Rates of reaction and energy changes — Edexcel GCSE Study Guide
Exam Board: Edexcel | Level: GCSE
Master the speed of chemical reactions and the energy they release or absorb. This high-yield topic connects collision theory to real-world applications and guarantees marks for graphing and calculations.
## Overview
Rates of reaction and energy changes is a fundamental topic in GCSE Chemistry that bridges theoretical concepts with observable phenomena. It explores not just *what* happens in a chemical reaction, but *how fast* it happens and *why* energy is released or absorbed. This topic is heavily tested because it allows examiners to assess multiple skills simultaneously: explaining abstract concepts using collision theory, interpreting and drawing graphical data (reaction profiles), and performing mathematical calculations (bond energies).
Understanding these concepts is crucial for industrial chemistry, where controlling the rate and energy of reactions can make processes safer and more economically viable. You will often find synoptic links between this topic and others, such as reversible reactions, equilibrium, and the Haber process. Exam questions typically range from simple recall of the factors affecting rate to complex, multi-step calculations and 6-mark extended response questions requiring detailed explanations using particle models.

## Key Concepts
### Concept 1: Collision Theory
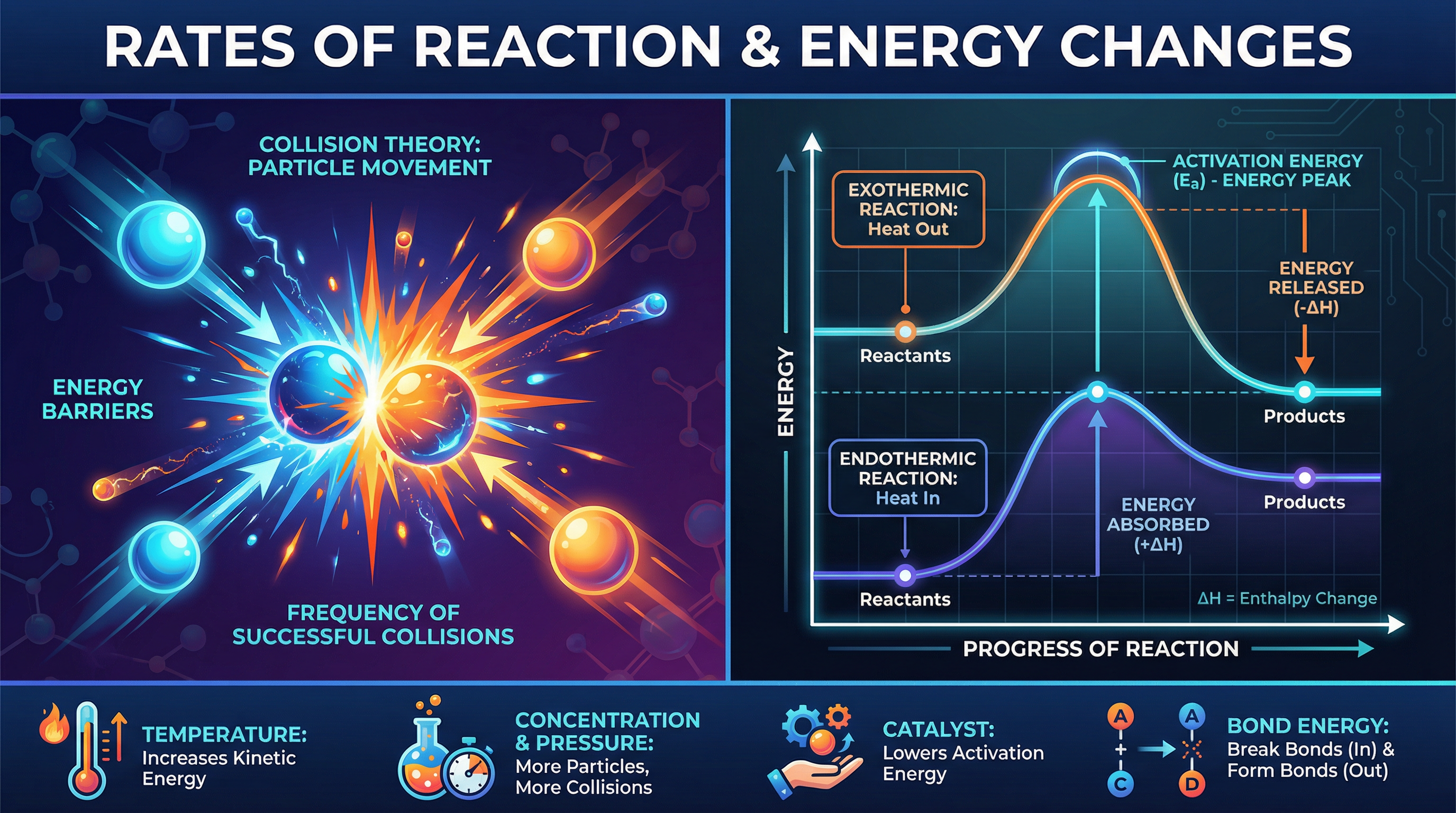
For a chemical reaction to occur, reactant particles must collide with each other. However, a collision only results in a reaction if two conditions are met: the particles must collide with the correct orientation, and they must collide with sufficient energy. This minimum amount of energy required to break the existing bonds and start the reaction is known as the **activation energy ($E_a$)**.
Think of activation energy as a toll booth on a road. If a car (particle) doesn't have enough money (energy) to pay the toll, it can't pass through and continue its journey (react). Therefore, the rate of a chemical reaction depends entirely on the **frequency of successful collisions**.
### Concept 2: Factors Affecting Rate of Reaction
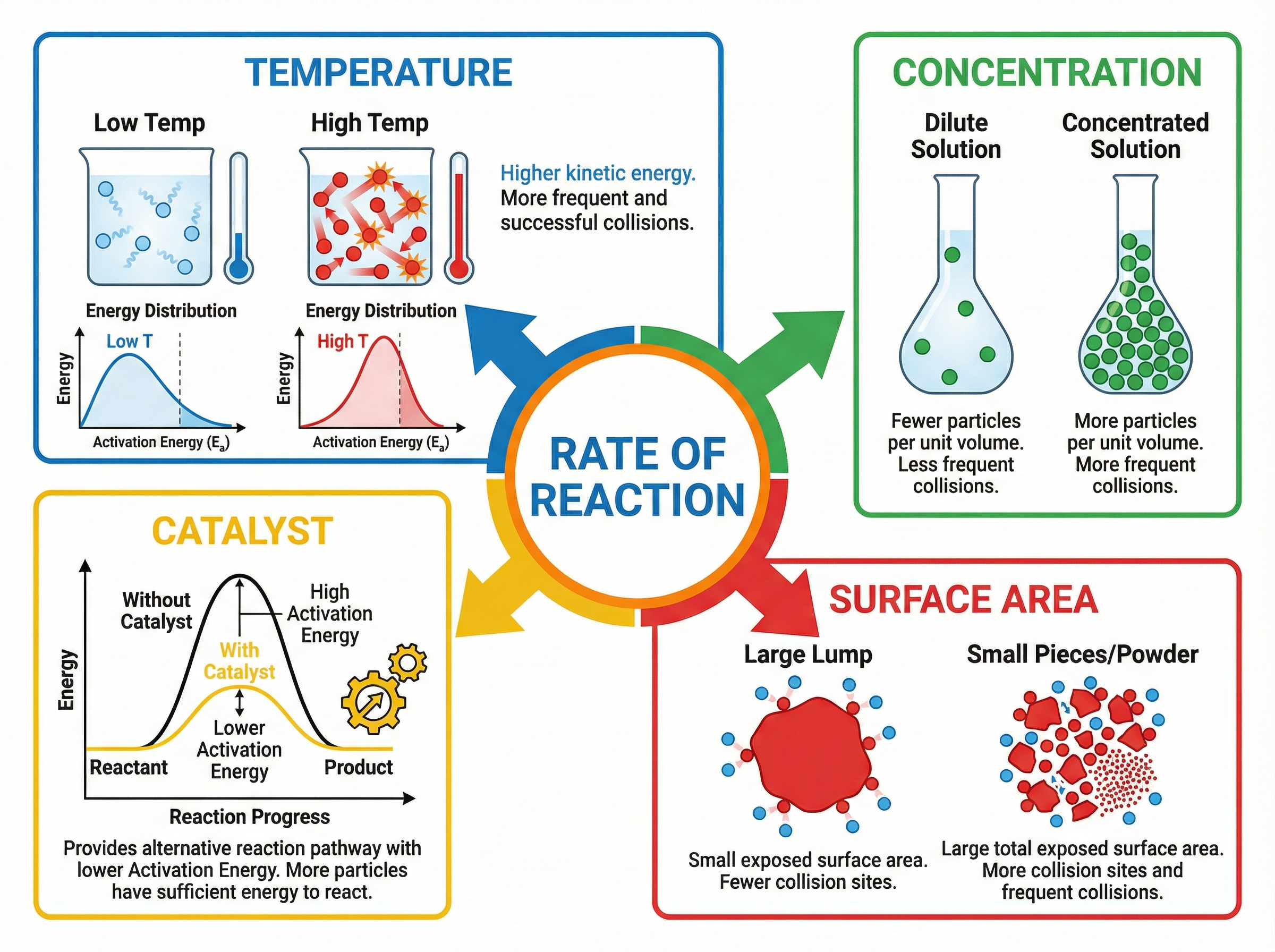
There are four primary factors that can increase the rate of a reaction:
1. **Temperature:** Increasing the temperature gives particles more kinetic energy. They move faster, resulting in more frequent collisions. Crucially, because they have more energy, a higher proportion of collisions will have energy equal to or greater than the activation energy. This leads to a significant increase in the frequency of *successful* collisions.
2. **Concentration (Solutions) / Pressure (Gases):** Increasing concentration or pressure means there are more reactant particles in the same given volume. This crowding forces the particles closer together, leading to more frequent collisions.
3. **Surface Area (Solids):** If a solid reactant is broken into smaller pieces or ground into a powder, its surface area to volume ratio increases. This exposes more particles to the other reactant, providing more sites for collisions to occur, thus increasing the collision frequency.
4. **Catalysts:** A catalyst is a substance that speeds up a chemical reaction without being used up or chemically changed itself. It achieves this by providing an alternative reaction pathway that has a lower activation energy. Consequently, a greater proportion of particles will have sufficient energy to react upon collision.
### Concept 3: Energy Changes (Exothermic and Endothermic)
Chemical reactions involve energy transfers between the reacting substances and their surroundings, usually in the form of heat.
* **Exothermic Reactions:** These reactions transfer energy *to* the surroundings, causing the temperature of the surroundings to increase. Examples include combustion, oxidation, and neutralisation.
* **Endothermic Reactions:** These reactions take in energy *from* the surroundings, causing the temperature of the surroundings to decrease. Examples include thermal decomposition and the reaction between citric acid and sodium hydrogencarbonate.
### Concept 4: Reaction Profiles
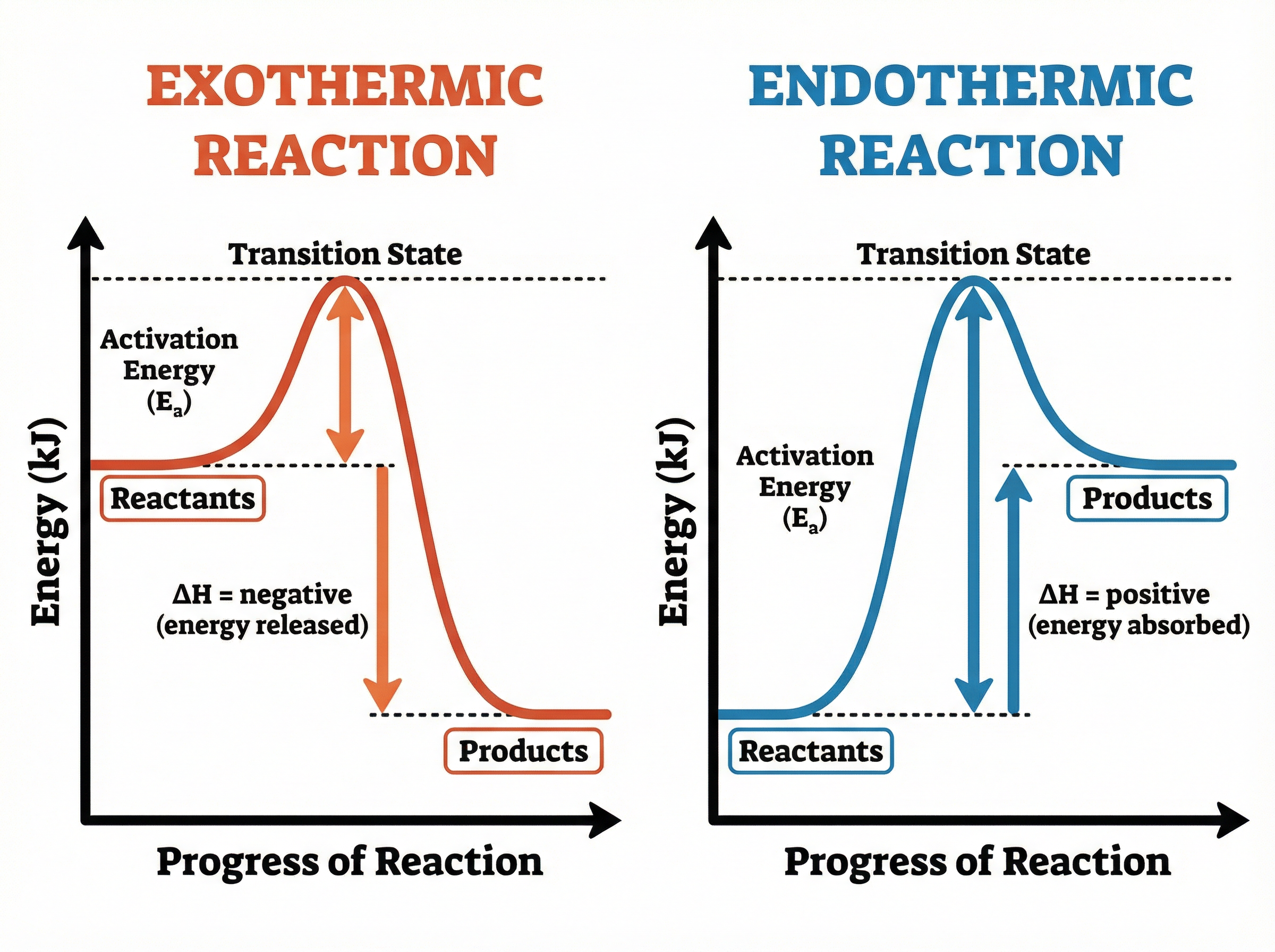
Reaction profiles (or energy level diagrams) are graphical representations of the energy changes during a reaction. They show the relative energies of reactants and products, the activation energy, and the overall energy change ($\\Delta H$).
* **Exothermic Profile:** The products are at a lower energy level than the reactants. The difference in height represents the energy released (negative $\\Delta H$).
* **Endothermic Profile:** The products are at a higher energy level than the reactants. The difference in height represents the energy absorbed (positive $\\Delta H$).
In both profiles, the activation energy is shown as an upward arrow starting from the reactant energy level to the peak of the curve.
### Concept 5: Bond Energy Calculations
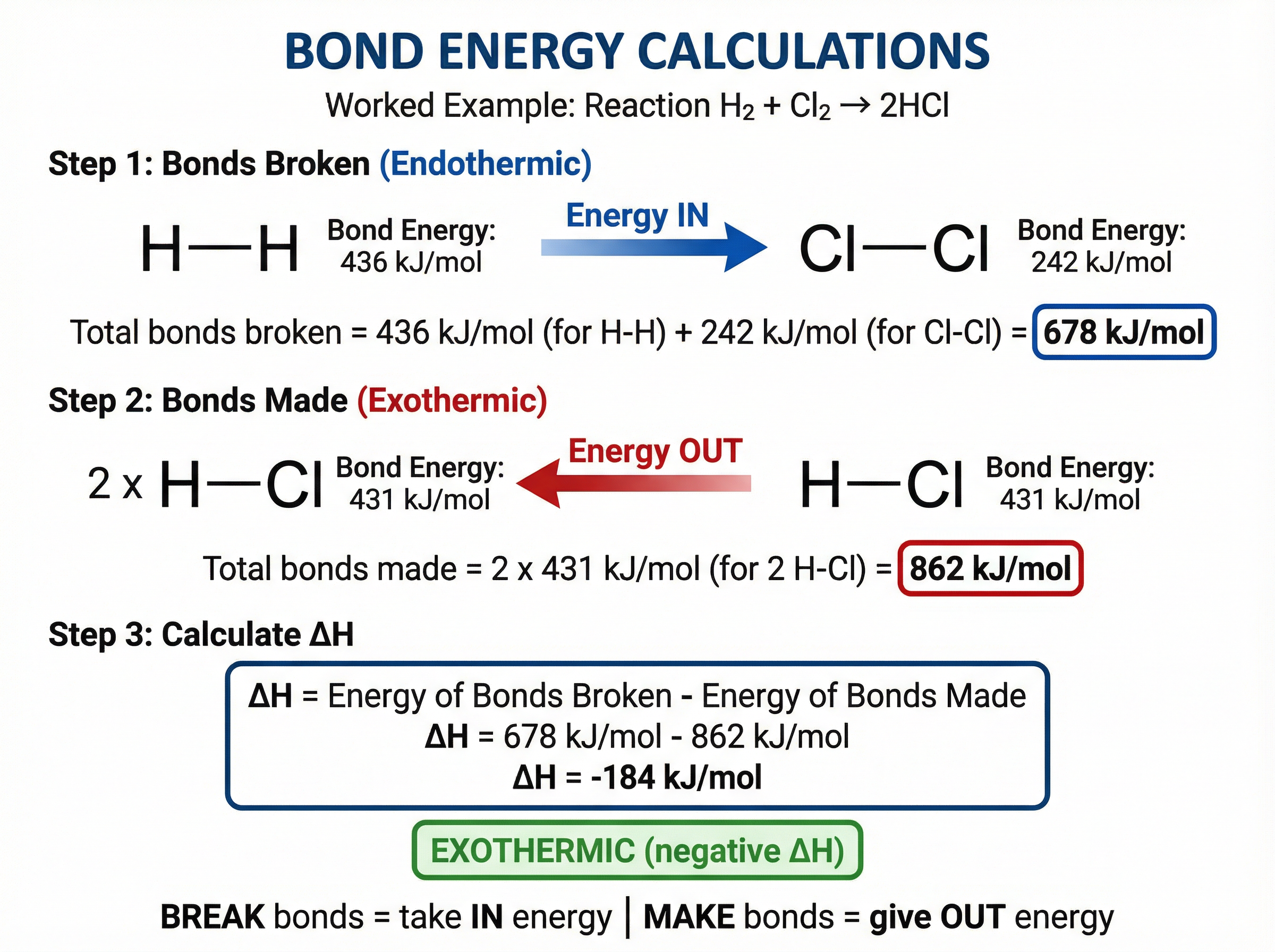
During a chemical reaction, existing bonds in the reactants must be broken, and new bonds in the products must be formed.
* **Bond breaking is endothermic:** Energy must be supplied to break bonds.
* **Bond making is exothermic:** Energy is released when new bonds form.
To calculate the overall energy change ($\\Delta H$) for a reaction, you use the formula:
$\\Delta H = \\text{Total energy needed to break bonds} - \\text{Total energy released making bonds}$
If the calculated $\\Delta H$ is negative, the reaction is exothermic. If it is positive, the reaction is endothermic.
## Mathematical/Scientific Relationships
* **Rate of Reaction Formula:**
* $\\text{Rate} = \\frac{\\text{Amount of reactant used}}{\\text{Time}}$
* $\\text{Rate} = \\frac{\\text{Amount of product formed}}{\\text{Time}}$
* *Units:* g/s, cm³/s, or mol/s.
* **Overall Energy Change ($\\Delta H$):**
* $\\Delta H = \\sum(\\text{Bonds Broken}) - \\sum(\\text{Bonds Made})$
* *Units:* kJ/mol.
## Practical Applications
**Required Practical: Investigating the effect of concentration on the rate of reaction.**
There are two common methods examiners test:
1. **Measuring the volume of gas produced:** Reacting magnesium ribbon with hydrochloric acid and collecting the hydrogen gas in a gas syringe or inverted measuring cylinder. You measure the volume of gas produced at regular time intervals.
2. **Observing a colour change (The 'Disappearing Cross' experiment):** Reacting sodium thiosulfate with hydrochloric acid produces a cloudy precipitate of solid sulfur. You place the flask over a piece of paper with a black cross drawn on it and measure the time it takes for the cross to disappear from view when looking down through the solution.
*Common Examiner Focus:* They often ask about control variables (e.g., keeping temperature and total volume constant), identifying anomalous results on a graph, and explaining why the rate decreases over time as reactants are used up.