Key concepts in chemistry — Edexcel GCSE Study Guide
Exam Board: Edexcel | Level: GCSE
Master the foundation of quantitative chemistry with this comprehensive guide to moles, formulas, and the conservation of mass. These core concepts unlock the ability to calculate reacting masses and are essential for securing top marks in your GCSE exam.
## Overview
Welcome to Key Concepts in Chemistry. This topic forms the mathematical backbone of your GCSE Chemistry course. It is fundamentally about how chemists count particles, measure masses, and determine the formulas of compounds. Understanding these concepts is absolutely crucial, not just for this specific topic, but because quantitative chemistry is woven into almost every other module you will study, from electrolysis to rates of reaction.
Examiners consistently use this topic to test your mathematical skills, logical reasoning, and ability to apply formulas. You will encounter questions ranging from simple 1-mark relative formula mass calculations to complex 6-mark multi-step problems involving limiting reactants and reacting masses. By mastering the mole concept and the law of conservation of mass, you will be well-equipped to tackle these challenging calculations and secure those higher-grade marks.

## Key Concepts
### Concept 1: Relative Formula Mass ($M_r$)
The relative formula mass ($M_r$) of a compound is simply the sum of the relative atomic masses ($A_r$) of all the atoms present in its chemical formula. It is a fundamental calculation that you must be able to perform quickly and accurately, as it is the first step in almost all quantitative chemistry problems.
To calculate $M_r$, you need to use the periodic table to find the $A_r$ of each element. Then, multiply the $A_r$ by the number of atoms of that element in the formula (indicated by the small subscript number) and add them all together.
**Example**: Calculate the $M_r$ of sulfuric acid, $\text{H}_2\text{SO}_4$.
- $A_r$ of H = 1, S = 32, O = 16
- $(2 \times 1) + 32 + (4 \times 16) = 2 + 32 + 64 = 98$
### Concept 2: The Mole and Avogadro's Constant
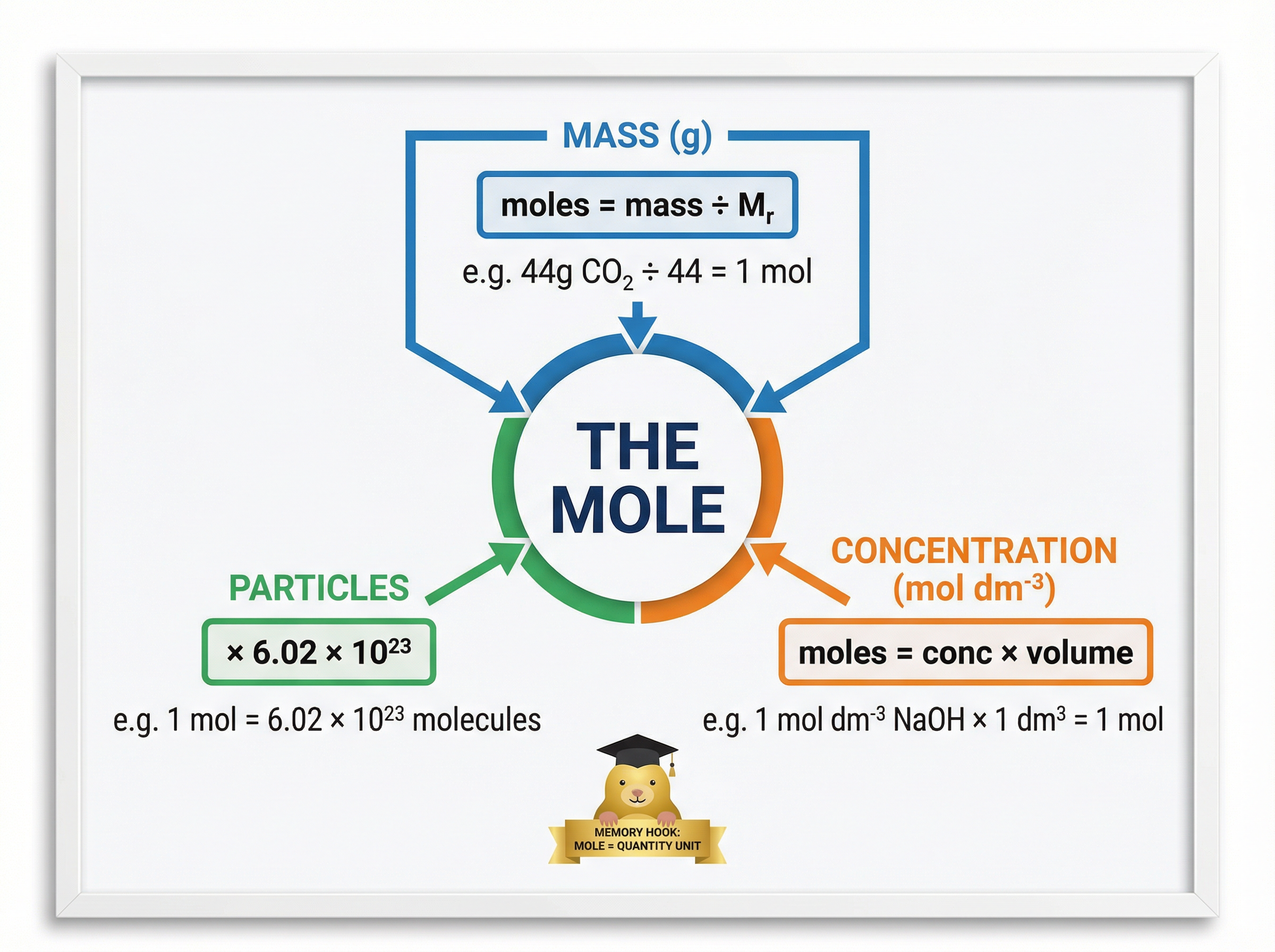
Atoms and molecules are incredibly small, making it impossible to count them individually. Instead, chemists use a unit called the **mole** (mol). One mole of any substance contains exactly $6.02 \times 10^{23}$ particles (atoms, molecules, or ions). This massive number is known as the **Avogadro constant**.
The mass of one mole of a substance in grams is numerically equal to its relative formula mass ($M_r$). For example, the $M_r$ of carbon is 12, so one mole of carbon weighs exactly 12g. This relationship allows chemists to convert between the mass of a substance and the number of particles it contains.
**Example**: How many moles are in 88g of carbon dioxide ($\text{CO}_2$)?
- $M_r$ of $\text{CO}_2 = 12 + (2 \times 16) = 44$
- $\text{Moles} = \text{Mass} \div M_r = 88 \div 44 = 2 \text{ moles}$
### Concept 3: Conservation of Mass
The law of conservation of mass states that no atoms are lost or made during a chemical reaction. Therefore, the total mass of the products must always equal the total mass of the reactants. This principle is why we must always balance chemical equations.
In a **closed system** (where nothing can enter or leave), the mass remains exactly the same throughout the reaction. However, in an **open system** (like an unsealed flask), the mass may appear to change. If a gas is produced and escapes, the mass will decrease. If a gas from the air reacts with a solid, the mass will increase. Examiners frequently test your understanding of why mass appears to change in open systems.
**Example**: 24g of magnesium reacts with oxygen to form 40g of magnesium oxide. What mass of oxygen reacted?
- $\text{Mass of reactants} = \text{Mass of products}$
- $24\text{g} + \text{Mass of oxygen} = 40\text{g}$
- $\text{Mass of oxygen} = 40\text{g} - 24\text{g} = 16\text{g}$
### Concept 4: Empirical and Molecular Formulas

The **empirical formula** is the simplest whole-number ratio of atoms of each element in a compound. The **molecular formula** is the actual number of atoms of each element in a single molecule of the compound.
To find the empirical formula from percentage composition or reacting masses, you must calculate the number of moles of each element, then find the simplest ratio by dividing by the smallest mole value.
**Example**: A compound contains 75% carbon and 25% hydrogen by mass. Find its empirical formula.
- Moles of C = $75 \div 12 = 6.25$
- Moles of H = $25 \div 1 = 25$
- Ratio = $6.25 : 25$
- Divide by smallest (6.25): $1 : 4$
- Empirical formula = $\text{CH}_4$
### Concept 5: Limiting Reactants
In many chemical reactions, one reactant is completely used up while the other is left over (in excess). The reactant that is completely consumed is called the **limiting reactant** because it limits the amount of product that can be formed.
To determine the limiting reactant, you must calculate the number of moles of each reactant and compare them using the balanced chemical equation. The reactant that produces the smaller amount of product is the limiting reactant.
## Mathematical/Scientific Relationships
- **Moles from Mass**: $\text{Moles} = \frac{\text{Mass (g)}}{M_r}$
*(Must memorise)*
- **Percentage by Mass**: $\% \text{ mass} = \frac{\text{Total } A_r \text{ of element}}{M_r \text{ of compound}} \times 100$
*(Must memorise)*
- **Concentration (g/dm³)**: $\text{Concentration} = \frac{\text{Mass (g)}}{\text{Volume (dm}^3\text{)}}$
*(Must memorise)*
- **Number of Particles**: $\text{Number of particles} = \text{Moles} \times (6.02 \times 10^{23})$
*(Given on formula sheet, but must know how to apply)*
## Practical Applications
These calculations are not just theoretical; they are essential in industrial chemistry. For example, pharmaceutical companies must use reacting masses to calculate exactly how much of each reactant is needed to produce a specific amount of a drug, minimizing waste and maximising profit. Similarly, calculating the concentration of solutions is crucial in medical settings, such as preparing intravenous drips for patients.