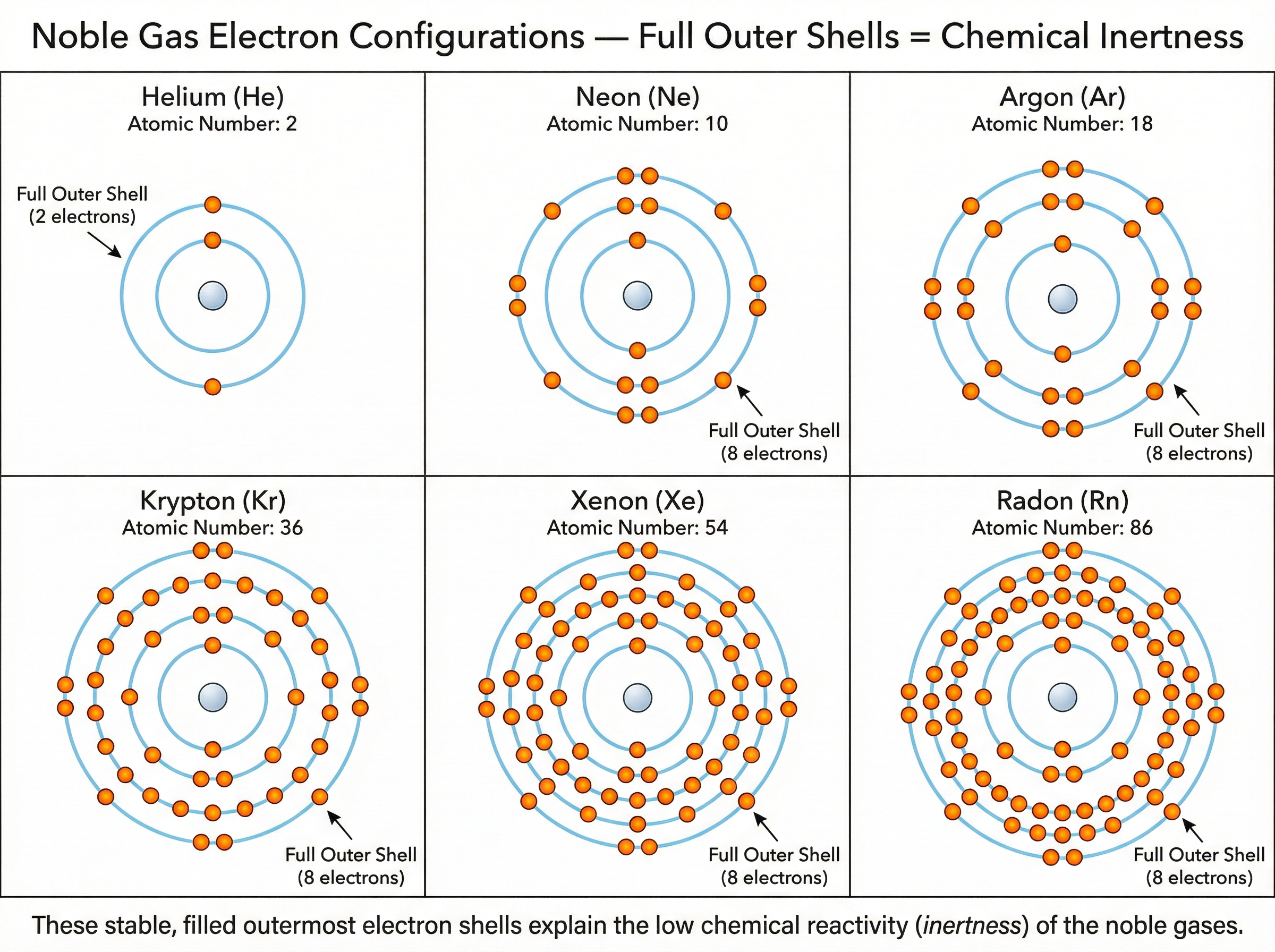
The defining feature of all noble gases is their electronic configuration. Every element in Group 0 has a **full outer electron shell**. For helium, this means two electrons in its single shell. For neon, it is eight electrons in its second shell (2,8), and for argon, it is eight electrons in its third shell (2,8,8).
In chemistry, atoms react—by gaining, losing, or sharing electrons—to achieve a stable, full outer shell. Because noble gases already possess this full outer shell, they have no tendency to transfer or share electrons. Consequently, they exist as single, monatomic atoms rather than molecules.
**Exam Tip**: When asked to explain their lack of reactivity, you must explicitly state that they have a "full outer electron shell". Saying they are "stable" or "happy" will not earn you the mark.
### Concept 2: Trends in Physical Properties
While noble gases share chemical inertness, their physical properties exhibit clear patterns as you move down the group from helium to radon.
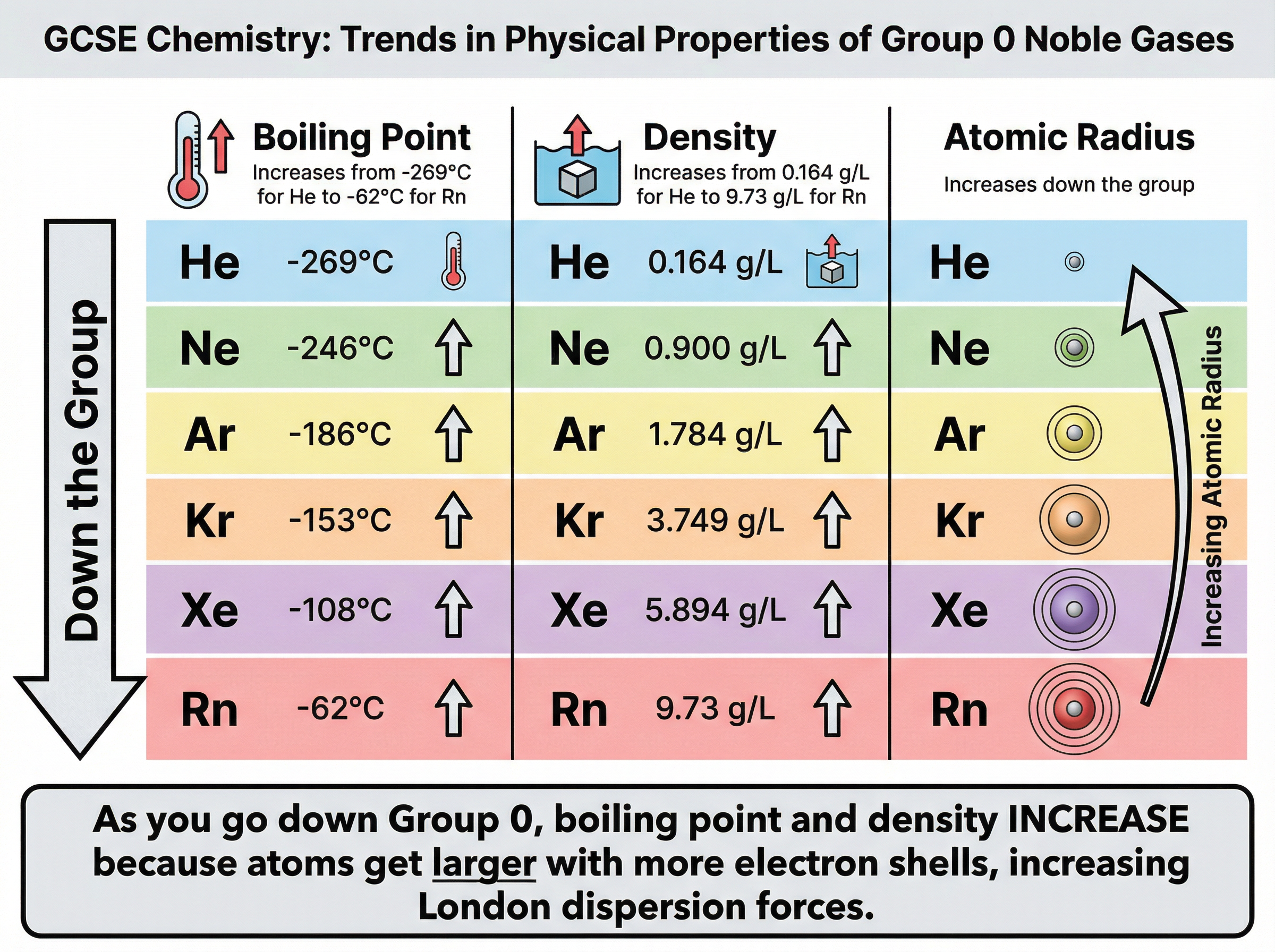
As you go down Group 0, the **boiling point** increases. Helium has the lowest boiling point of any element at -269°C, while radon boils at -62°C. This occurs because the atoms become larger as they gain more electron shells. Larger atoms have more electrons, which leads to stronger intermolecular forces (London dispersion forces) between the atoms. Stronger forces require more thermal energy to overcome, resulting in a higher boiling point.
Similarly, the **density** of the noble gases increases down the group. This is due to the increasing atomic mass being packed into the atomic volume. Helium is much less dense than air, which is why helium balloons float, whereas heavier noble gases like krypton and xenon are significantly denser than air.
### Concept 3: Linking Properties to Uses
Examiners love to test your ability to apply knowledge by linking the properties of noble gases to their everyday uses.
- **Helium** is used in party balloons and airships because it has a very low density (lighter than air) and is non-flammable, making it a much safer alternative to hydrogen. Its extremely low boiling point also makes it ideal for cooling superconducting magnets in MRI scanners.
- **Neon** is famous for its use in advertising signs. When an electric current is passed through neon gas under low pressure, it emits a characteristic bright red-orange glow.
- **Argon** is used to fill incandescent light bulbs. Because it is inert, it prevents the hot tungsten filament from reacting with oxygen and burning away. It is also used as a shielding gas in welding to stop the hot metal from oxidising in the air.

## Mathematical/Scientific Relationships
While Group 0 does not involve complex mathematical formulas, you must be able to interpret data tables showing physical properties. You may be asked to predict the boiling point or density of a noble gas based on the values of the elements above and below it in the group.
When predicting a value, find the mean of the values for the element above and the element below. For example, if you know the boiling point of Neon and Krypton, you can estimate the boiling point of Argon by averaging the two.
## Practical Applications
Understanding noble gases has profound real-world impacts. The discovery of argon by Lord Rayleigh and William Ramsay in 1894 solved the mystery of why nitrogen extracted from the air was slightly heavier than nitrogen obtained from chemical compounds—the air contained a small amount of this unreactive, heavier gas. Today, the inert nature of these gases is exploited in everything from preserving historical documents (like the US Declaration of Independence, which is encased in argon) to providing the bright flashes in high-speed photography (using krypton).