Separate chemistry 2 — Edexcel GCSE Study Guide
Exam Board: Edexcel | Level: GCSE
Master the fundamentals of hydrocarbons with this essential guide to alkanes and alkenes. You'll learn to draw structural formulas, distinguish saturated from unsaturated molecules, and ace the classic bromine water test—key skills that frequently appear in 4-6 mark exam questions.
## Overview
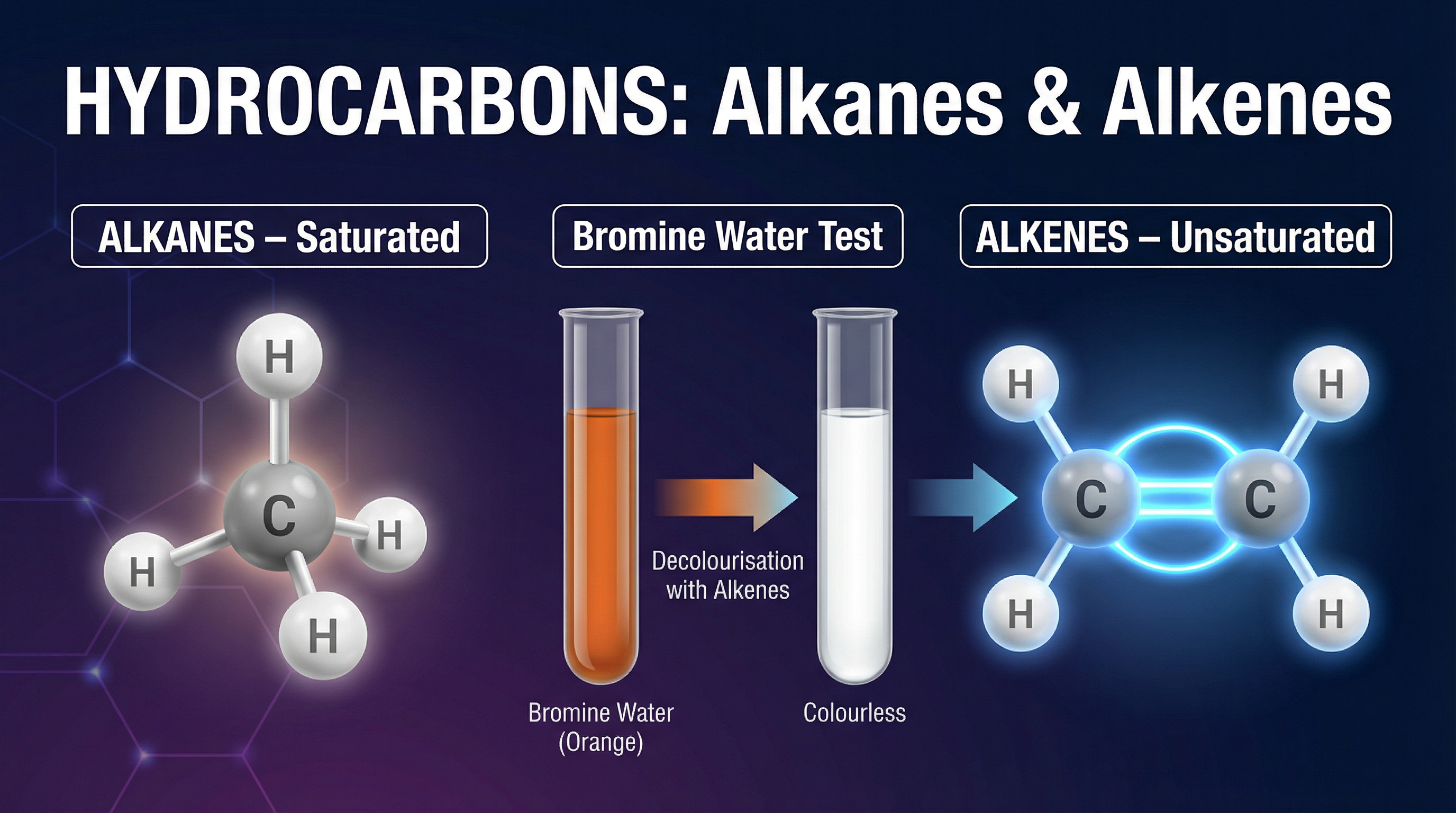
This topic is the foundation of organic chemistry. Hydrocarbons—compounds containing only carbon and hydrogen—are the building blocks of fuels, plastics, and many materials we use daily. In your exam, you will be expected to draw their structures accurately, explain their properties, and write balanced chemical equations for their reactions.
Understanding the difference between alkanes (saturated) and alkenes (unsaturated) is crucial. Examiners frequently test this through the bromine water practical and by asking you to identify functional groups. This topic also links heavily to crude oil fractional distillation and the environmental impacts of combustion.

## Key Concepts
### Concept 1: Alkanes (Saturated Hydrocarbons)
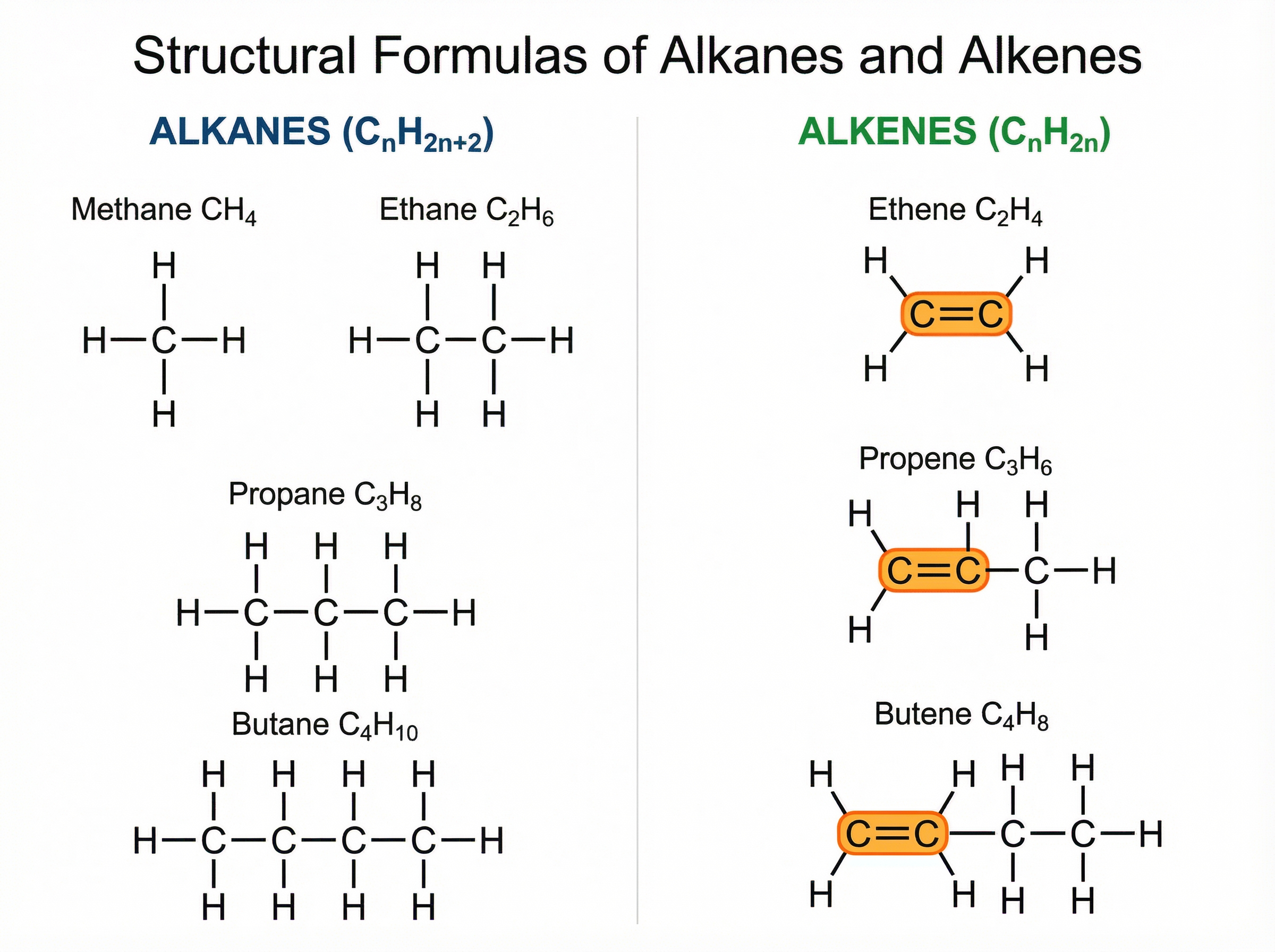
Alkanes are a homologous series of hydrocarbons with the general formula C\textsubscript{n}H\textsubscript{2n+2}. They are described as **saturated** because they contain only single covalent bonds between carbon atoms (C-C). Every carbon atom is bonded to the maximum possible number of hydrogen atoms.
**Example**: Methane (CH\textsubscript{4}) is the simplest alkane. Ethane (C\textsubscript{2}H\textsubscript{6}), propane (C\textsubscript{3}H\textsubscript{8}), and butane (C\textsubscript{4}H\textsubscript{10}) follow in the series. When drawing these, you must show every single bond as a distinct line connecting the atoms.
### Concept 2: Alkenes (Unsaturated Hydrocarbons)
Alkenes are a homologous series with the general formula C\textsubscript{n}H\textsubscript{2n}. They are **unsaturated** because they contain a carbon-carbon double bond (C=C). This double bond is their functional group and makes them significantly more reactive than alkanes.
**Example**: Ethene (C\textsubscript{2}H\textsubscript{4}) is the simplest alkene. The double bond means there is 'room' for other atoms to add onto the molecule, which is why alkenes undergo addition reactions.
### Concept 3: The Bromine Water Test
This is the standard test to distinguish between an alkane and an alkene. Bromine water is an orange-brown solution.
- **With an Alkane**: There is no reaction under normal conditions. The solution remains orange.
- **With an Alkene**: The solution decolourises (turns colourless). This happens because the alkene undergoes an **addition reaction** with the bromine across the C=C double bond, forming a colourless dibromoalkane.
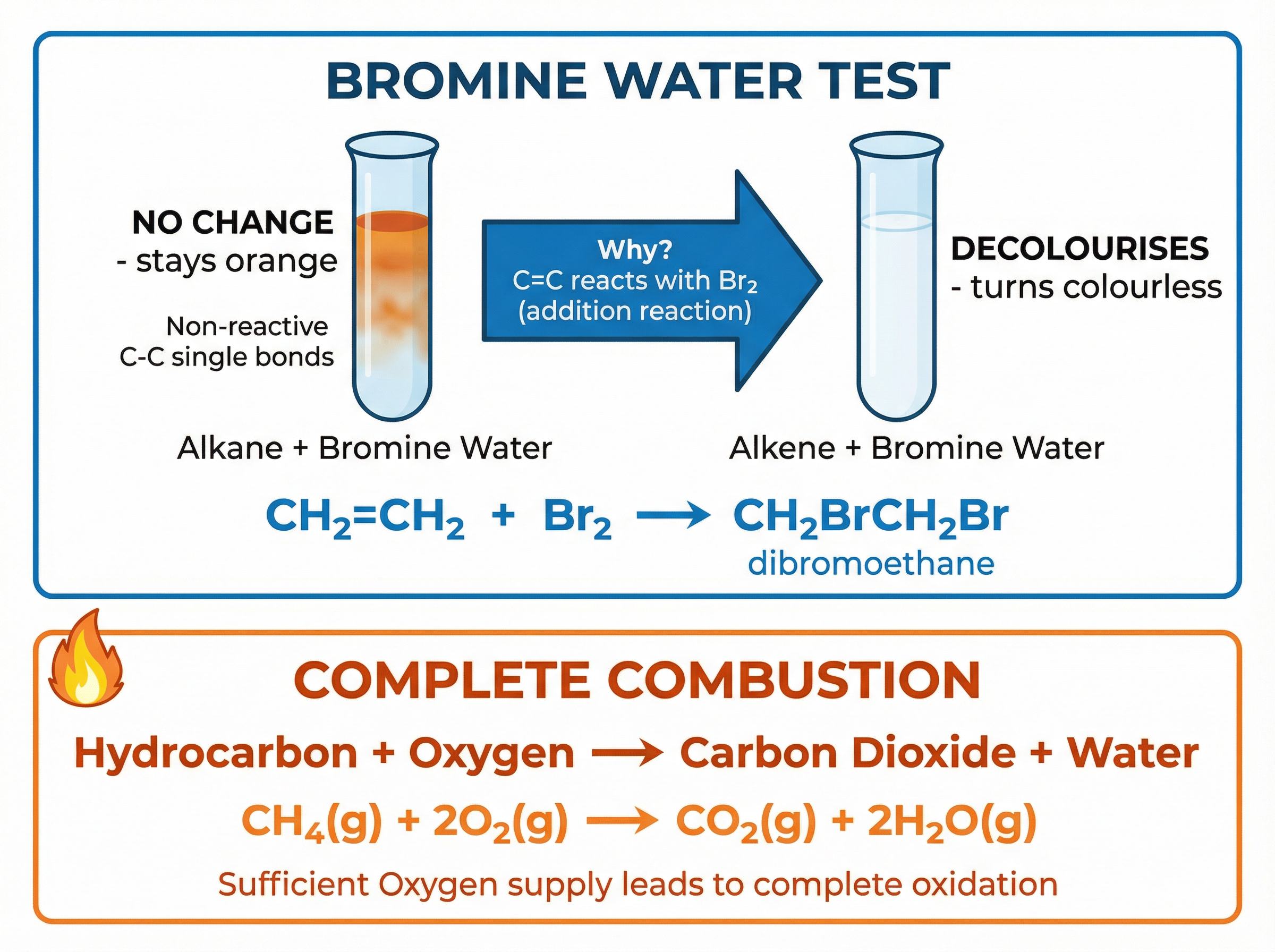
### Concept 4: Complete Combustion
When hydrocarbons burn in a plentiful supply of oxygen, they undergo complete combustion. The carbon is oxidised to carbon dioxide, and the hydrogen is oxidised to water. This reaction releases a significant amount of energy.
**Example Equation**: CH\textsubscript{4}(g) + 2O\textsubscript{2}(g) $\rightarrow$ CO\textsubscript{2}(g) + 2H\textsubscript{2}O(g)
## Mathematical/Scientific Relationships
- **Alkane General Formula**: $C_nH_{2n+2}$ (Must memorise)
- **Alkene General Formula**: $C_nH_{2n}$ (Must memorise)
## Practical Applications
Alkanes are primarily used as fuels (e.g., methane in natural gas, octane in camping stoves) due to the large amount of energy released during combustion. Alkenes are the crucial starting materials for making polymers (plastics) like poly(ethene), which is used in shopping bags and bottles.