Rates of reaction and energy changes — Edexcel GCSE Study Guide
Exam Board: Edexcel | Level: GCSE
Master the energy changes in chemical reactions, a high-yield topic that appears in every chemistry exam. Learn to confidently distinguish between exothermic and endothermic processes, draw perfect reaction profiles, and calculate bond energies to secure maximum marks.
## Overview
Every chemical reaction involves an energy change. When chemicals react, old bonds are broken and new bonds are formed. This fundamental topic explores whether a reaction releases energy to its surroundings (getting hotter) or absorbs energy from its surroundings (getting colder). Understanding energy changes is crucial in Chemistry because it explains why some reactions need continuous heating to keep going, while others can cause explosions. It connects deeply to your understanding of atomic structure and bonding, and it is frequently tested in exams through both descriptive questions and multi-mark calculations.
Examiners love to test this topic through a combination of skills: asking you to draw or interpret reaction profile diagrams, explain the difference between bond breaking and bond making, and calculate overall energy changes using bond energy values. Let's break down exactly how to secure those marks.
## Key Concepts
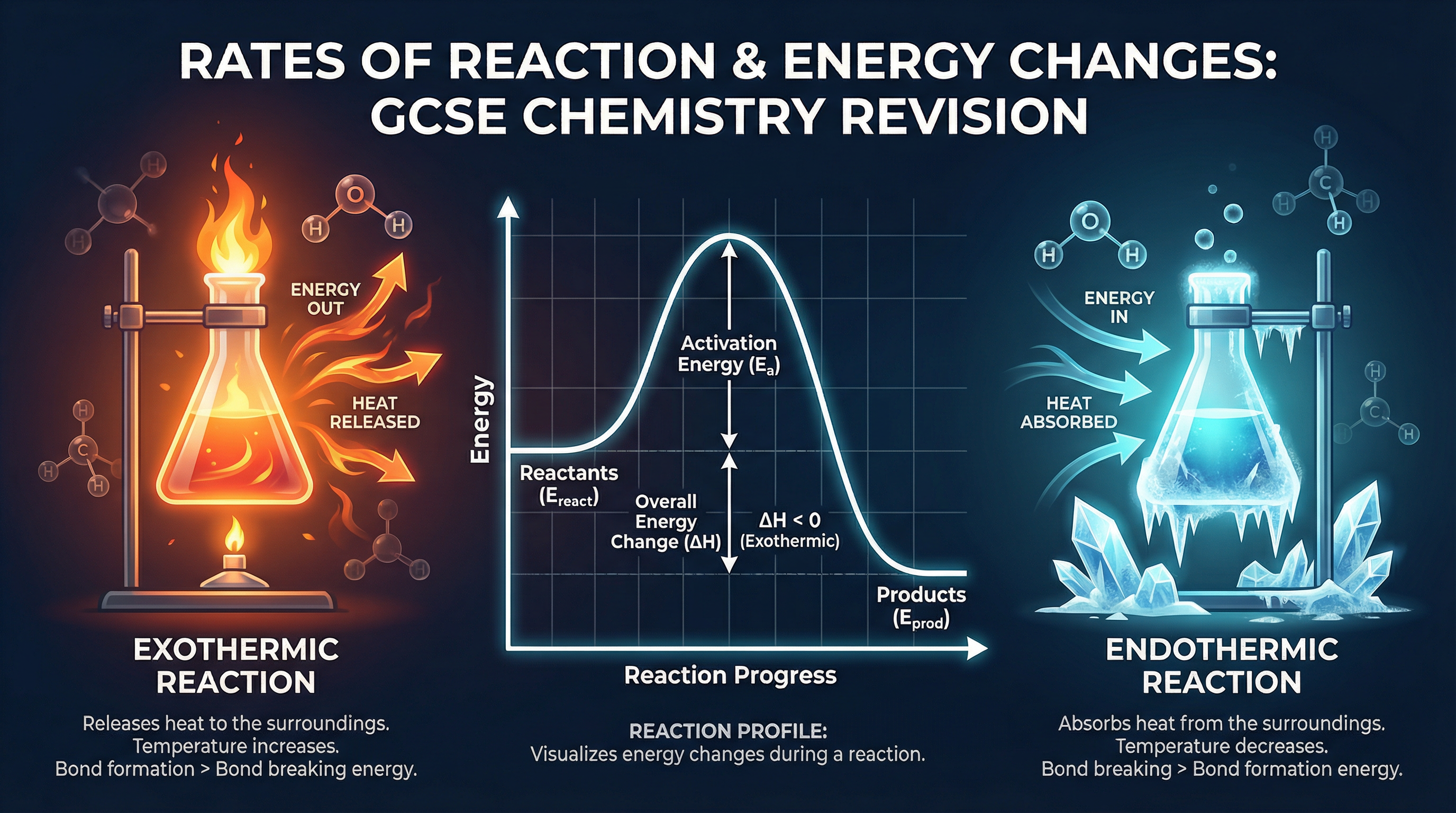
### Concept 1: Exothermic and Endothermic Reactions
An **exothermic reaction** is one that transfers energy to the surroundings, causing the temperature of the surroundings to increase. The energy exits the reaction. Common examples include combustion (burning fuels), neutralisation reactions, and many oxidation reactions. Hand warmers use exothermic oxidation of iron to release heat.
An **endothermic reaction** is one that takes in energy from the surroundings, causing the temperature of the surroundings to decrease. The energy enters the reaction. Common examples include thermal decomposition (like heating calcium carbonate) and the reaction of citric acid and sodium hydrogencarbonate. Sports injury cold packs use endothermic reactions to instantly cool down.
**Crucial Examiner Point**: Always remember that we measure the temperature of the *surroundings*, not the reaction itself. If the thermometer reading goes up, energy has been released *into* the surroundings (exothermic).
### Concept 2: Reaction Profiles (Energy Level Diagrams)
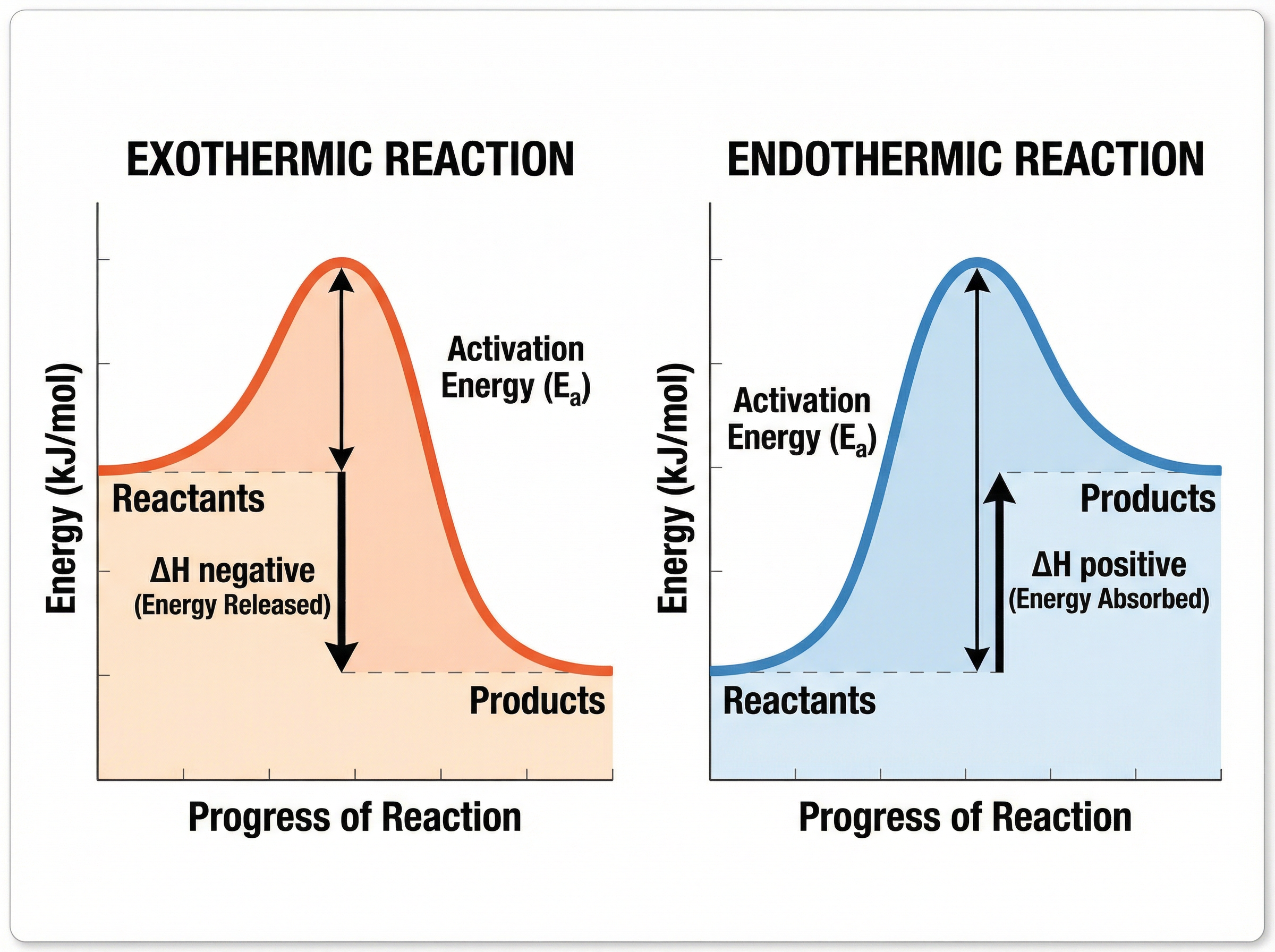
Reaction profiles show the relative energies of reactants and products, as well as the activation energy of the reaction.
- **Activation Energy ($E_a$)**: The minimum amount of energy that particles must have to react when they collide. On a diagram, it is the energy difference from the reactants to the peak of the curve.
- **Overall Energy Change ($\Delta H$)**: The difference in energy between the reactants and the products.
In an **exothermic** profile, the products are at a lower energy level than the reactants. The difference in height represents the energy released to the surroundings.
In an **endothermic** profile, the products are at a higher energy level than the reactants. The difference in height represents the energy absorbed from the surroundings.
### Concept 3: Bond Breaking and Bond Making
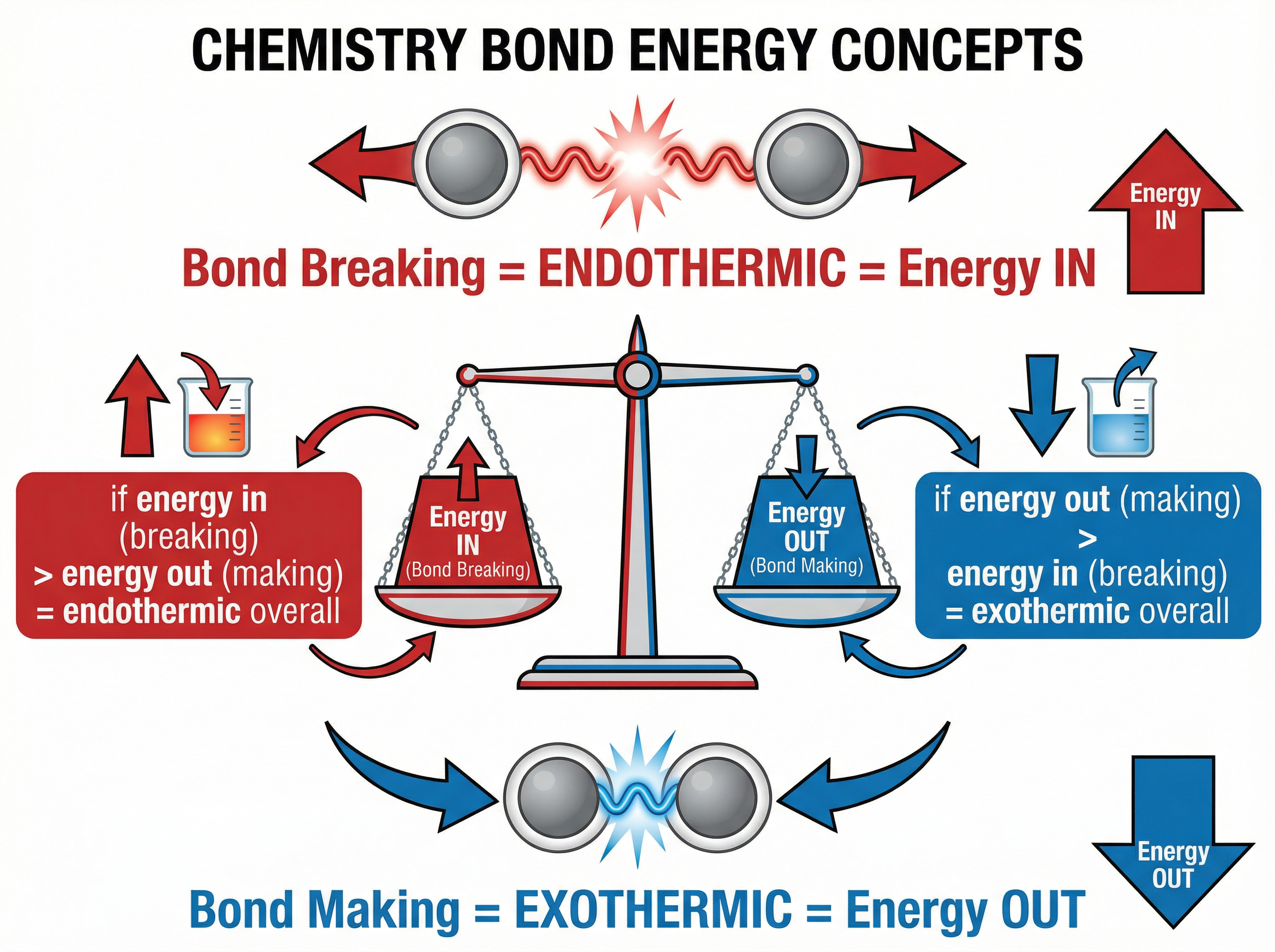
During a chemical reaction, bonds in the reactants must be broken, and new bonds must be formed to make the products.
- **Bond Breaking is Endothermic**: Energy must be supplied to break existing bonds. Think of it like snapping a stick; you have to put energy in to break it.
- **Bond Making is Exothermic**: Energy is released when new bonds are formed.
The overall energy change depends on the balance between these two processes:
- If the energy released forming new bonds is **greater** than the energy needed to break existing bonds, the reaction is **exothermic**.
- If the energy needed to break existing bonds is **greater** than the energy released forming new bonds, the reaction is **endothermic**.
## Mathematical/Scientific Relationships
### Calculating Energy Changes using Bond Energies
Every chemical bond has a specific 'bond energy' measured in kJ/mol. You can calculate the overall energy change for a reaction using the formula:
**Overall Energy Change = Total Energy IN (breaking bonds) - Total Energy OUT (making bonds)**
1. **Step 1**: Calculate the total energy required to break all bonds in the reactants.
2. **Step 2**: Calculate the total energy released when all bonds in the products are formed.
3. **Step 3**: Subtract the total energy out from the total energy in.
- A **negative** answer means the reaction is **exothermic** (energy is released overall).
- A **positive** answer means the reaction is **endothermic** (energy is absorbed overall).
## Practical Applications
### Required Practical: Temperature Changes
You must be familiar with the practical method to investigate the variables that affect temperature changes in reacting solutions (e.g., acid plus alkali).
**Method Highlights**:
1. Measure a set volume of acid into a polystyrene cup (which acts as an insulator to reduce heat loss).
2. Place the cup in a beaker for stability.
3. Measure the initial temperature of the acid.
4. Add a set volume/mass of the second reactant (e.g., alkali or metal powder).
5. Put a lid on the cup to reduce heat loss to the air, and stir gently with the thermometer.
6. Record the highest (or lowest) temperature reached.
7. Calculate the temperature change.
**Examiner Tip**: A common question asks how to improve this experiment. The answer is always about reducing heat loss to the surroundings: use a polystyrene cup with a lid, or add extra insulation like cotton wool around the cup.
## Listen to the Podcast
For a full audio review of this topic, including a quick-fire recall quiz, listen to our podcast episode:
