Separate chemistry 1 — Edexcel GCSE Study Guide
Exam Board: Edexcel | Level: GCSE
Master the principles of chemical cells and hydrogen-oxygen fuel cells. This topic is essential for understanding modern clean energy technology and frequently appears in 4-6 mark evaluation questions.
## Overview

This topic explores the fascinating world of electrochemistry, focusing on how chemical reactions can be harnessed to generate electricity. You will investigate standard chemical cells, which power everyday devices like remote controls, and compare them with the cutting-edge technology of hydrogen-oxygen fuel cells.
Understanding these cells is crucial in modern chemistry, as the world transitions towards sustainable energy sources and electric vehicles. Examiners frequently test this topic through comparative and evaluative questions, requiring you to weigh the environmental and practical benefits of fuel cells against their limitations. You will also need to master the specific half-equations occurring at the electrodes.
## Key Concepts
### Concept 1: Standard Chemical Cells
A standard chemical cell converts chemical energy directly into electrical energy. It consists of two different metal electrodes dipped into an electrolyte solution. The fundamental principle is that the two metals must have different reactivities.
The more reactive metal acts as the negative electrode (the anode in a galvanic cell). Here, the metal atoms lose electrons (oxidation) and form positive ions that enter the solution. These released electrons travel through the external circuit (the wire) to the less reactive metal, which acts as the positive electrode (the cathode). This flow of electrons constitutes the electric current that can power a bulb or a motor.
The crucial point examiners look for is that a chemical cell produces a voltage **only until one of the reactants is used up**. Once the more reactive metal is fully oxidised or the electrolyte is depleted, the chemical reaction stops, and the cell goes 'flat'. It has a finite lifespan.
**Example**: In a simple cell using zinc and copper electrodes in a sodium chloride electrolyte, zinc is more reactive. Zinc atoms oxidise to form Zn²⁺ ions, releasing electrons that flow to the copper electrode.
### Concept 2: The Hydrogen-Oxygen Fuel Cell
Unlike standard chemical cells, a fuel cell does not rely on solid metal electrodes that get used up. Instead, it generates electricity from a continuous external supply of fuel (hydrogen) and oxygen (from the air). As long as these gases are supplied, the fuel cell will produce a continuous voltage.
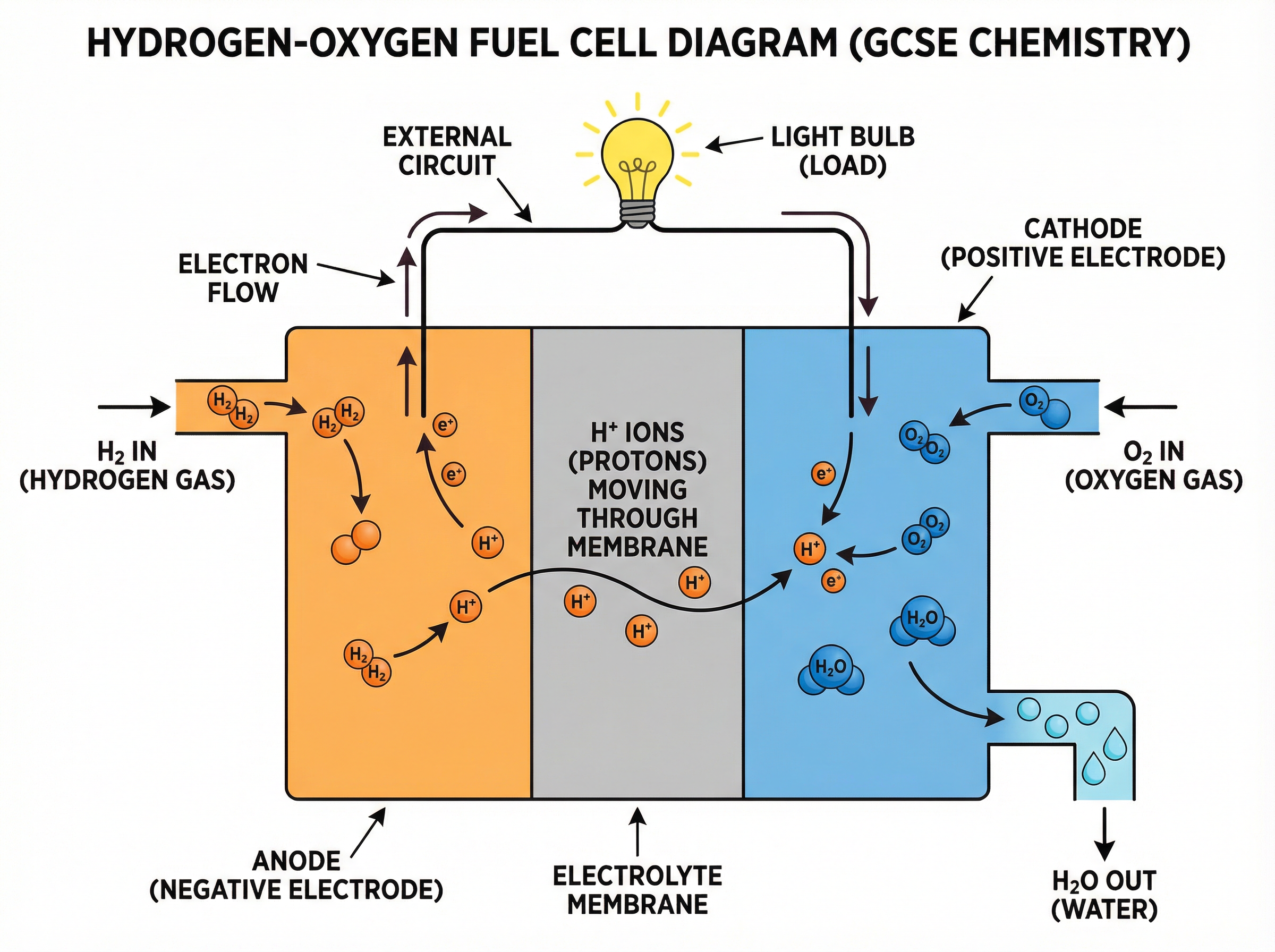
Inside the cell, hydrogen gas enters at the negative electrode (anode). Here, the hydrogen molecules are oxidised, losing electrons to form hydrogen ions (H⁺, or protons). The electrons travel through the external circuit, providing the electrical power.
Meanwhile, the hydrogen ions migrate through a special electrolyte membrane to the positive electrode (cathode). At the cathode, oxygen gas enters and is reduced, gaining the electrons that have travelled around the circuit. The oxygen then reacts with the hydrogen ions to form water.
This leads to the most important fact about hydrogen-oxygen fuel cells for your exam: **Water is the only product**. There are no carbon emissions, no toxic gases, just pure H₂O.

## Mathematical/Scientific Relationships
To secure top marks, you must be able to write the overall balanced equation for the reaction in a hydrogen-oxygen fuel cell:
**2H₂ + O₂ → 2H₂O**
This equation shows that two molecules of hydrogen gas react with one molecule of oxygen gas to produce two molecules of liquid water.
*Higher Tier Only*: You may also be asked for the half-equations at each electrode.
At the negative electrode (oxidation of hydrogen):
**H₂ → 2H⁺ + 2e⁻**
At the positive electrode (reduction of oxygen):
**O₂ + 4H⁺ + 4e⁻ → 2H₂O**
## Practical Applications
Hydrogen fuel cells are currently being developed and used to power electric vehicles, including cars, buses, and even some trains. Because their only emission is water vapour, they offer a promising solution to urban air pollution and greenhouse gas emissions associated with traditional internal combustion engines. However, the practical challenges of safely storing highly flammable hydrogen gas at high pressures, and the current lack of refuelling infrastructure, remain significant hurdles to widespread adoption.
Listen to our summary podcast for more revision tips:
