Separate chemistry 1 — Edexcel GCSE Study Guide
Exam Board: Edexcel | Level: GCSE
Master the principles of reversible reactions, dynamic equilibrium, and the Haber process. This essential guide breaks down how conditions like temperature, pressure, and concentration affect equilibrium, equipping you to score top marks on 6-mark industrial chemistry questions.
## Overview

Reversible reactions and dynamic equilibrium form the backbone of industrial chemistry. This topic explores reactions that can go in both directions and how chemists manipulate conditions to maximise the yield of useful products like ammonia in the Haber Process. It is a crucial area of the GCSE Chemistry specification because it tests your ability to apply theoretical principles—specifically Le Chatelier's Principle—to real-world economic and safety considerations.
Examiners frequently assess this topic through extended 6-mark questions where you must evaluate the compromises made in industrial conditions. Understanding the 'why' behind these compromises is just as important as memorising the numbers. This topic synoptically links to rates of reaction, energy changes (exothermic and endothermic reactions), and the environmental impact of fertilisers.
Listen to our comprehensive revision podcast to solidify your understanding:

## Key Concepts
### Concept 1: Reversible Reactions and Dynamic Equilibrium
Most chemical reactions are irreversible; reactants turn into products and the reaction stops. However, in **reversible reactions**, the products can react together to reform the original reactants. We represent this using the equilibrium symbol (⇌).
When a reversible reaction occurs in a **closed system** (where no reactants or products can escape), it eventually reaches a state called **dynamic equilibrium**. At this point, the forward and reverse reactions are occurring at exactly the same rate. Because they are balanced, the overall concentrations of reactants and products remain constant. It is vital to remember that the reactions have *not* stopped; they are simply happening at equal speeds, like a person walking up a down escalator at the exact same speed the escalator is moving.
**Example**: The thermal decomposition of ammonium chloride.
Ammonium chloride ⇌ Ammonia + Hydrogen chloride
$NH_4Cl_{(s)} \rightleftharpoons NH_3_{(g)} + HCl_{(g)}$
Heating moves the reaction forward (endothermic), while cooling moves it backward (exothermic).
### Concept 2: Le Chatelier's Principle
Le Chatelier's Principle is your key to predicting how an equilibrium will shift. It states that if a change is made to the conditions of a system at equilibrium, the system will shift to oppose that change.
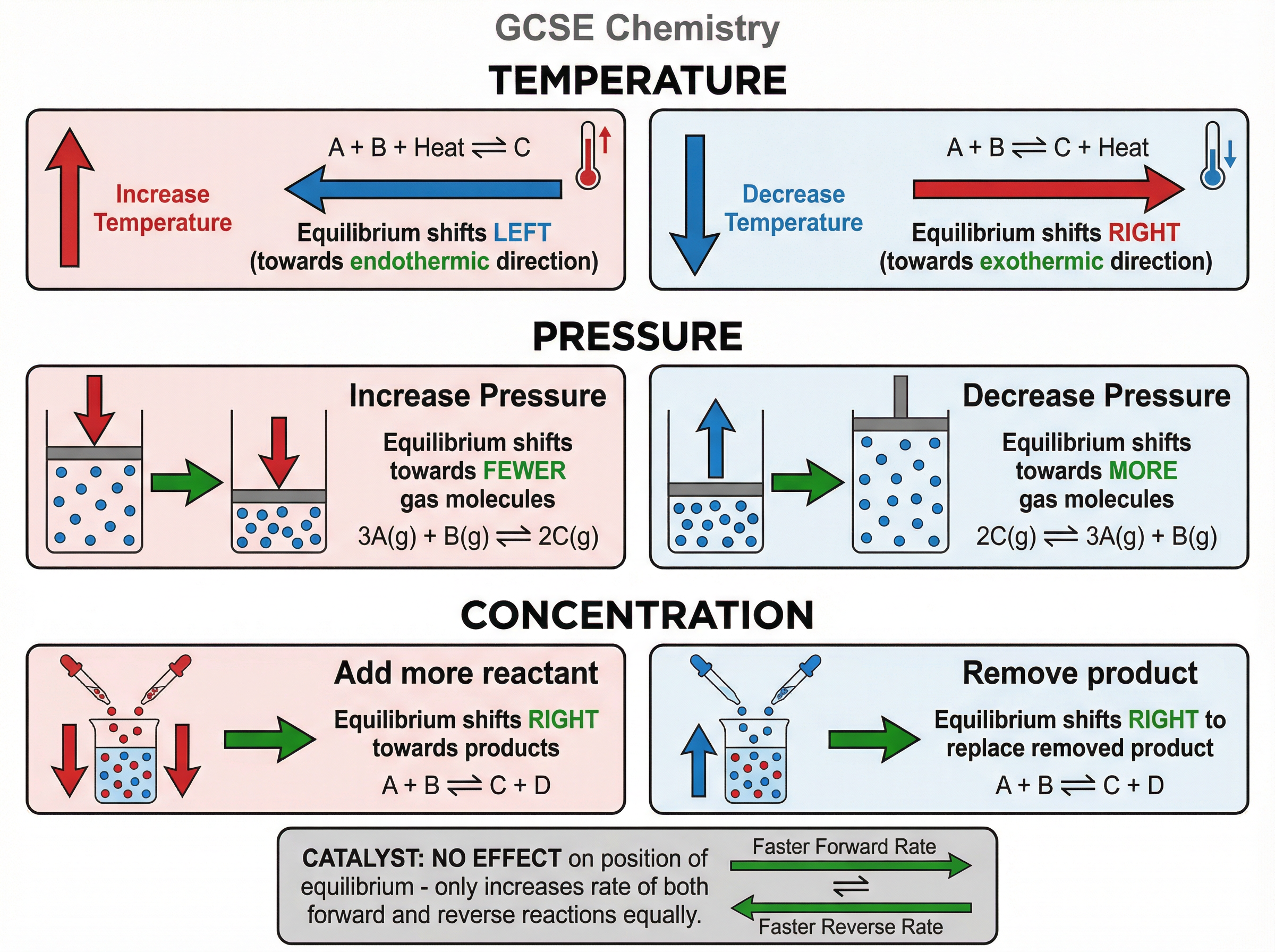
* **Temperature**: Increasing the temperature shifts the equilibrium in the **endothermic** direction to absorb the extra heat. Decreasing the temperature shifts it in the **exothermic** direction to release heat.
* **Pressure** (for gases): Increasing pressure shifts the equilibrium to the side with **fewer molecules of gas** to reduce the pressure. Decreasing pressure shifts it to the side with more gas molecules.
* **Concentration**: Adding more reactant shifts the equilibrium to the right (towards products) to use it up. Removing a product also shifts it to the right to replace what was lost.
### Concept 3: The Haber Process
The Haber Process is the industrial method for manufacturing ammonia ($NH_3$), which is essential for making nitrogen-based fertilisers. The raw materials are nitrogen (extracted from the air) and hydrogen (obtained from natural gas/methane).
The reversible equation is:
$N_{2(g)} + 3H_{2(g)} \rightleftharpoons 2NH_{3(g)}$
*(The forward reaction is exothermic)*
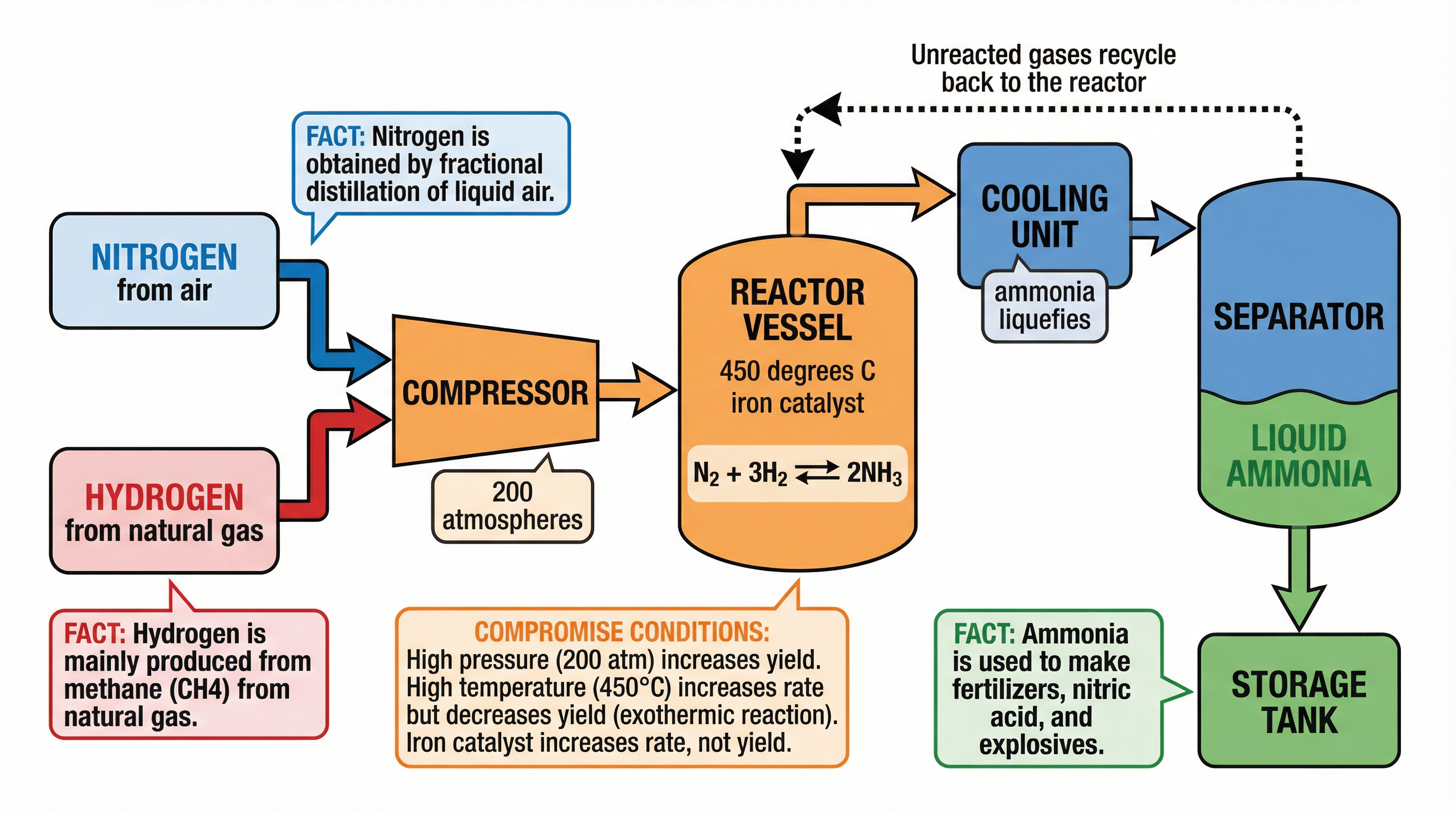
To make the process economically viable, chemists must balance the *yield* of ammonia with the *rate* of reaction and the *cost* of equipment.
## Mathematical/Scientific Relationships
* **Molecule counting for pressure shifts**: In the Haber equation ($N_2 + 3H_2 \rightleftharpoons 2NH_3$), there are 4 moles of gas on the left and 2 moles of gas on the right. Therefore, high pressure favours the right-hand side.
* **Yield percentage**: The actual industrial yield of the Haber process is only around 15%. However, unreacted nitrogen and hydrogen are continuously recycled back into the reactor, making the overall process highly efficient and reducing waste.
## Practical Applications
The primary application of the Haber Process is the production of ammonia for **fertilisers** (e.g., ammonium nitrate). These fertilisers provide plants with the essential nitrogen needed to synthesise proteins for growth. Without the Haber Process, global agricultural output would plummet, leading to widespread food shortages. Ammonia is also a precursor for manufacturing nitric acid and explosives.