Key concepts in chemistry — Edexcel GCSE Study Guide
Exam Board: Edexcel | Level: GCSE
Master the foundation of all chemistry: the Periodic Table. Discover how Mendeleev's brilliant predictions and the modern atomic number arrangement unlock the secrets of electronic configuration, metals, and non-metals to secure top marks.

## Overview
The Periodic Table is the ultimate cheat sheet for chemists, but to unlock its power, you need to understand how it is built. This topic covers the fascinating history of how the elements were organised, moving from Dmitri Mendeleev's groundbreaking 1869 table to the modern arrangement we use today. It is a cornerstone of GCSE Chemistry because the position of an element in the table dictates its properties, how it reacts, and what type of bonds it forms.
Examiners love to test your understanding of the link between an element's position (its group and period) and its electronic configuration. You will frequently encounter questions asking you to compare Mendeleev's table with the modern one, or to deduce the electron arrangement of the first 20 elements. Mastering this topic provides the foundation for almost every other chemistry module, from bonding to quantitative chemistry.
Listen to the companion podcast for a complete audio walkthrough of this topic:

## Key Concepts
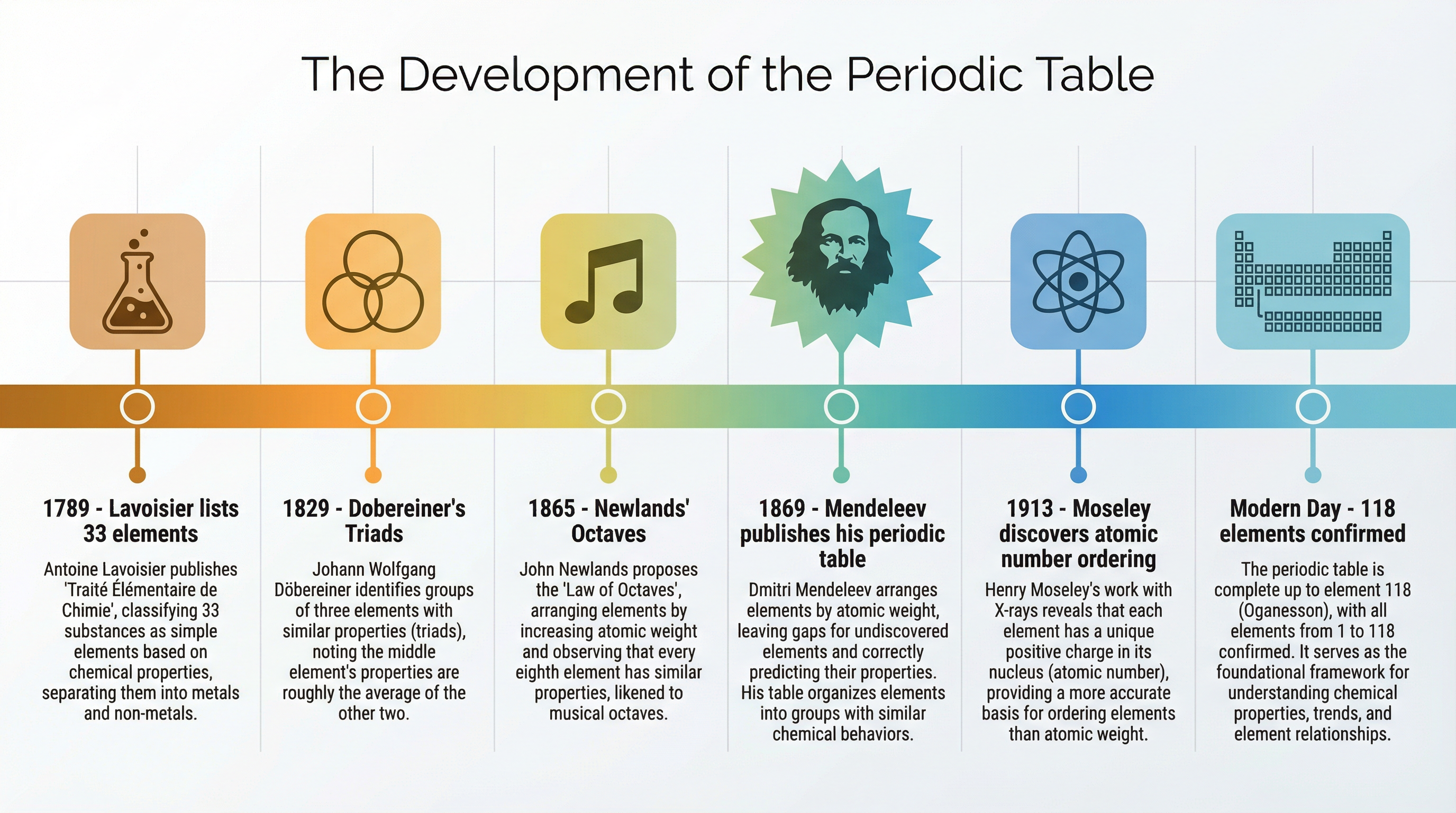
### Concept 1: The Historical Development of the Periodic Table
Before the 20th century, scientists did not know about protons, neutrons, or electrons. They had to rely on observable properties and atomic mass (then called atomic weight) to organise the elements. In 1869, the Russian chemist Dmitri Mendeleev achieved a major breakthrough. He arranged the known elements in order of increasing atomic mass, but crucially, he grouped elements with similar chemical properties together.
Mendeleev's genius lay in two key decisions:
1. **Leaving gaps:** When the pattern of properties didn't fit, he left gaps for undiscovered elements, rather than forcing known elements into the wrong groups.
2. **Making predictions:** He predicted the properties of these missing elements (such as 'eka-silicon'). When elements like germanium were later discovered and matched his predictions perfectly, his table was accepted by the scientific community.
However, Mendeleev's table had anomalies. Sometimes he had to swap the order of elements so they fit the pattern of properties, even if it meant breaking the atomic mass rule (e.g., placing tellurium before iodine).
### Concept 2: The Modern Periodic Table and Atomic Number
The discovery of isotopes explained why Mendeleev's atomic mass ordering sometimes failed. Isotopes are atoms of the same element with different numbers of neutrons, meaning they have different masses but identical chemical properties. Because atomic mass is an average of all isotopes, it can skew the order.
In 1913, Henry Moseley discovered that each element has a unique positive charge in its nucleus — the **atomic number** (number of protons). The modern periodic table is arranged in order of **increasing atomic number**, not atomic mass. This completely resolved Mendeleev's anomalies.
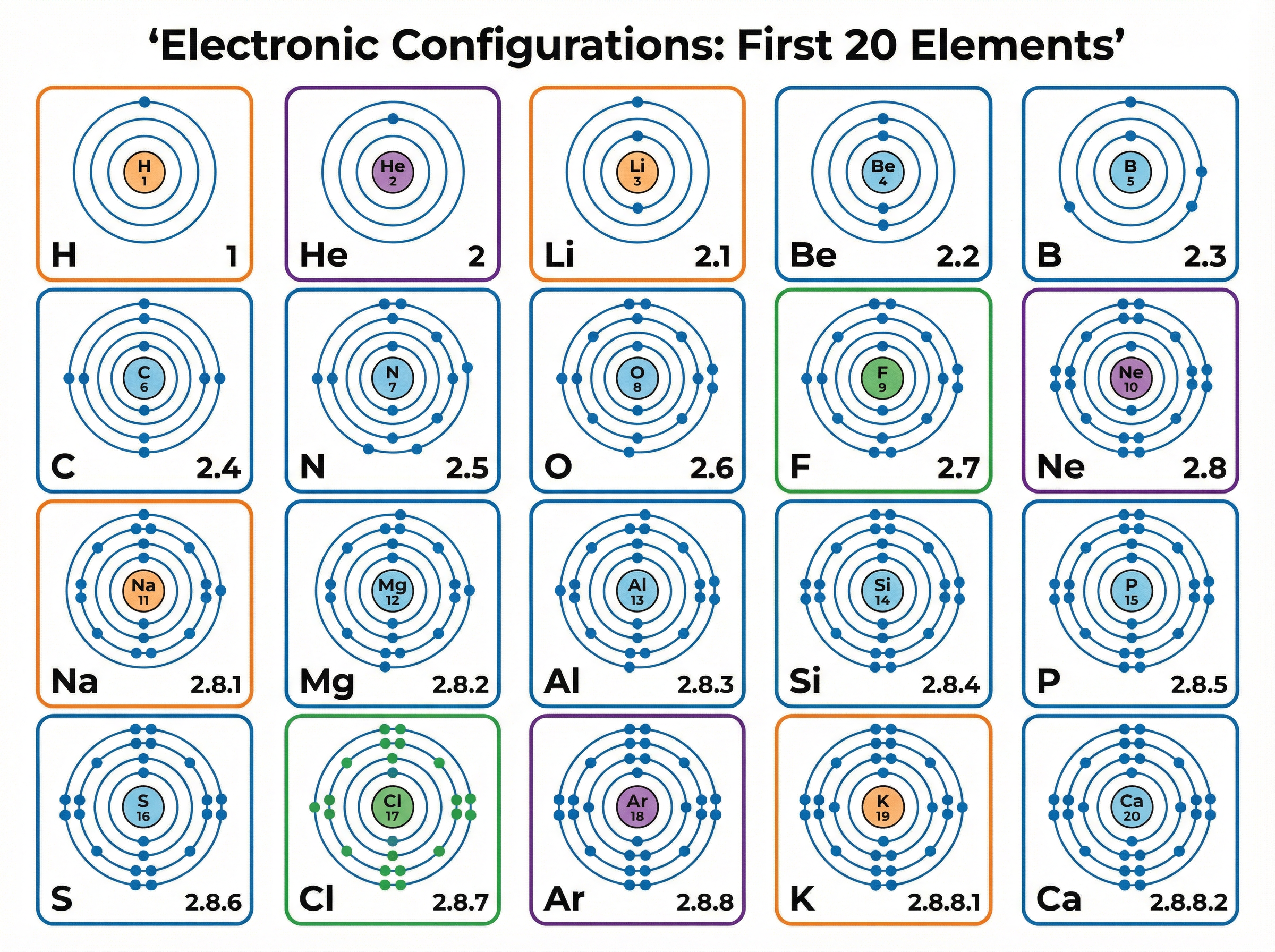
### Concept 3: Groups, Periods, and Electronic Configuration
The modern table is a grid of rows and columns:
- **Periods (horizontal rows):** The period number tells you the **number of electron shells** an atom has.
- **Groups (vertical columns):** The group number tells you the **number of electrons in the outer shell**.
Electrons occupy shells around the nucleus. The rules for the first 20 elements are:
- 1st shell holds a maximum of 2 electrons.
- 2nd shell holds a maximum of 8 electrons.
- 3rd shell holds a maximum of 8 electrons.
**Example:** Sodium (Na) has an atomic number of 11. This means it has 11 protons and 11 electrons.
Filling the shells: 2 in the first, 8 in the second, leaving 1 for the third.
Electronic configuration: **2.8.1**
Because it has 3 shells, it is in **Period 3**. Because it has 1 outer electron, it is in **Group 1**.
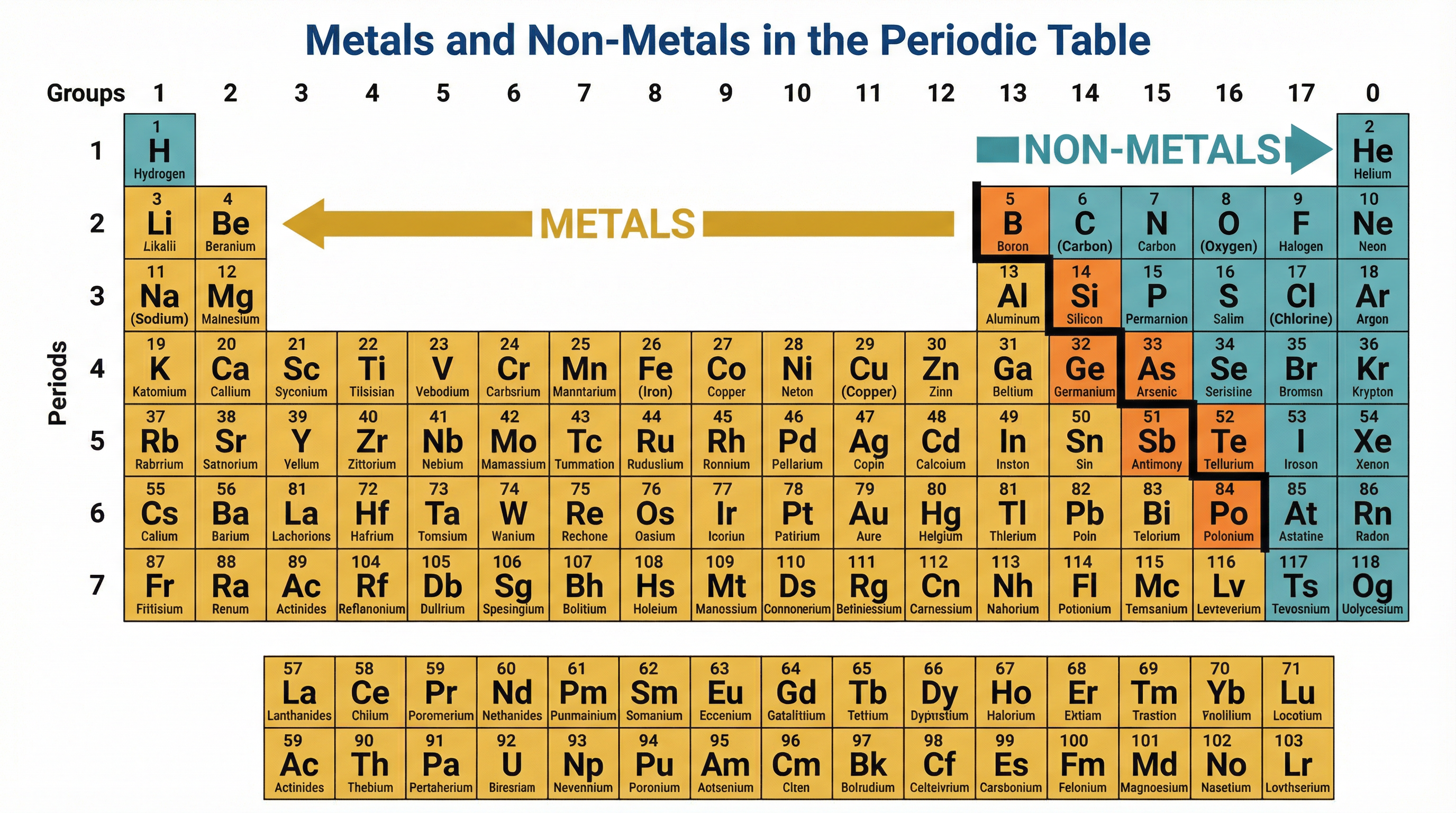
### Concept 4: Metals and Non-Metals
The periodic table is divided into metals and non-metals by a 'staircase' line starting near boron.
- **Metals** are found on the left and in the centre. They react to **lose** electrons and form positive ions.
- **Non-metals** are found on the right. They react to **gain** or share electrons to form negative ions or covalent bonds.
## Mathematical/Scientific Relationships
There are no complex mathematical formulas in this specific topic, but there is a crucial numerical relationship you must apply:
**Atomic Number = Number of Protons = Number of Electrons (in a neutral atom)**
You must use the atomic number (the smaller number on the periodic table tile) to determine the total number of electrons before writing the electronic configuration.
## Practical Applications
Understanding the periodic table allows chemists to predict how entirely new or highly reactive elements will behave. For example, knowing that francium is at the bottom of Group 1 allows us to predict it will be explosively reactive with water, even though it is too rare and radioactive to test safely in a school lab.