Key concepts in chemistry — Edexcel GCSE Study Guide
Exam Board: Edexcel | Level: GCSE
Master the fundamental concept of ionic bonding, where electron transfer creates oppositely charged ions held together by strong electrostatic forces. This topic is guaranteed to appear in your GCSE Chemistry exam and forms the foundation for understanding chemical properties and structures.
## Overview
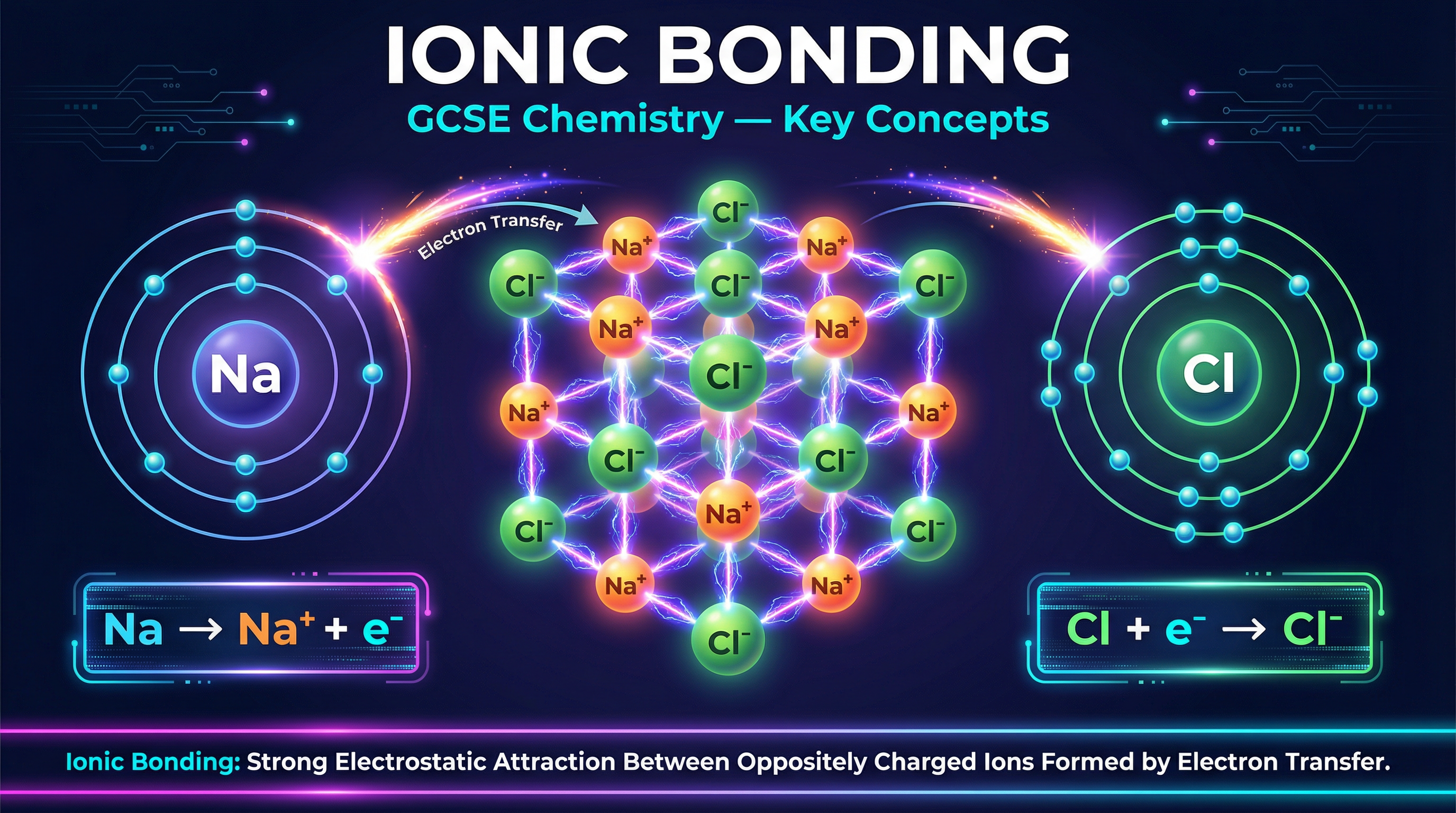
Ionic bonding is a cornerstone of GCSE Chemistry, appearing in virtually every exam paper. It explains how metals and non-metals react to form compounds with entirely new properties. By understanding the transfer of electrons, you can explain why sodium (a highly reactive metal) and chlorine (a toxic gas) combine to form sodium chloride (table salt), a stable solid we eat every day.
Examiners love testing this topic because it links atomic structure to macroscopic properties. You'll frequently see questions asking you to draw dot and cross diagrams, deduce formulae, or explain why ionic compounds have high melting points. Mastering these concepts will secure you significant marks across multiple papers.
## Key Concepts
### Concept 1: Electron Transfer and Ion Formation
Ionic bonding occurs between a metal and a non-metal. Atoms are most stable when they have a full outer electron shell, like the noble gases. To achieve this, metal atoms **lose** electrons from their outer shell to form positively charged ions (cations). Non-metal atoms **gain** these electrons into their outer shell to form negatively charged ions (anions).
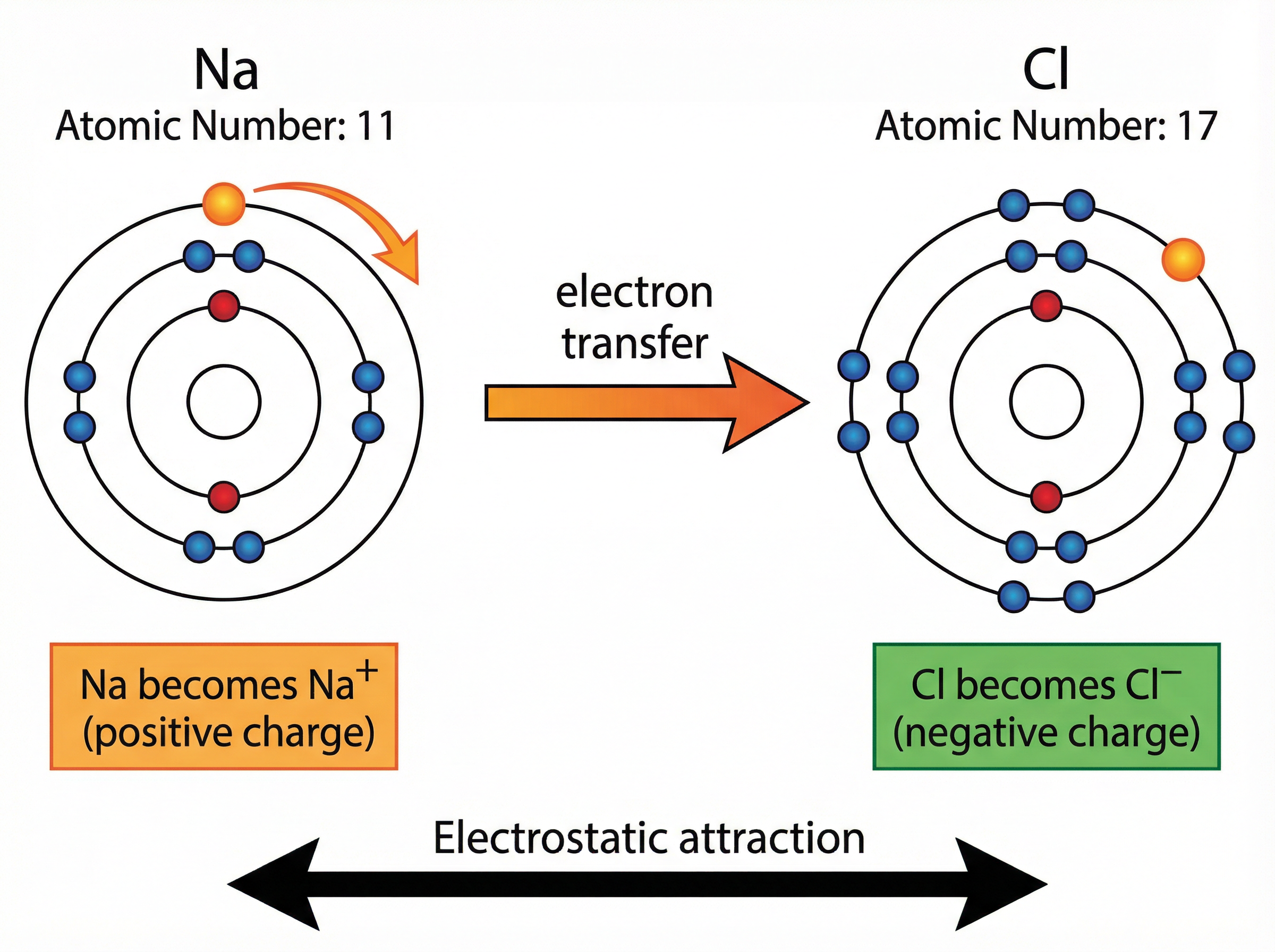
**Example**: Sodium (Na) has the electron configuration 2,8,1. It loses its 1 outer electron to become Na⁺ (2,8). Chlorine (Cl) is 2,8,7. It gains 1 electron to become Cl⁻ (2,8,8). The electron lost by sodium is the exact same electron gained by chlorine.
### Concept 2: Dot and Cross Diagrams
These diagrams show the arrangement of electrons in atoms and ions. Examiners are incredibly strict here: when drawing the ions, you **must** use square brackets around each ion and place the charge outside the top right corner. The electrons from one atom are drawn as dots, and from the other as crosses, proving exactly where the transferred electrons originated.
### Concept 3: The Giant Ionic Lattice
Ionic compounds do not exist as simple molecules. Instead, the oppositely charged ions attract each other in all directions, forming a massive, regular, repeating 3D structure called a **giant ionic lattice**.
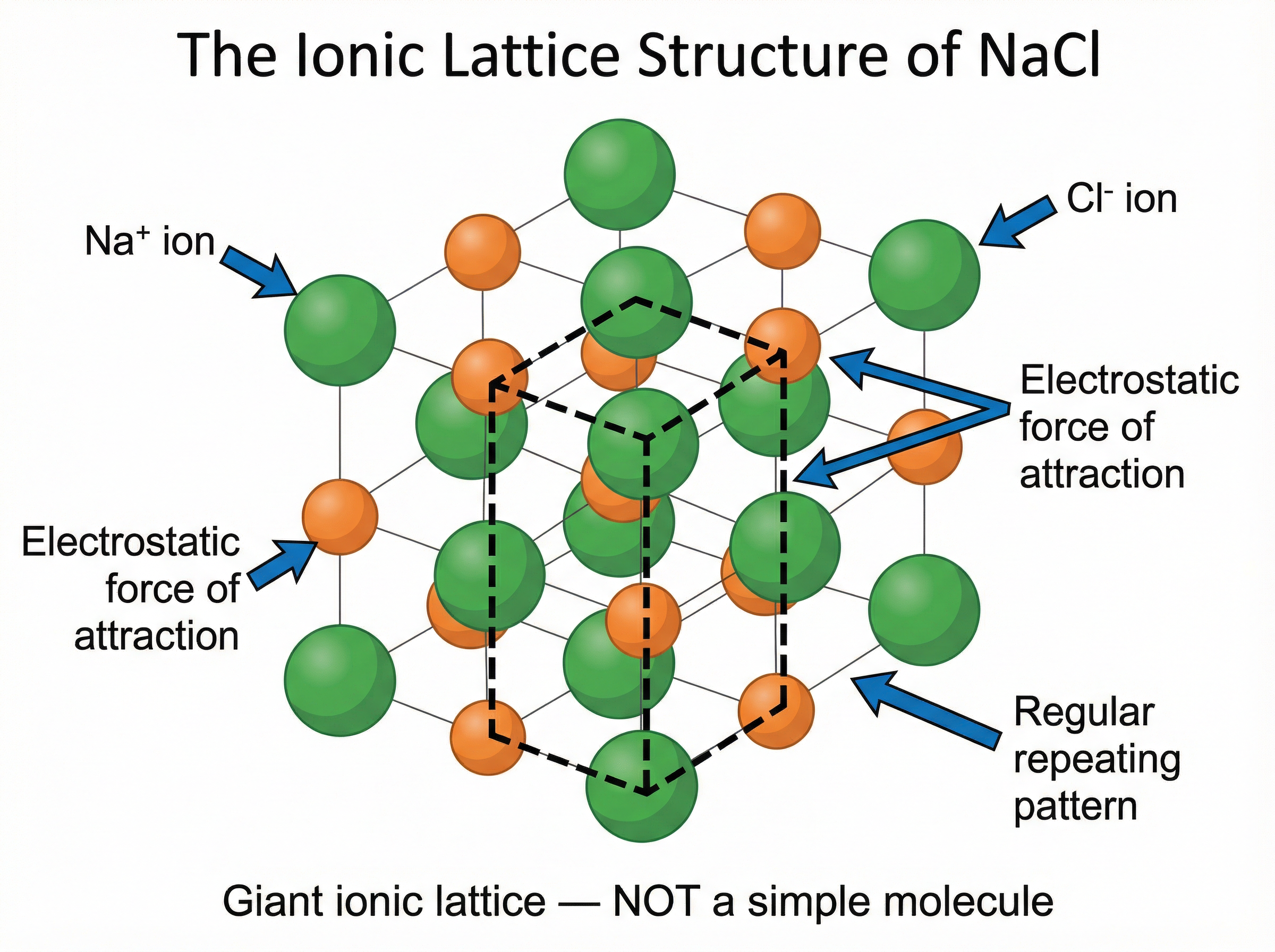
**Example**: In a crystal of sodium chloride, every Na⁺ ion is surrounded by six Cl⁻ ions, and every Cl⁻ ion is surrounded by six Na⁺ ions. The strong electrostatic forces of attraction between these oppositely charged ions hold the entire structure together tightly.
### Concept 4: Deducing Formulae
The formula of an ionic compound tells you the simplest ratio of ions in the lattice. Because compounds must be electrically neutral overall, the total positive charge must exactly balance the total negative charge.
**Example**: Aluminium is in Group 3 (forms Al³⁺). Oxygen is in Group 6 (forms O²⁻). To balance the charges, you need two Al³⁺ ions (total +6) and three O²⁻ ions (total -6). Therefore, the formula is Al₂O₃.
## Mathematical/Scientific Relationships
- **Charge of Group 1 metals**: +1
- **Charge of Group 2 metals**: +2
- **Charge of Group 3 metals**: +3
- **Charge of Group 5 non-metals**: -3
- **Charge of Group 6 non-metals**: -2
- **Charge of Group 7 non-metals**: -1
To find the number of subatomic particles in an ion:
- **Protons** = Atomic number (does not change)
- **Neutrons** = Mass number - Atomic number (does not change)
- **Electrons** = Atomic number - Charge (e.g., for Na⁺: 11 - (+1) = 10 electrons)
## Practical Applications
While there isn't a required practical specifically for forming ionic bonds, you will use ionic compounds constantly. The conductivity of ionic compounds is tested in the electrolysis practicals. Solid ionic compounds (like NaCl) cannot conduct electricity because the ions are fixed in the lattice. However, when melted (molten) or dissolved in water (aqueous), the lattice breaks apart and the ions are free to move and carry charge.
## Podcast Summary
Listen to our 10-minute revision podcast covering all these concepts, common mistakes, and a quick-fire quiz!
