Key concepts in chemistry — Edexcel GCSE Study Guide
Exam Board: Edexcel | Level: GCSE
Master the fundamentals of covalent bonding, from drawing precise dot and cross diagrams to explaining the physical properties of simple molecules. This core topic forms the foundation for understanding how non-metals interact, a guaranteed source of marks in your chemistry exams.
## Overview
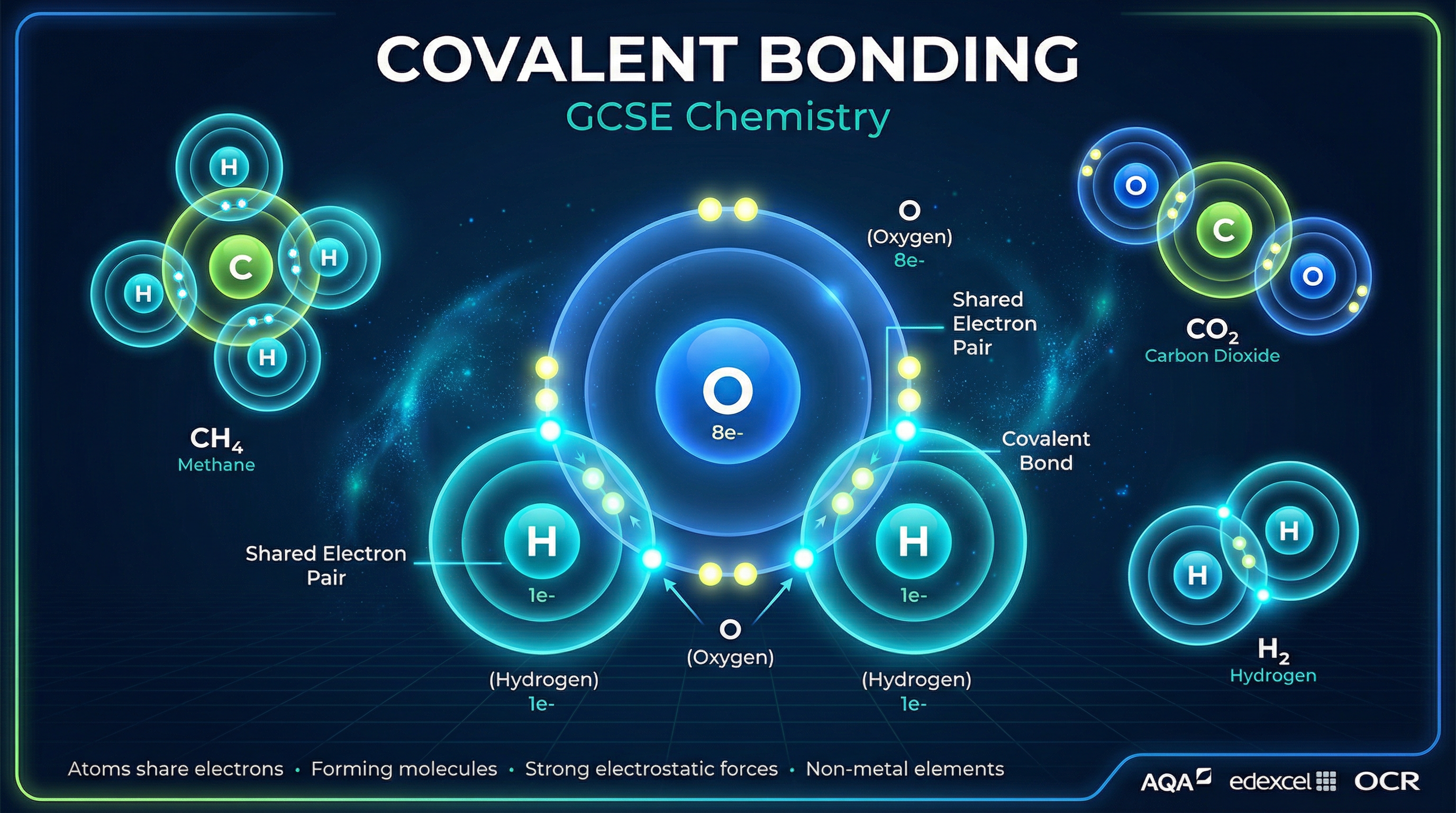
Covalent bonding is a fundamental concept in chemistry that explains how non-metal elements interact to form the vast array of molecules in our universe. In this topic, we explore the mechanics of electron sharing, a process driven by atoms seeking the stability of a full outer electron shell. Understanding covalent bonding is crucial because it not only explains the structure of vital substances like water ($H_2O$) and carbon dioxide ($CO_2$), but it also underpins organic chemistry and biochemistry.
Examiners frequently test this topic through drawing tasks, requiring you to construct precise dot and cross diagrams, and through explanatory questions where you must link molecular structure to physical properties. A classic pitfall is confusing the strong covalent bonds within a molecule with the weak intermolecular forces between molecules. By mastering this distinction, you will secure high marks in extended response questions.

## Key Concepts
### Concept 1: The Nature of the Covalent Bond
A covalent bond is defined as a **shared pair of electrons between two non-metal atoms**. Atoms are most stable when they have a full outer shell of electrons (typically eight, known as an octet, except for hydrogen which requires two). When non-metal atoms react, neither is willing to fully give up an electron (as occurs in ionic bonding). Instead, they compromise by sharing electrons in the overlapping region of their outer shells.
The bond itself is the strong electrostatic attraction between the positively charged nuclei of the bonded atoms and the negatively charged shared pair of electrons situated between them.
**Example**: In a molecule of hydrogen ($H_2$), two hydrogen atoms, each with one electron, share their electrons to form a single covalent bond. Both atoms now have a full outer shell of two electrons, achieving stability.
### Concept 2: Representing Molecules with Dot and Cross Diagrams
Dot and cross diagrams are the standard method for representing covalent bonds in exams. The electrons from one atom are drawn as dots ($\bullet$), and the electrons from the other atom are drawn as crosses ($\times$). This visual distinction helps examiners see that you understand where the shared electrons originated.
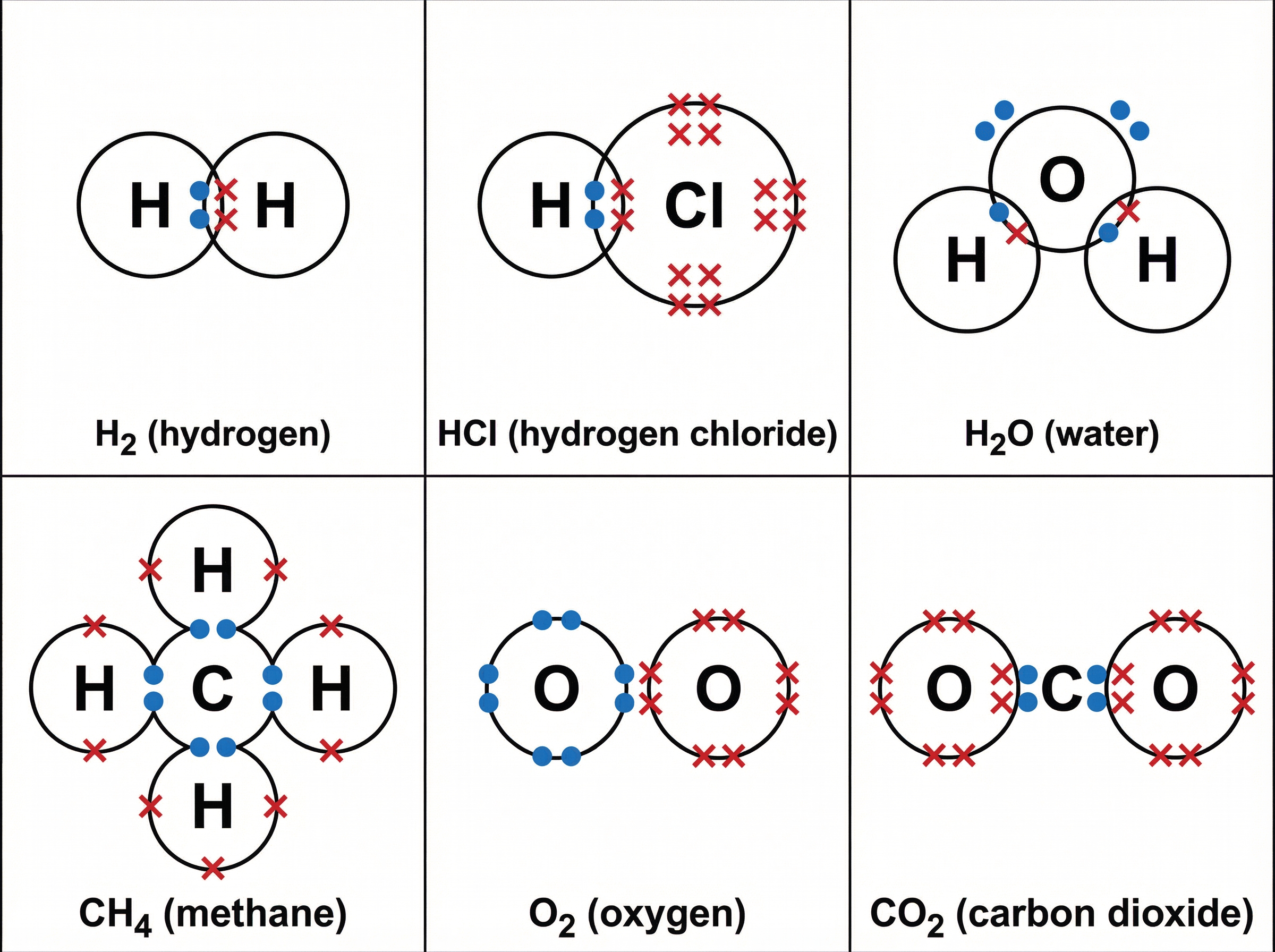
When drawing these diagrams, you must ensure:
1. The shared electrons are placed clearly in the overlapping region of the atomic circles.
2. **All** outer shell electrons are shown, not just the bonding ones.
3. Non-bonding pairs of electrons (lone pairs) are drawn on the outer edge of the respective atom's circle.
For example, in water ($H_2O$), oxygen shares one electron with each of the two hydrogen atoms. The oxygen atom originally has six outer electrons. Two are involved in bonding, leaving four electrons (two lone pairs) which must be clearly drawn on the oxygen atom.
### Concept 3: Simple Molecular Structures and Their Properties
Substances consisting of small molecules (like $H_2O$, $CH_4$, $CO_2$) have simple molecular structures. The covalent bonds holding the atoms together *within* the molecule are very strong. However, the forces of attraction *between* separate molecules—known as **intermolecular forces**—are very weak.
When you boil water, you are not breaking the strong O-H covalent bonds; you are only overcoming the weak intermolecular forces separating the $H_2O$ molecules. Because these forces are weak, very little thermal energy is required to overcome them. Therefore, simple molecular substances have **low melting and boiling points** and are often gases or liquids at room temperature.
Furthermore, simple molecular substances **do not conduct electricity**. Electrical conductivity requires the presence of mobile charged particles (delocalised electrons or free ions). In simple molecules, all electrons are tightly held in covalent bonds or lone pairs, and there is no overall electrical charge.
### Concept 4: The Scale of Atoms and Molecules
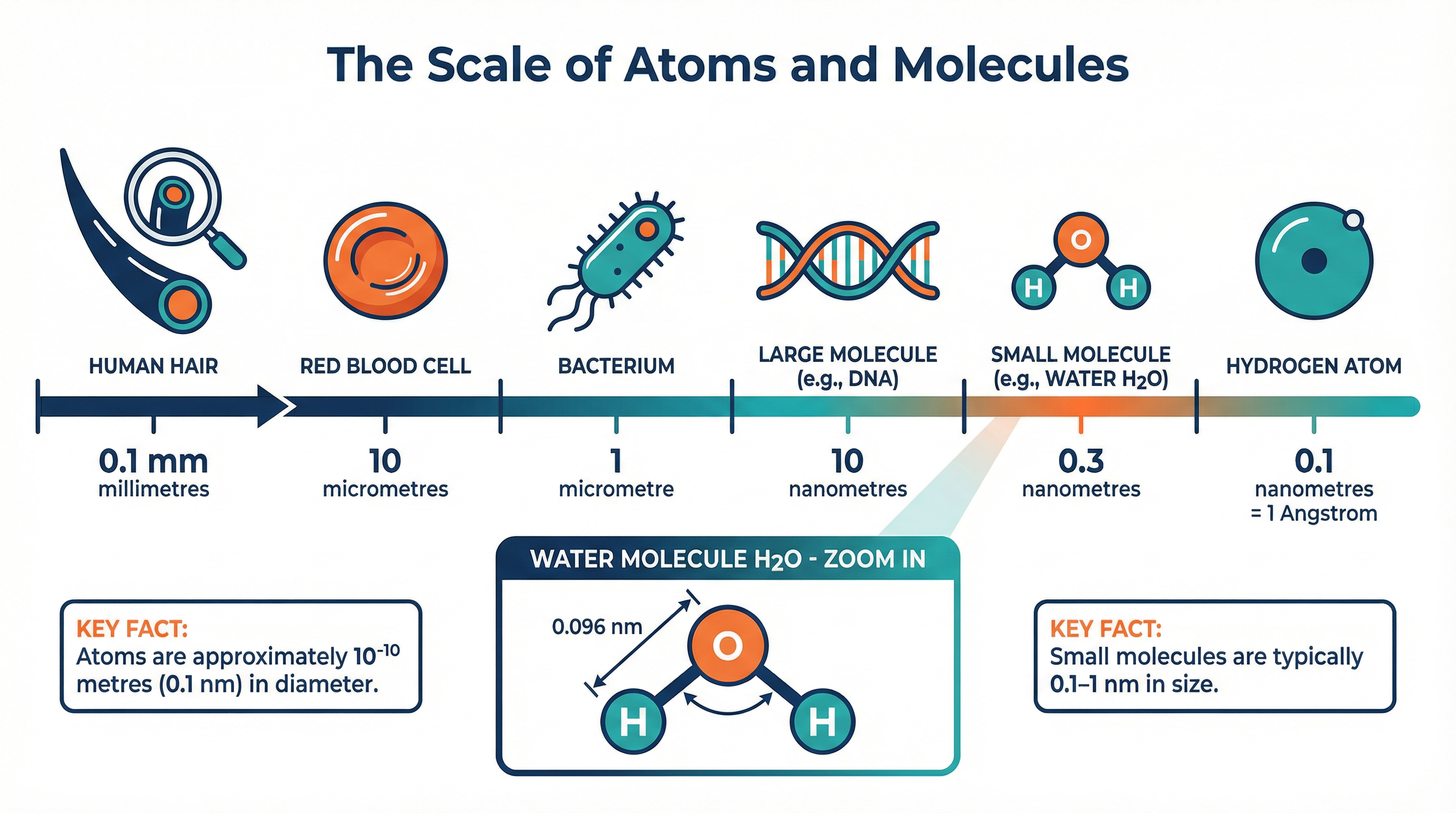
To appreciate chemistry, you must understand the order of magnitude of the particles involved. Atoms are incredibly small, with a typical radius of about $0.1 \text{ nanometres}$ ($1 \times 10^{-10} \text{ m}$). Small molecules, such as water or methane, are on a similar scale, typically ranging from $0.1$ to $1 \text{ nm}$.
Examiners expect you to know this standard form representation ($10^{-10} \text{ m}$) and be able to compare it to larger structures, such as polymers or giant covalent lattices, which are orders of magnitude larger.
## Mathematical/Scientific Relationships
There are no complex formulas to calculate in this specific subtopic, but you must understand the numerical relationship of the octet rule:
- Number of electrons needed for a full shell = Number of covalent bonds the atom typically forms.
- E.g., Carbon is in Group 4 (4 outer electrons). It needs 4 more to reach 8. Therefore, carbon forms 4 covalent bonds.
- E.g., Oxygen is in Group 6 (6 outer electrons). It needs 2 more to reach 8. Therefore, oxygen forms 2 covalent bonds.
## Practical Applications
The principles of covalent bonding are the basis of the entire pharmaceutical industry, plastics, and biochemistry. The specific shapes and bonds of molecules like DNA and proteins dictate how our bodies function. The low boiling points of simple molecules are why the oxygen we breathe and the carbon dioxide we exhale are gases at room temperature, enabling respiration.