Separate chemistry 1 — Edexcel GCSE Study Guide
Exam Board: Edexcel | Level: GCSE
Master the mathematics of chemistry! This topic covers the essential calculations needed to determine concentrations, evaluate reaction efficiency, and predict gas volumes — skills that are heavily rewarded with method marks in your final exams.
## Overview
Quantitative Chemistry is the mathematics of chemical reactions. It is the bridge between the theoretical equations you balance on paper and the practical realities of working in a laboratory or an industrial chemical plant. This topic focuses on advanced quantitative techniques specifically required for the Separate Chemistry qualification (Topic 5).
Examiners heavily weight this topic because it tests your ability to apply logic, manipulate formulas, and handle units correctly. You will learn how to calculate the concentration of solutions (crucial for titrations), evaluate how efficient a reaction is using percentage yield and atom economy, and predict the volume of gases produced in reactions using the molar volume constant.
This topic links directly to practical skills, particularly the titration required practical, and builds on your foundational knowledge of moles and relative formula mass. Expect multi-step calculation questions where showing your working is just as important as the final answer.
Listen to the revision podcast below for a complete audio walkthrough of this topic:

## Key Concepts
### Concept 1: Solution Concentration and Titrations
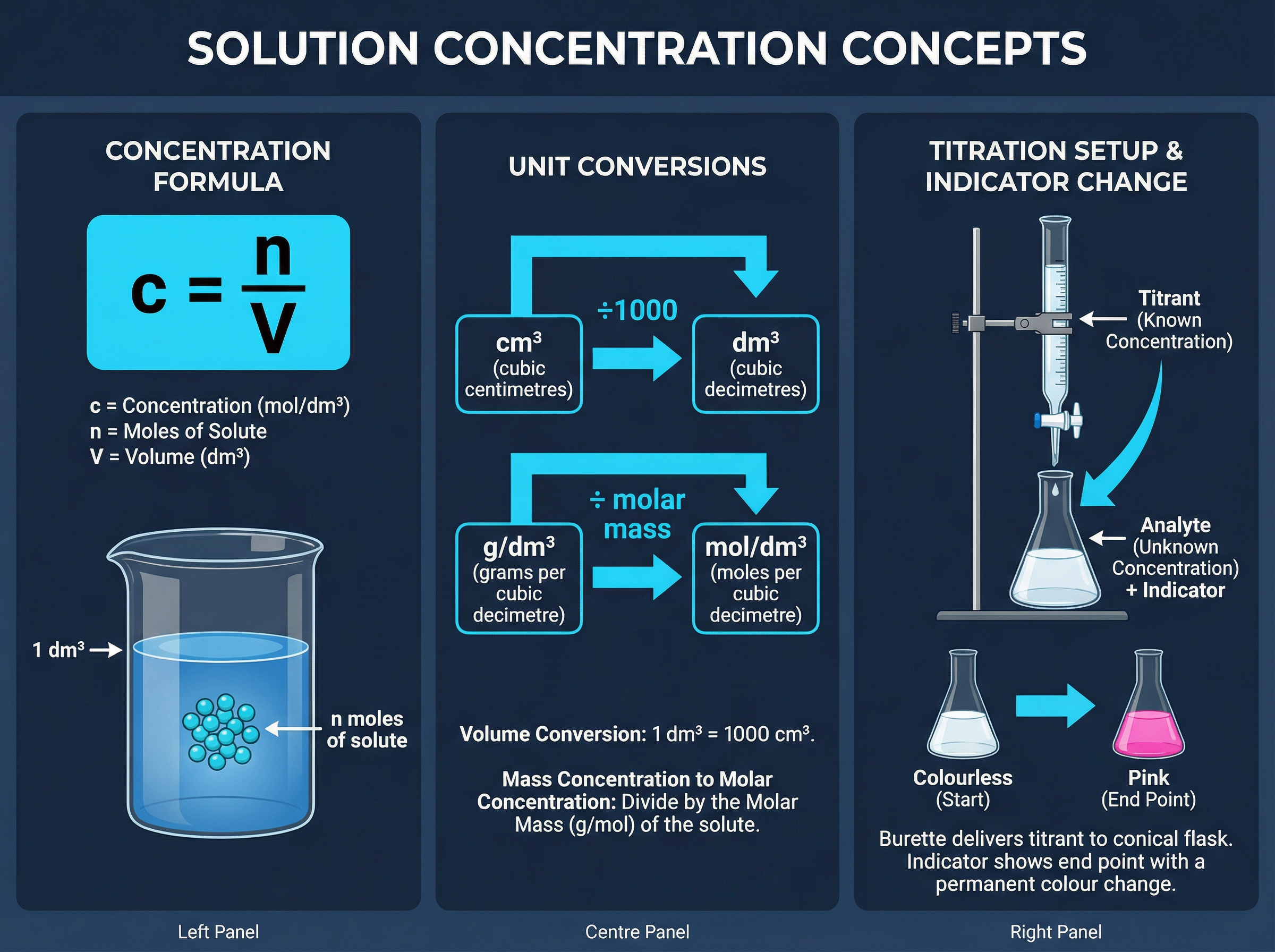
Concentration is a measure of how crowded the solute particles are in a given volume of solvent. Think of it like making squash — adding more squash (solute) to the same amount of water makes it more concentrated. In chemistry, we measure the 'amount' in moles and the 'volume' in cubic decimetres (dm³).
To calculate concentration, we use the relationship: **Concentration = Moles ÷ Volume**. The standard unit is mol/dm³.
**Crucial Exam Skill**: You must ensure your volume is in dm³. Examiners will almost always give you the volume in cm³ to test if you remember to convert it. To convert cm³ to dm³, you must divide by 1000. For example, 250 cm³ is 0.25 dm³.
Titrations are a practical application of concentration. By reacting a known volume of a solution with an unknown concentration against a solution with a known concentration (the titrant), you can calculate the unknown. This is a classic 3-step calculation: find the moles of the known, use the balanced equation ratio to find the moles of the unknown, and then calculate its concentration.
### Concept 2: Percentage Yield
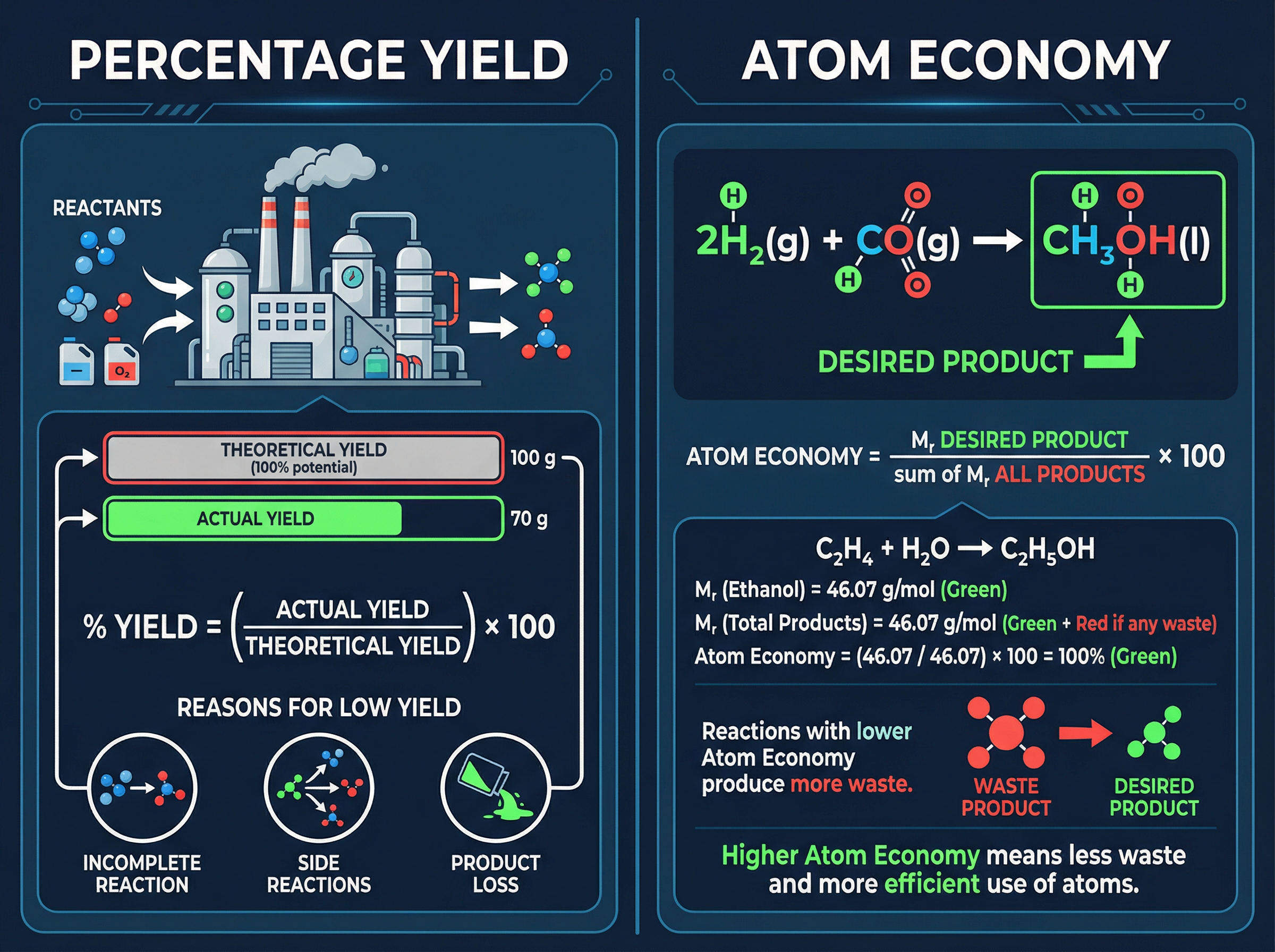
When you calculate how much product a reaction *should* make based on the moles of reactants, you are calculating the **theoretical yield**. This is the absolute maximum amount possible if everything goes perfectly. However, in the real world, you never get 100% of the theoretical yield. The amount you actually collect is the **actual yield**.
The percentage yield compares the actual yield to the theoretical yield. It tells you how successful the practical process was.
Examiners frequently ask *why* the percentage yield is less than 100%. You must be able to state three distinct reasons:
1. The reaction may be reversible and not go to completion.
2. Some of the reactants may react in unexpected ways (side reactions).
3. Some of the product may be lost when it is separated from the reaction mixture (e.g., left on filter paper or spilt during transfer).
### Concept 3: Atom Economy
While percentage yield measures practical efficiency, **atom economy** measures theoretical efficiency. It asks: of all the atoms we put into this reaction, what percentage ended up in the product we actually want, rather than in waste products?
High atom economy is vital for sustainable development and economic viability in the chemical industry. A reaction with 100% atom economy means every single atom from the reactants ended up in the desired product (typical of addition reactions). Reactions that produce multiple products (like substitution reactions) will always have an atom economy of less than 100%.
**Exam Trap**: When calculating atom economy, you must use the sum of the relative formula masses of **all products** on the bottom of the fraction, taking into account the large balancing numbers (stoichiometry) from the equation.
### Concept 4: Molar Volume of Gases
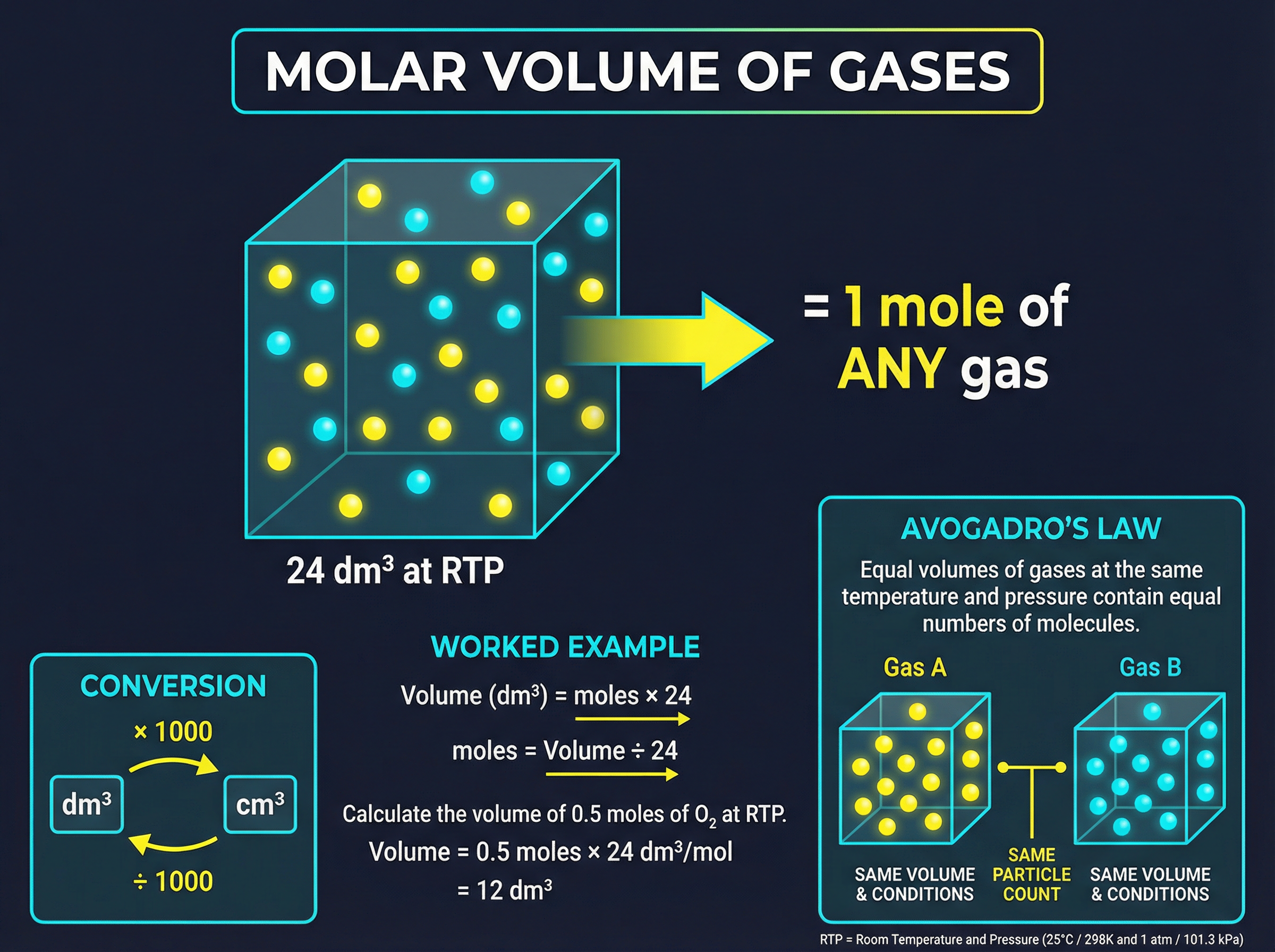
Avogadro's law states that equal volumes of different gases, at the same temperature and pressure, contain the same number of molecules. This leads to a incredibly useful constant: at room temperature and pressure (RTP, which is 20°C and 1 atmosphere), **one mole of any gas occupies exactly 24 dm³** (or 24,000 cm³).
This means you don't need to know the mass or the identity of the gas to find its volume, only the number of moles. The formula is simply: **Volume = Moles × 24** (when volume is in dm³).
This also allows you to read reacting gas volumes directly from the balanced equation. If 1 mole of methane reacts with 2 moles of oxygen, then 10 dm³ of methane will react with exactly 20 dm³ of oxygen.
## Mathematical/Scientific Relationships
Here are the core formulas you must memorise and apply:
| Formula | What it calculates | Units required |
| :--- | :--- | :--- |
| **$c = \frac{n}{V}$** | Concentration ($c$) from moles ($n$) and Volume ($V$) | $c$ in mol/dm³, $n$ in mol, $V$ in dm³ |
| **$V_{dm^3} = \frac{V_{cm^3}}{1000}$** | Converting volume from cm³ to dm³ | cm³ to dm³ |
| **$\text{Percentage Yield} = \frac{\text{Actual Yield}}{\text{Theoretical Yield}} \times 100$** | Efficiency of the practical process | Both yields must be in the same units (usually grams) |
| **$\text{Atom Economy} = \frac{M_r \text{ of desired product}}{\text{Sum of } M_r \text{ of all products}} \times 100$** | Theoretical efficiency of the reaction pathway | Use relative formula masses ($M_r$) including balancing numbers |
| **$\text{Volume} = n \times 24$** | Volume of a gas at RTP from moles ($n$) | Volume in dm³, $n$ in mol |
## Practical Applications
**Required Practical: Titration**
This topic is heavily linked to the titration required practical. You must know the apparatus (pipette for accurate fixed volume, burette for variable volume), the method (adding dropwise near the end point, swirling constantly), and how to read the burette (at eye level, bottom of the meniscus). You also need to know suitable indicators (phenolphthalein or methyl orange) and their colour changes, as universal indicator is not suitable for titrations due to its gradual colour change.