Separate chemistry 2 — Edexcel GCSE Study Guide
Exam Board: Edexcel | Level: GCSE
Master the fascinating world of Nanoparticles, where materials behave entirely differently due to their incredibly small size. This topic is a favourite for examiners to test your understanding of surface area to volume ratios, standard form calculations, and evaluating the risks versus benefits of cutting-edge technology.

## Overview
Welcome to the microscopic world of Nanoparticles, a cutting-edge topic in Separate Chemistry that bridges the gap between atomic structure and bulk materials. A nanoparticle is defined as a particle with dimensions ranging from 1 to 100 nanometres (nm). To put this in perspective, they are larger than individual atoms and molecules but vastly smaller than anything visible under a standard light microscope.
This topic is crucial because materials at the nanoscale exhibit entirely different properties compared to their bulk counterparts. Examiners frequently test your ability to explain *why* these properties change—primarily due to the massive increase in the surface area to volume ratio. You will also be expected to confidently calculate these ratios, convert units using standard form, and evaluate the real-world applications and potential risks of nanotechnology.
Listen to the podcast below for a comprehensive 10-minute revision session covering all the key concepts, common pitfalls, and a quick-fire recall quiz.

## Key Concepts
### Concept 1: Scale and Standard Form
Nanoparticles are incredibly small. A nanometre is one billionth of a metre, written in standard form as $1 \times 10^{-9}$ m. Therefore, nanoparticles range from $1 \times 10^{-9}$ m to $1 \times 10^{-7}$ m.
Examiners love to test your sense of scale. A typical atom has a diameter of about 0.1 nm ($1 \times 10^{-10}$ m). This means a small nanoparticle contains only a few dozen atoms, while larger ones contain thousands. For comparison, a human hair is roughly 80,000 nm thick. You must be able to convert between metres, millimetres, micrometres, and nanometres seamlessly.
**Example**: If a nanoparticle has a diameter of 50 nm, this is $50 \times 10^{-9}$ m. However, proper standard form requires the first number to be between 1 and 10. So, it must be written as $5.0 \times 10^{-8}$ m.
### Concept 2: Surface Area to Volume Ratio (SA:V)
This is the most critical concept in the topic. As a particle decreases in size, its surface area to volume ratio increases dramatically.
Why does this matter? Chemical reactions occur on the surface of a material. Because nanoparticles have such a massive surface area relative to their tiny volume, a much higher percentage of their atoms are exposed on the surface and available to react. This makes them incredibly reactive and highly effective as catalysts.
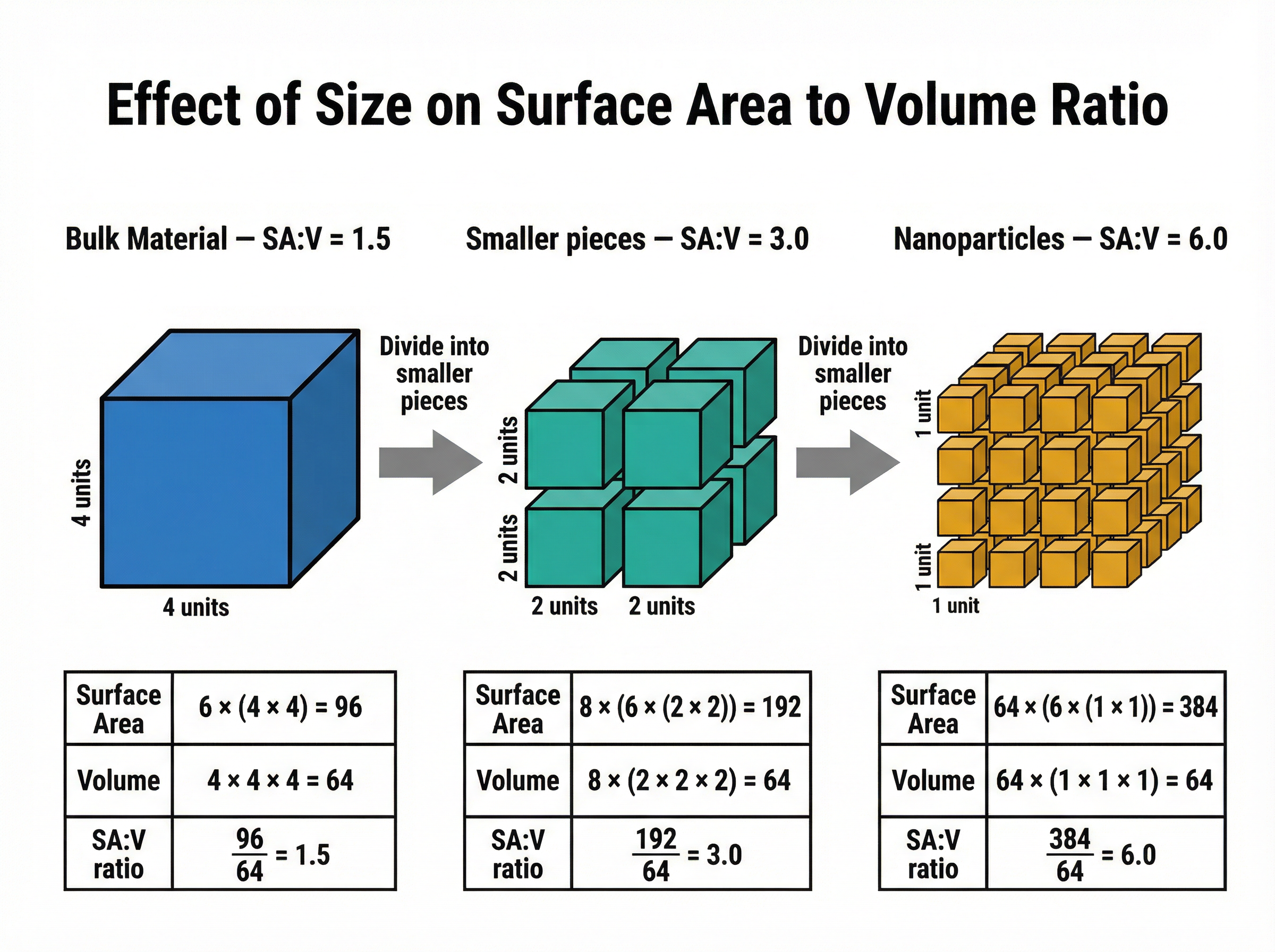
**Example**: Consider a cube with a side length of 10 cm.
- Surface Area = $6 \times (10 \times 10) = 600$ cm²
- Volume = $10 \times 10 \times 10 = 1000$ cm³
- SA:V Ratio = $600 / 1000 = 0.6$
Now, if we cut that cube into smaller 1 cm cubes:
- The total volume remains 1000 cm³.
- The new total surface area is $1000 \times (6 \times 1 \times 1) = 6000$ cm².
- The new SA:V Ratio = $6000 / 1000 = 6.0$.
The ratio has increased tenfold simply by making the pieces smaller!
### Concept 3: Applications and Risks
Because of their unique properties, nanoparticles have revolutionary applications. However, because the technology is relatively new, the long-term impacts on health and the environment are not fully understood.
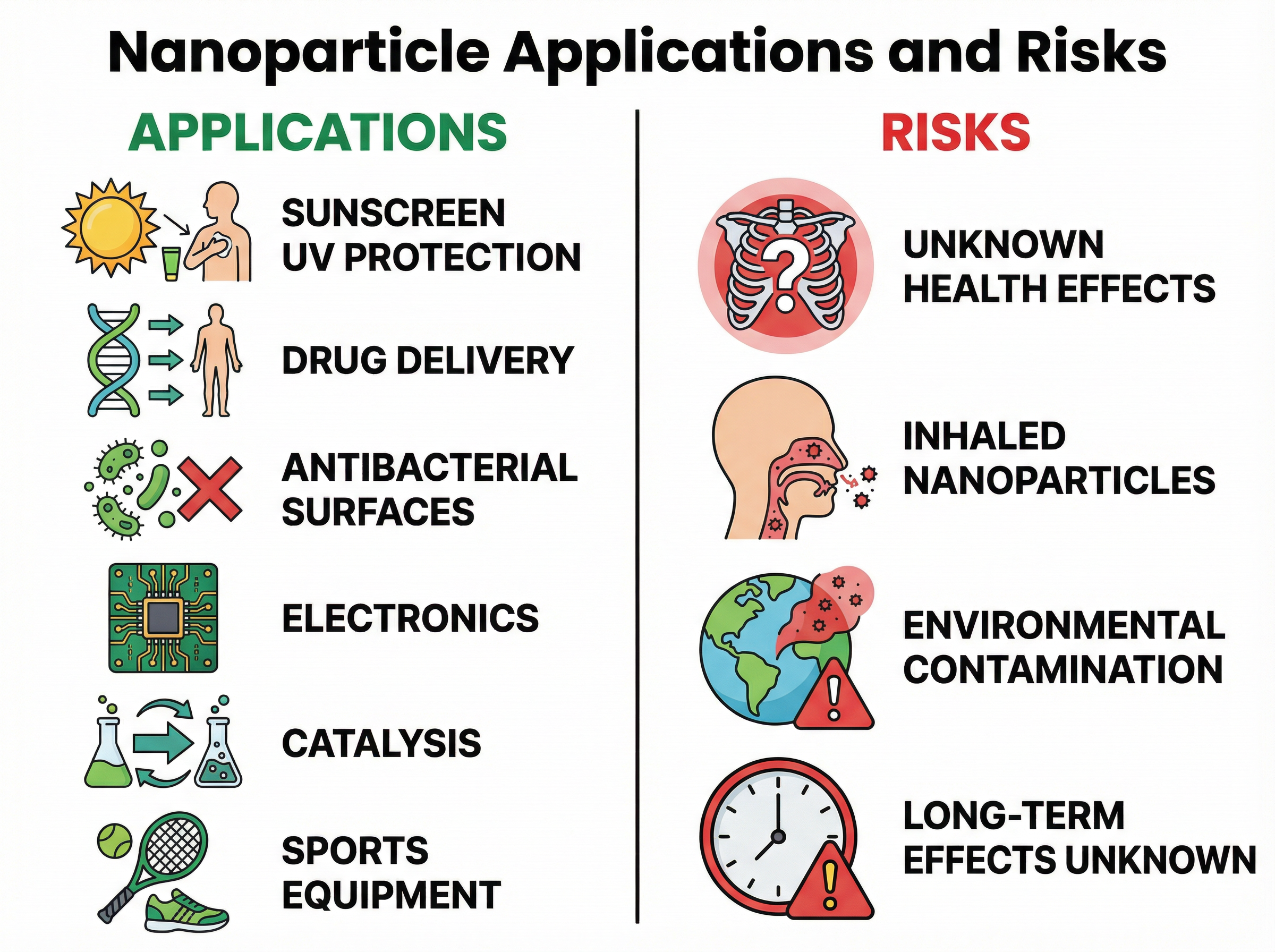
**Applications**:
- **Sunscreens**: Titanium dioxide and zinc oxide nanoparticles are used because they absorb harmful UV radiation but are so small they appear transparent on the skin, avoiding the white residue of traditional sunscreens.
- **Medicine**: Nanoparticles can deliver drugs directly to targeted cells (like cancer cells), improving efficacy and reducing side effects.
- **Antibacterial Materials**: Silver nanoparticles have strong antibacterial properties and are woven into socks and wound dressings.
- **Catalysts**: Their huge SA:V ratio means less material is needed to provide the same catalytic effect, saving money in industrial processes.
**Risks**:
- **Health**: Due to their tiny size, nanoparticles can be inhaled deep into the lungs or pass through cell membranes into the bloodstream. The long-term toxicity is unknown.
- **Environment**: When washed away (e.g., silver nanoparticles from socks), they may accumulate in aquatic ecosystems, potentially harming fish and disrupting food chains.
## Mathematical/Scientific Relationships
**Surface Area of a Cube**: $6 \times l^2$ (where $l$ is the side length)
**Volume of a Cube**: $l^3$
**Surface Area to Volume Ratio**: $\frac{\text{Surface Area}}{\text{Volume}}$
**Standard Form Rules**:
- 1 millimetre (mm) = $1 \times 10^{-3}$ m
- 1 micrometre (µm) = $1 \times 10^{-6}$ m
- 1 nanometre (nm) = $1 \times 10^{-9}$ m
*Must memorise*: You must know how to calculate the surface area and volume of a cube and calculate the ratio. This formula is NOT given in the exam.