Separate chemistry 1 — Edexcel GCSE Study Guide
Exam Board: Edexcel | Level: GCSE
Master the unique properties of transition metals, the structural secrets that make alloys so strong, and the vital chemistry behind corrosion. This topic is heavily tested on your ability to link atomic structure to macroscopic properties and real-world applications.
## Overview
Welcome to Separate Chemistry 1, Topic 5: Transition Metals, Alloys, and Corrosion. This topic bridges the gap between theoretical atomic structure and the materials we rely on every day, from the steel in our bridges to the catalysts in industrial processes.
Understanding this topic is crucial for the exam because examiners frequently use it to test your ability to apply fundamental chemical principles—like metallic bonding and oxidation—to unfamiliar contexts. You will often encounter questions asking you to compare the properties of pure metals and alloys, or to evaluate the effectiveness of different rust prevention methods.
By mastering how the microscopic arrangement of atoms dictates the macroscopic strength of an alloy, and precisely what conditions are required for iron to rust, you will secure high marks in some of the most common extended-response questions.
Listen to the companion podcast to reinforce your learning:

## Key Concepts
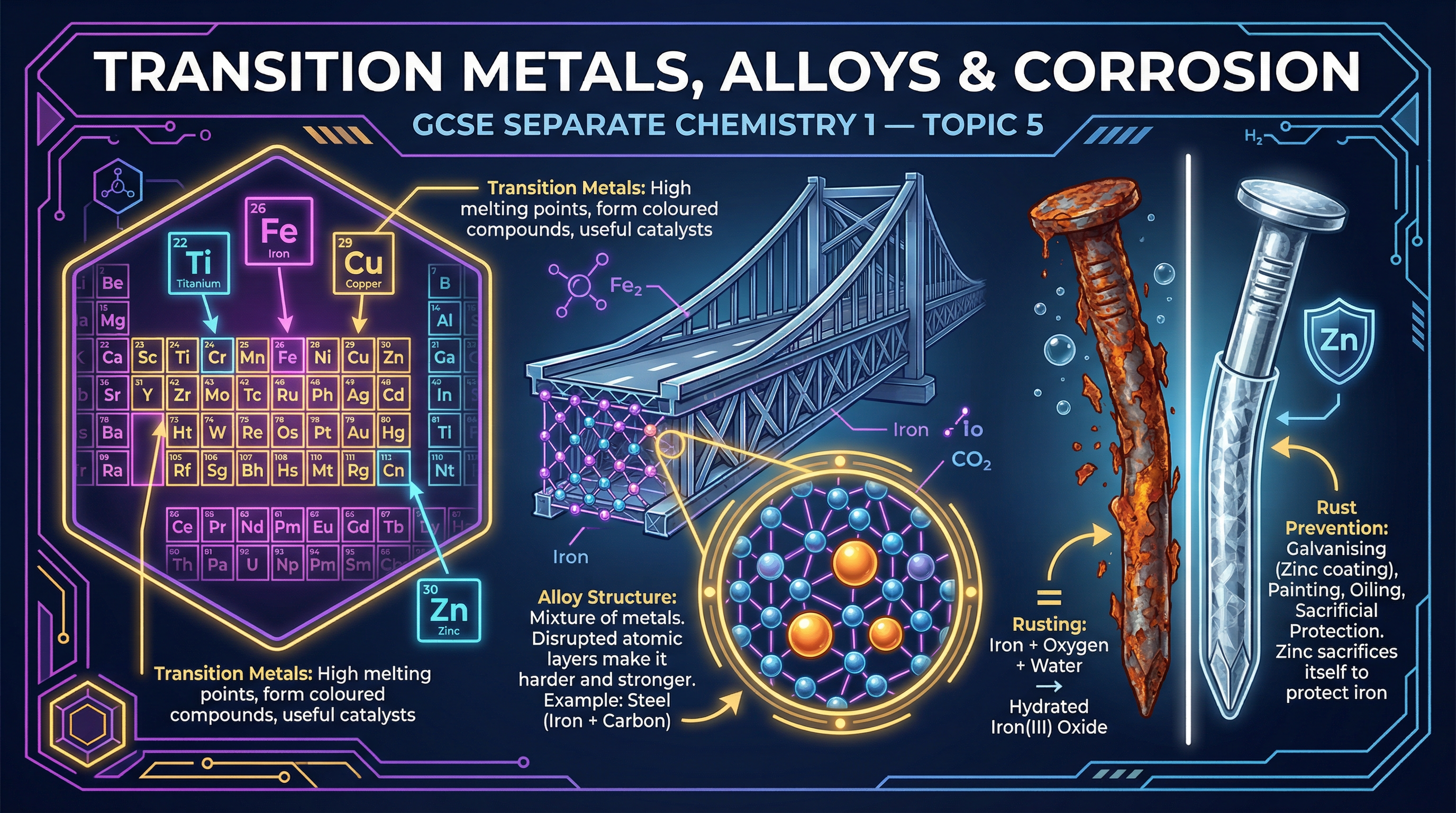
### Concept 1: Properties of Transition Metals
Transition metals are located in the central block of the periodic table. Unlike Group 1 alkali metals (which are soft and have low melting points), transition metals have high melting points and high densities. This makes them exceptionally useful as structural materials.
Crucially for your exams, transition metals exhibit three defining chemical characteristics:
1. **They form coloured compounds**: Copper(II) sulfate is blue, iron(II) compounds are pale green, and iron(III) compounds are orange-brown.
2. **They act as catalysts**: They speed up chemical reactions without being used up. For example, iron is the catalyst in the Haber process for making ammonia.
3. **They have variable oxidation states**: They can form ions with different charges (e.g., Fe²⁺ and Fe³⁺).
**Example**: If an exam question describes an unknown metal that is hard, melts at 1538°C, and forms a purple solution when reacted, you can confidently identify it as a transition metal.
### Concept 2: The Structure and Strength of Alloys
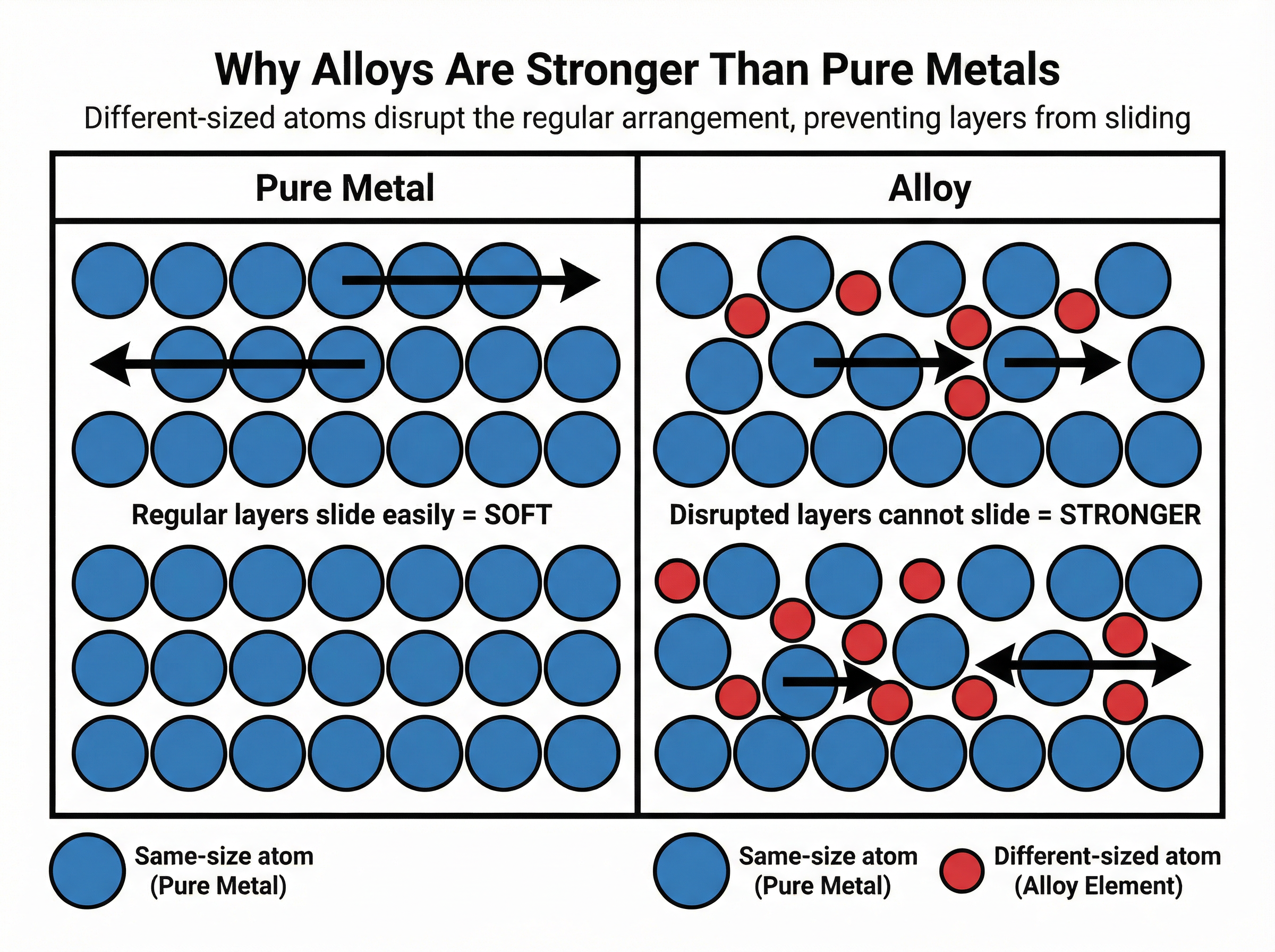
An alloy is a mixture of a metal with at least one other element. Pure metals are relatively soft because their atoms are arranged in regular layers that can easily slide over one another when a force is applied.
When another element is added to make an alloy, the different-sized atoms disrupt this regular lattice arrangement. Because the layers are no longer perfectly aligned, they cannot slide past each other easily. This disruption of the layers is exactly why alloys are harder and stronger than pure metals.
**Example**: Steel is an alloy of iron and carbon. The small carbon atoms fit into the spaces between the larger iron atoms, distorting the regular layers and making the steel significantly stronger than pure iron.
### Concept 3: Corrosion and Rusting
Corrosion is the destruction of materials by chemical reactions with substances in the environment. Rusting is a specific type of corrosion that only applies to iron (and its alloy, steel).
Rusting is an oxidation reaction where iron reacts with oxygen and water to form hydrated iron(III) oxide (rust). The golden rule for the exam is that **both oxygen AND water are required** for rusting to occur. If either is absent, the iron will not rust.
### Concept 4: Preventing Rusting
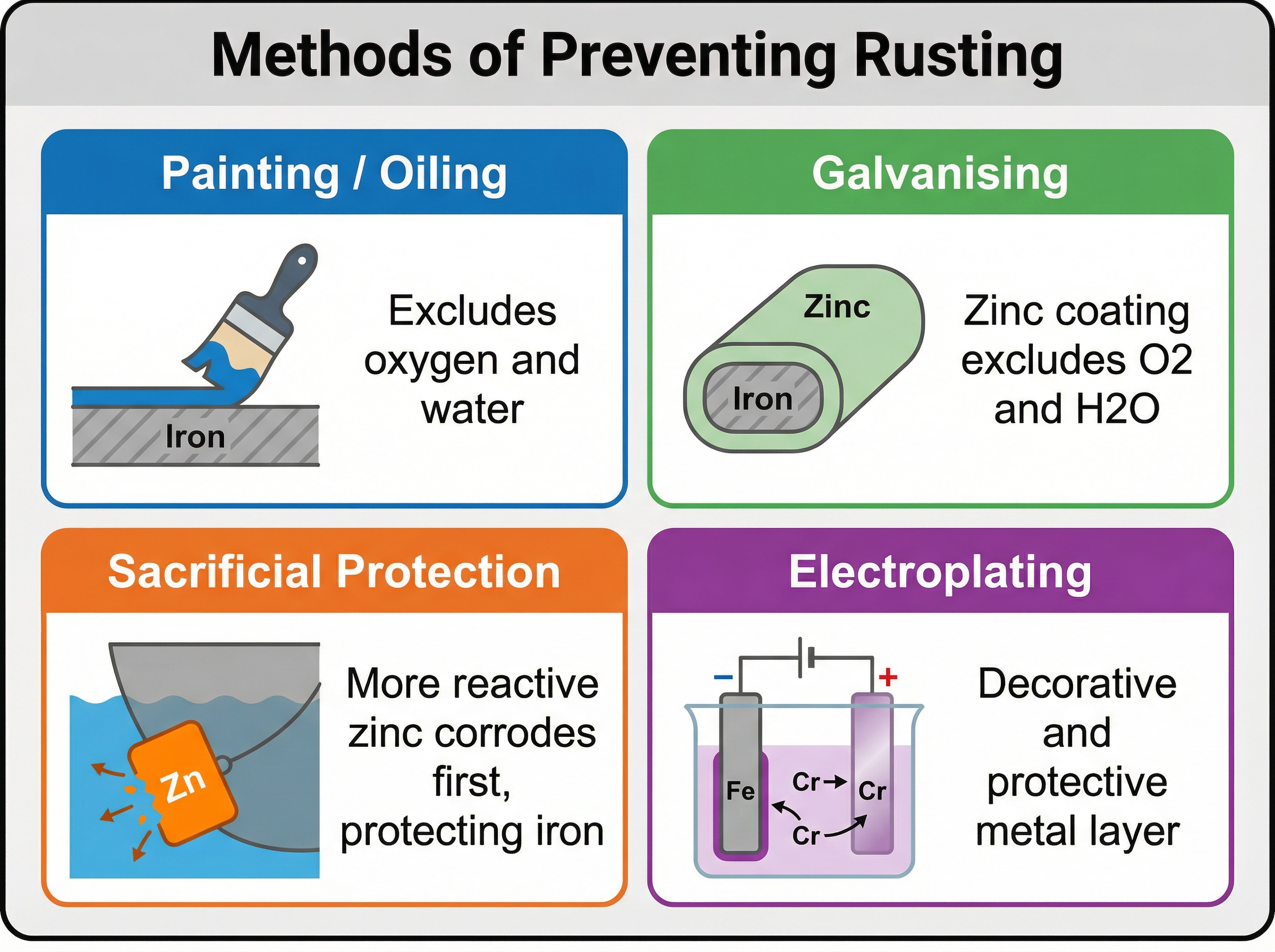
We can prevent rusting using several methods, which generally fall into two categories: barrier methods and sacrificial methods.
- **Barrier Methods**: Painting, oiling, greasing, or coating with plastic. These work by physically excluding oxygen and water from reaching the iron surface.
- **Galvanising**: Coating iron with a layer of zinc. This acts as a barrier, but even if scratched, the zinc provides sacrificial protection.
- **Sacrificial Protection**: Attaching a block of a more reactive metal (like zinc or magnesium) to the iron. Because the attached metal is higher in the reactivity series, it oxidises (corrodes) preferentially, sacrificing itself to protect the iron.
- **Electroplating**: Using electrolysis to coat the iron with a thin layer of another metal (like chromium) to improve appearance and prevent corrosion.
## Mathematical/Scientific Relationships
The word equation for rusting is frequently required in exams:
**Iron + Oxygen + Water → Hydrated Iron(III) Oxide**
*(Note: You are rarely asked for the balanced symbol equation for rusting due to the variable nature of hydration, but you must know the word equation perfectly).*
## Practical Applications
- **Aluminium Alloys**: Used in aircraft manufacture because they combine the low density of aluminium with the high strength of an alloy.
- **Bronze (Copper + Tin)**: Used for statues and medals due to its hardness and resistance to corrosion.
- **Brass (Copper + Zinc)**: Used for musical instruments and door fittings because it is hard but can be machined into intricate shapes.
- **Sacrificial Protection in Action**: Blocks of magnesium or zinc are bolted to the steel hulls of ships or underground iron pipes to prevent them from rusting away.