Key concepts in chemistry — Edexcel GCSE Study Guide
Exam Board: Edexcel | Level: GCSE
Master the fundamental building blocks of Chemistry! This topic covers how atoms bond together to form ionic, simple molecular, giant covalent, and metallic structures, and crucially, how these microscopic bonds dictate the macroscopic properties we observe.
## Overview
Bonding and structure form the very foundation of Chemistry. Understanding how atoms connect to one another is essential because the microscopic arrangement of particles completely dictates the macroscopic properties of a substance—such as whether it will melt easily, dissolve in water, or conduct electricity.
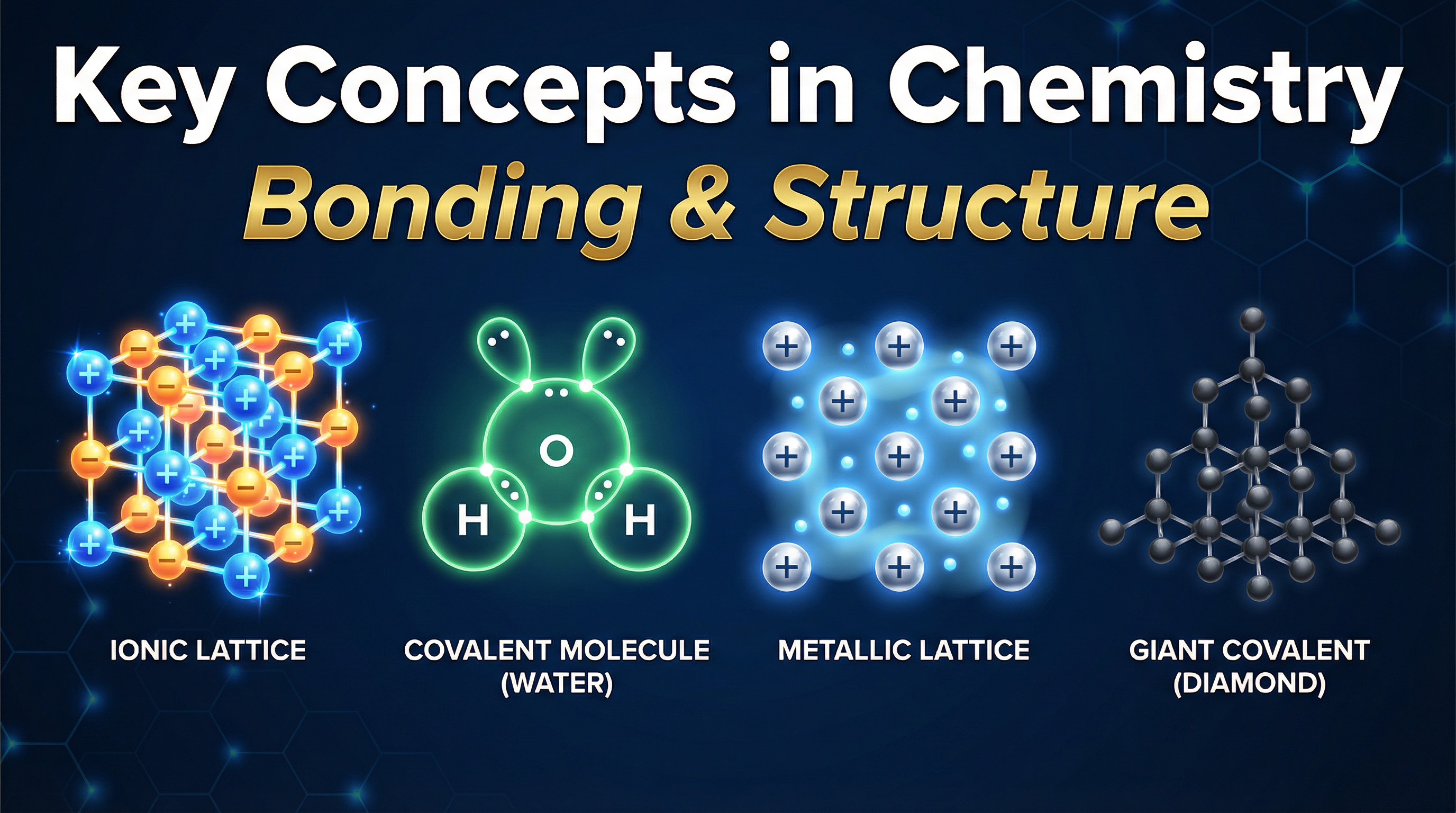
In this topic, we classify all substances into four main structural models: ionic lattices, simple molecular structures, giant covalent structures, and metallic lattices. Examiners love testing this area because it requires logical, step-by-step reasoning linking structure to properties. It also synoptically links to almost every other topic in the specification, from electrolysis and quantitative chemistry to organic chemistry and the reactivity series.
Listen to our comprehensive audio guide for a detailed breakdown of this topic:

## Key Concepts
### Concept 1: Ionic Bonding and Giant Ionic Lattices
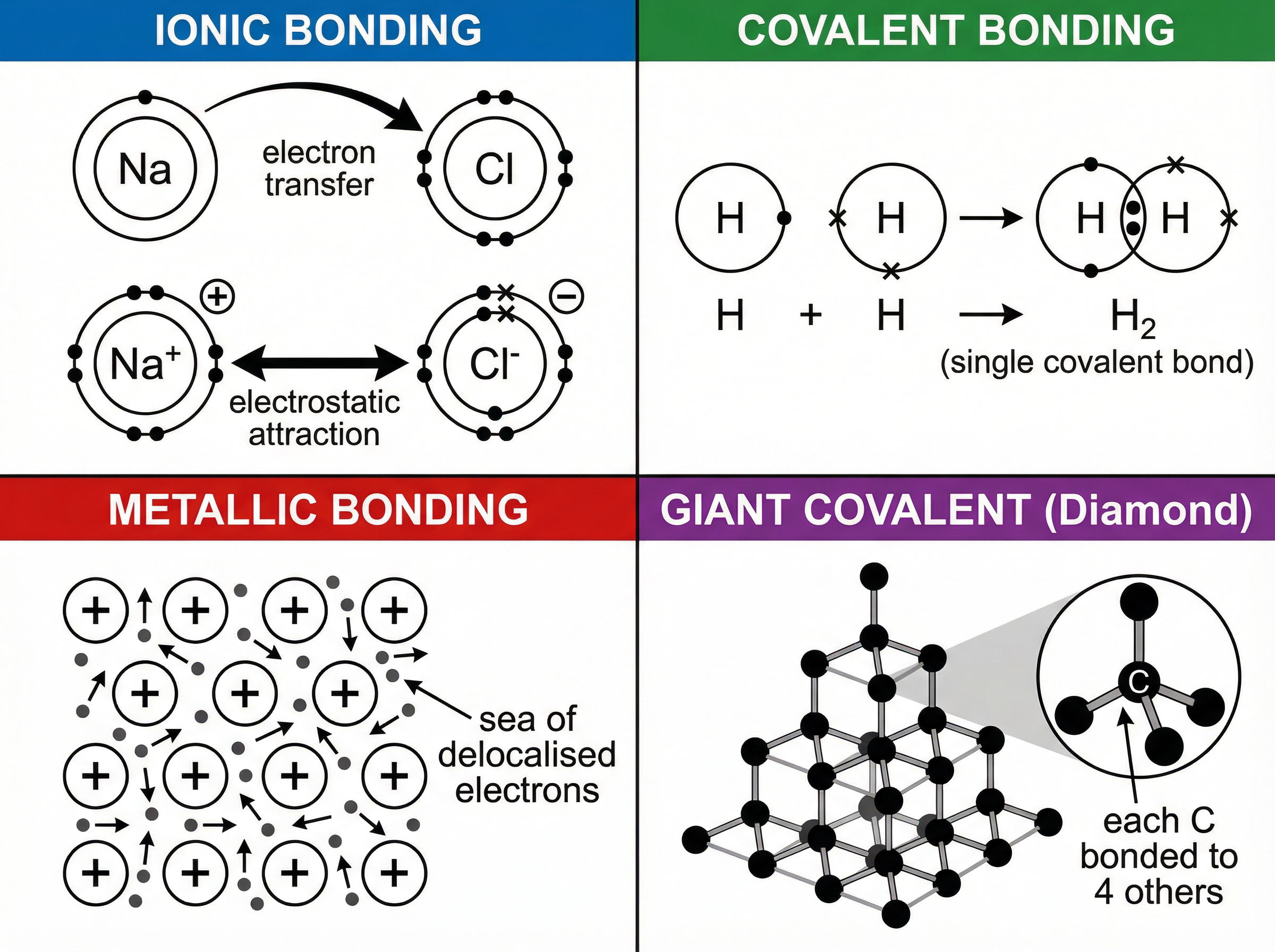
Ionic bonding occurs between a metal and a non-metal. The metal atom loses electrons to form a positively charged ion (cation), while the non-metal atom gains these electrons to form a negatively charged ion (anion).
Crucially, these ions do not exist in isolated pairs. They arrange themselves into a **giant ionic lattice**—a regular, three-dimensional structure of alternating positive and negative ions. The force holding this lattice together is the **strong electrostatic attraction** between oppositely charged ions, acting in all directions.
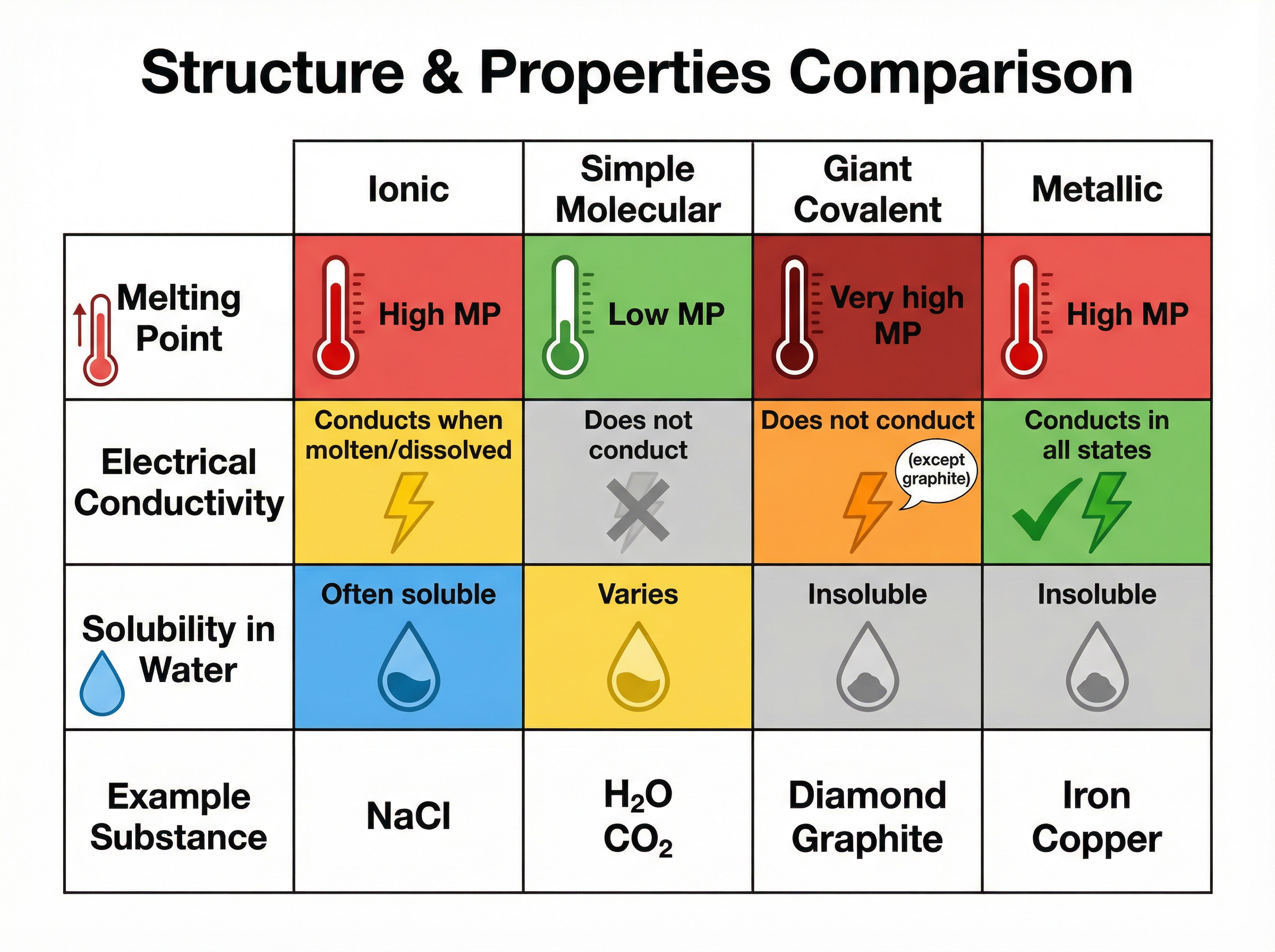
Because these electrostatic forces are very strong and numerous, a large amount of energy is required to overcome them. This explains why ionic compounds have high melting and boiling points.
**Conductivity**: Ionic compounds cannot conduct electricity when solid because the ions are fixed in the lattice and cannot move. However, when molten (melted) or dissolved in water (aqueous), the ions are free to move and carry charge, allowing the substance to conduct electricity.
### Concept 2: Simple Molecular Covalent Structures
Covalent bonding occurs between non-metal atoms that share pairs of electrons to achieve a full outer shell. In simple molecular substances (like water, carbon dioxide, or methane), the atoms within each molecule are held together by strong covalent bonds.
However, the forces *between* the individual molecules—known as **intermolecular forces**—are weak. When you melt or boil a simple molecular substance, you are only overcoming these weak intermolecular forces, not breaking the strong covalent bonds. Therefore, they require little energy to separate, resulting in low melting and boiling points.
**Conductivity**: Simple molecular substances do not conduct electricity in any state because their molecules are uncharged and there are no free electrons or ions to carry a charge.
### Concept 3: Giant Covalent Structures
Some non-metals form giant covalent structures (macromolecules), where millions of atoms are joined by strong covalent bonds in a continuous three-dimensional lattice.
**Diamond**: Each carbon atom forms four strong covalent bonds with other carbon atoms in a rigid tetrahedral arrangement. This makes diamond exceptionally hard with a very high melting point, as millions of covalent bonds must be broken to melt it. It does not conduct electricity because all outer electrons are involved in bonding.
**Graphite**: Each carbon atom forms only three covalent bonds, creating flat hexagonal layers. The layers are held together by weak intermolecular forces, allowing them to slide over one another (making graphite soft and slippery). The fourth outer electron from each carbon atom is **delocalised** and free to move between the layers, allowing graphite to conduct electricity and thermal energy.
### Concept 4: Metallic Bonding
Metals consist of a giant structure of positive metal ions arranged in a regular lattice, surrounded by a "sea" of delocalised electrons. These electrons are free to move throughout the structure.
The metallic bond is the strong electrostatic attraction between the positive metal ions and the negative delocalised electrons. This strong attraction gives metals their generally high melting and boiling points.
**Conductivity**: Metals are excellent conductors of electricity and heat because the delocalised electrons can move freely through the structure, carrying electrical charge or thermal energy.
**Malleability**: Metals are malleable (can be bent and shaped) because the layers of positive ions can slide over each other without breaking the metallic bonds, as the sea of delocalised electrons simply shifts to accommodate the new shape.
## Mathematical/Scientific Relationships
While this topic is primarily descriptive, it connects to chemical formulae:
- **Empirical Formula**: For giant structures (ionic, metallic, giant covalent), the formula represents the simplest ratio of atoms/ions in the lattice (e.g., NaCl means a 1:1 ratio of Na+ to Cl- ions).
- **Molecular Formula**: For simple molecular substances, the formula shows the exact number of atoms in one molecule (e.g., H2O means exactly 2 hydrogen atoms and 1 oxygen atom bonded together).
## Practical Applications
- **Lubricants**: Graphite's sliding layers make it an excellent dry lubricant for machinery where oil cannot be used.
- **Electrical Wiring**: Copper is used for wiring due to its excellent electrical conductivity (thanks to delocalised electrons) and ductility.
- **Cutting Tools**: Diamond's rigid tetrahedral lattice makes it hard enough to be used on the tips of industrial drill bits and saw blades.