Key concepts in chemistry — Edexcel GCSE Study Guide
Exam Board: Edexcel | Level: GCSE
Master the fundamental building blocks of all matter. This essential topic covers the structure of atoms, the role of subatomic particles, and how to calculate relative atomic mass from isotopes — key skills that underpin your entire GCSE Chemistry course.

## Overview
Welcome to Key Concepts in Chemistry. This topic explores the absolute foundation of the subject: atomic structure. Everything in the universe is made of atoms, and understanding their internal architecture is crucial for explaining how elements behave, why they react, and how they bond together.
In this section, you will learn about the three subatomic particles—protons, neutrons, and electrons—and how their arrangement defines the properties of an element. We will also delve into isotopes and the mathematical calculation of relative atomic mass, a frequent target for high-mark exam questions.
Examiners consistently test this area through calculation questions, diagram interpretation, and short-answer definitions. A strong grasp of these concepts will not only secure marks here but will also provide the necessary grounding for subsequent topics like chemical bonding, quantitative chemistry, and the periodic table.
Listen to the companion podcast below for a comprehensive audio review of these concepts, including a quick-fire recall quiz.

## Key Concepts
### Concept 1: Subatomic Particles and Atomic Structure
Atoms are incredibly small, with a radius of approximately $1 \times 10^{-10}$ metres. Despite their size, they possess a distinct internal structure consisting of a central nucleus surrounded by electron shells.
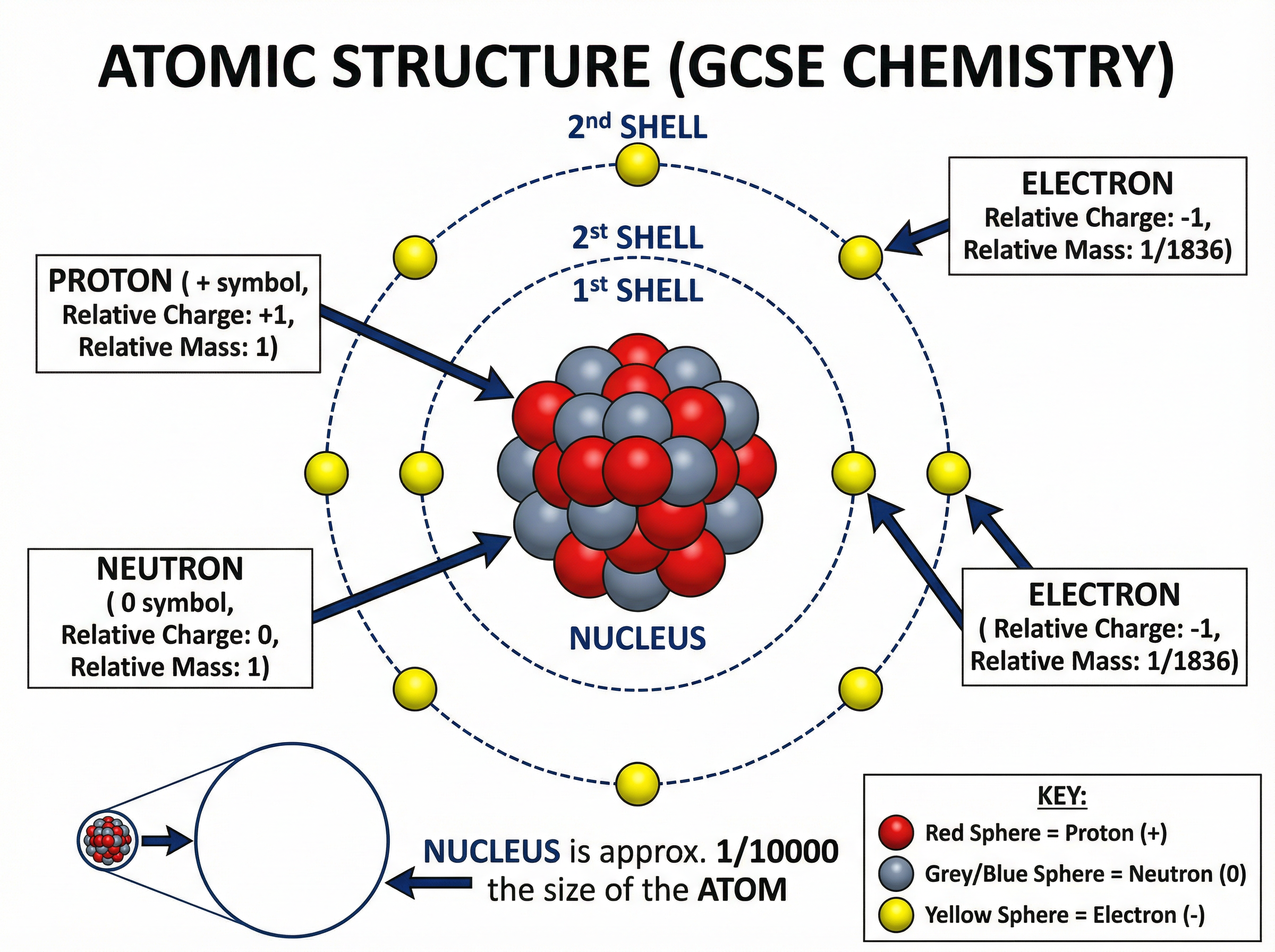
The nucleus is the dense core of the atom, containing **protons** and **neutrons**. It is positively charged and contains almost all of the atom's mass, even though its radius is only about $1/10000$ of the total atomic radius.
Orbiting the nucleus in distinct energy levels (or shells) are the **electrons**.
You must memorise the relative charges and masses of these three subatomic particles:
| Particle | Relative Charge | Relative Mass |
| :--- | :--- | :--- |
| Proton | +1 | 1 |
| Neutron | 0 | 1 |
| Electron | -1 | Very small (approx. 1/1836) |
**Examiner Tip**: Never describe the mass of an electron as "zero" in an exam. Use "very small" or "negligible".
### Concept 2: Atomic Number and Mass Number
Every element on the periodic table is defined by its **atomic number** (the smaller number). This is the number of protons in the nucleus. Because atoms are electrically neutral overall, the number of electrons is always equal to the number of protons.
The **mass number** (the larger number) is the total number of protons and neutrons combined.
To find the number of neutrons in an atom, you must subtract the atomic number from the mass number.
**Example**: For Sodium ($^{23}_{11}\text{Na}$):
- Atomic number = 11 (so it has 11 protons and 11 electrons)
- Mass number = 23
- Number of neutrons = $23 - 11 = 12$
### Concept 3: Isotopes
**Isotopes** are atoms of the same element that have the same number of protons but a different number of neutrons.
Because they have the same number of protons (and therefore electrons), isotopes have identical chemical properties. However, because they have different numbers of neutrons, they have different mass numbers and slightly different physical properties (such as density).
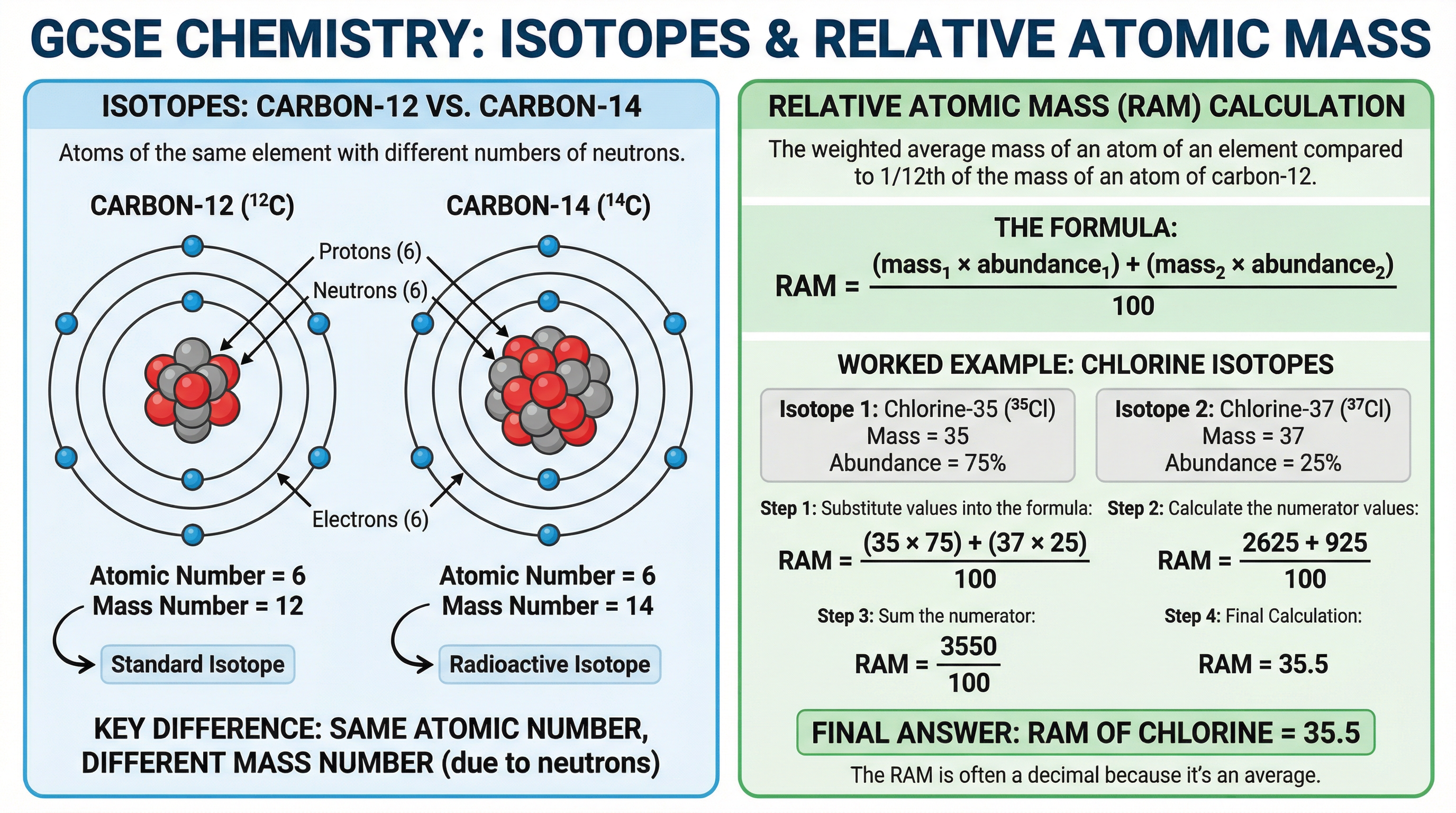
### Concept 4: Relative Atomic Mass ($A_r$)
Because elements can exist as a mixture of different isotopes, the mass number shown on the periodic table is often not a whole number (e.g., Chlorine is 35.5). This value is the **relative atomic mass** ($A_r$).
The relative atomic mass is a weighted average that takes into account the masses of all the isotopes of an element and their relative abundances (how common they are).
## Mathematical/Scientific Relationships
### Calculating Number of Neutrons
$$\text{Number of Neutrons} = \text{Mass Number} - \text{Atomic Number}$$
**When to use**: Use this formula whenever you are asked to determine the subatomic composition of an atom from its nuclear symbol.
### Calculating Relative Atomic Mass ($A_r$)
$$A_r = \frac{(\text{Mass}_1 \times \text{Abundance}_1) + (\text{Mass}_2 \times \text{Abundance}_2)}{100}$$
**When to use**: Use this formula when given the mass numbers and percentage abundances of two or more isotopes. You must memorise this formula as it is not provided in the exam.
## Practical Applications
Understanding isotopes has profound real-world applications. For example, Carbon-14 is a radioactive isotope of carbon used in **radiocarbon dating** to determine the age of ancient biological artefacts. Similarly, specific isotopes of elements like Iodine and Technetium are used in medicine as radioactive tracers to image internal organs and diagnose diseases.