Separate chemistry 2 — Edexcel GCSE Study Guide
Exam Board: Edexcel | Level: GCSE
Master the structure, properties, and reactions of alcohols and carboxylic acids. These essential organic compounds are central to industrial chemistry, from the fermentation of ethanol to the production of vinegar, and frequently feature in high-mark exam questions.
## Overview
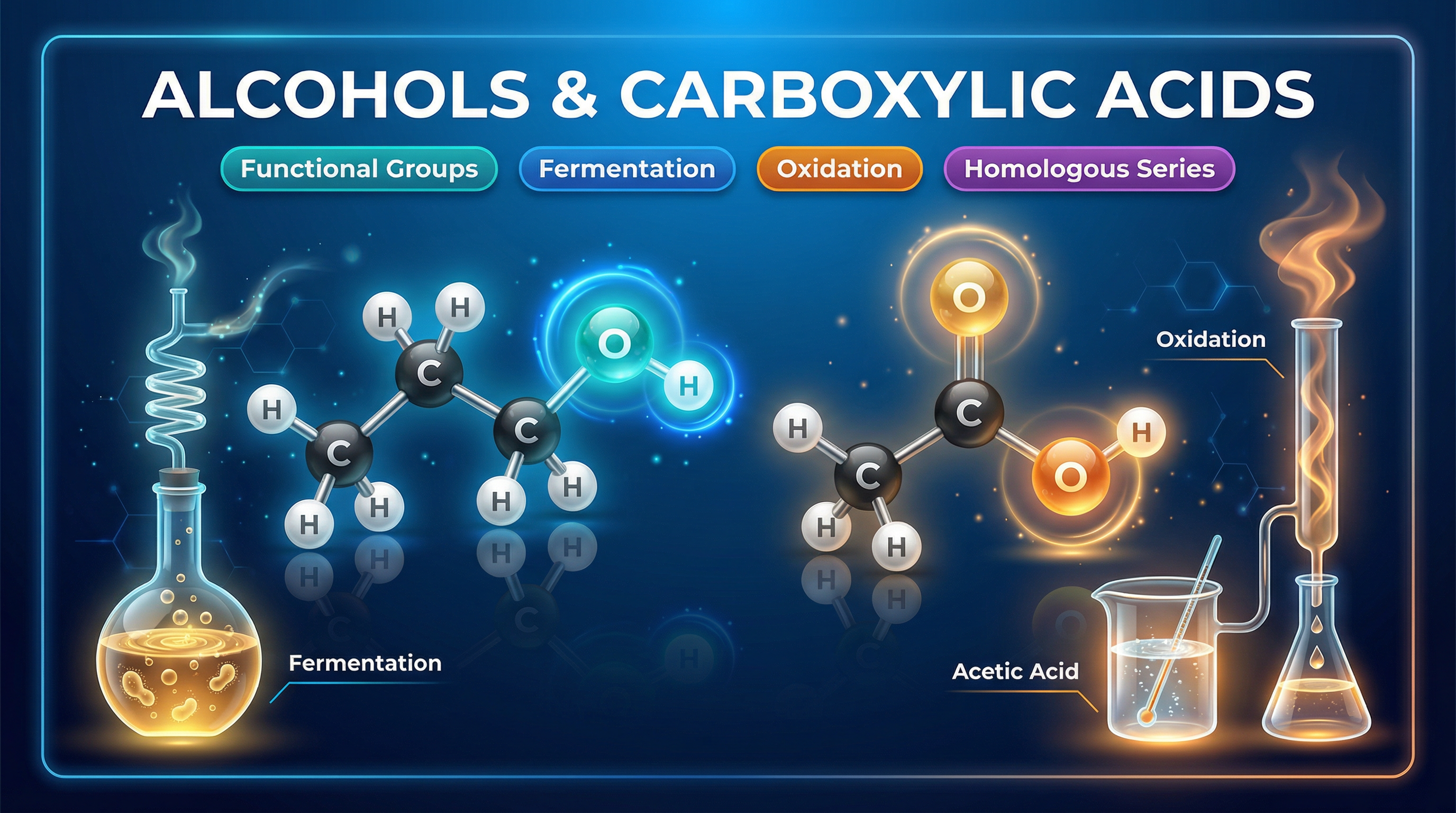
Welcome to Separate Chemistry 2: Alcohols and Carboxylic Acids. This topic is a cornerstone of organic chemistry, building directly on your knowledge of alkanes and alkenes. It introduces two vital homologous series that you interact with every day: alcohols (like the ethanol in biofuels and drinks) and carboxylic acids (like the ethanoic acid in vinegar).
Examiners love this topic because it links structural chemistry with practical applications. You will be expected to draw molecular structures, explain the conditions required for industrial processes like fermentation, and predict the outcomes of oxidation and acid-base reactions. Understanding the specific functional groups (-OH for alcohols and -COOH for carboxylic acids) is the key to unlocking the top grades. Let's dive into the chemistry that makes these molecules so unique and useful.
## Key Concepts
### Concept 1: Functional Groups and Homologous Series
Organic chemistry is organised by families called homologous series. A homologous series is a group of related organic compounds that have the same functional group, share similar chemical properties, and show a gradual variation in physical properties (such as boiling point). Each successive member of a homologous series differs from the previous one by a -CH₂- unit.
The functional group is the specific atom or group of atoms responsible for the characteristic chemical reactions of that molecule.
For alcohols, the functional group is the **hydroxyl group**, written as **-OH**.
For carboxylic acids, the functional group is the **carboxyl group**, written as **-COOH**.
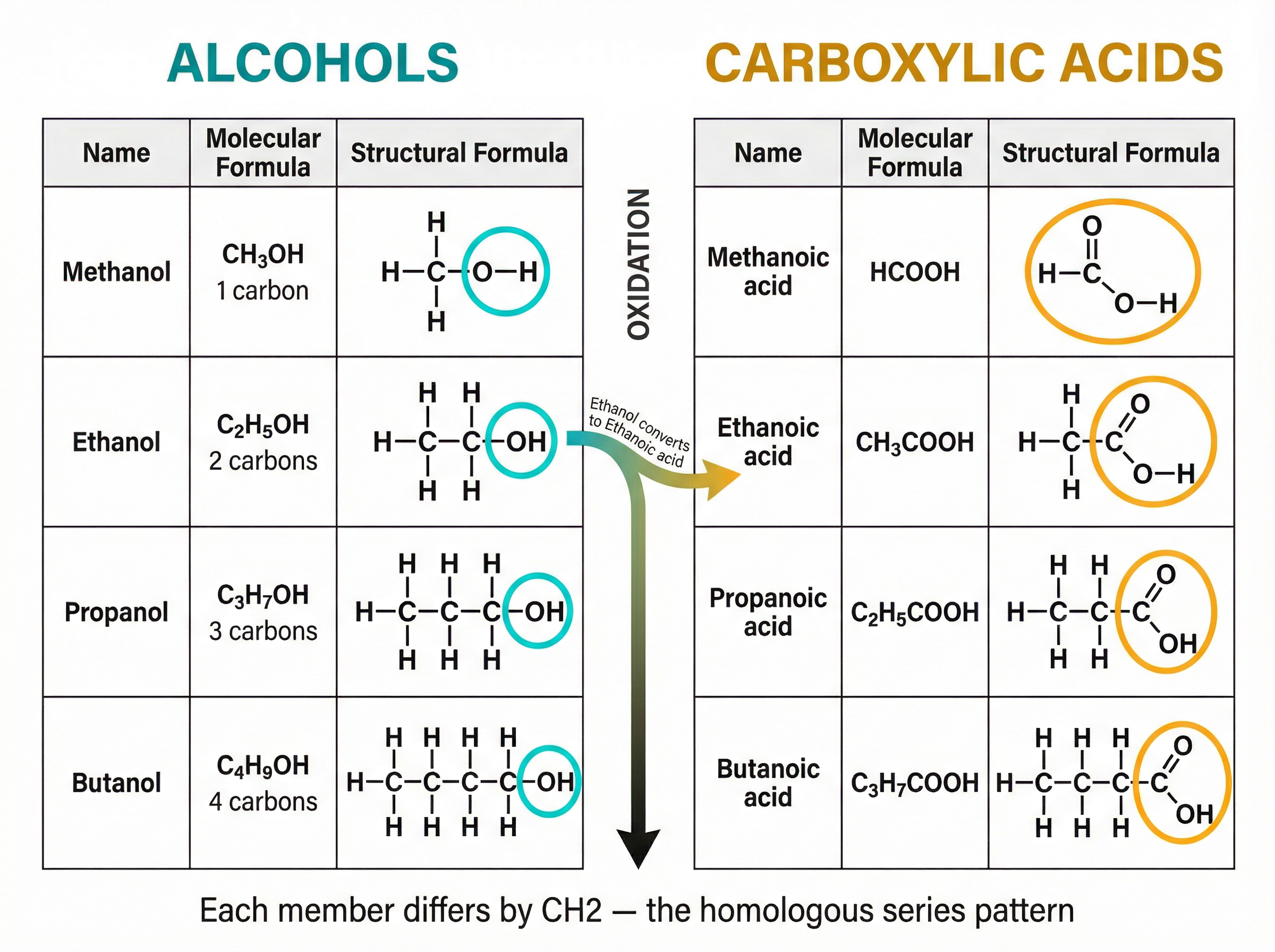
**Example**: Methanol (CH₃OH) and ethanol (C₂H₅OH) are both alcohols. They react similarly because they both contain the -OH group. However, ethanol has a higher boiling point because it has a longer carbon chain, leading to stronger intermolecular forces.
### Concept 2: The Production of Ethanol
Ethanol is the most widely used alcohol. You must know two methods for producing it: fermentation and the hydration of ethene.
**Fermentation** is a biological process where yeast enzymes convert glucose into ethanol and carbon dioxide.
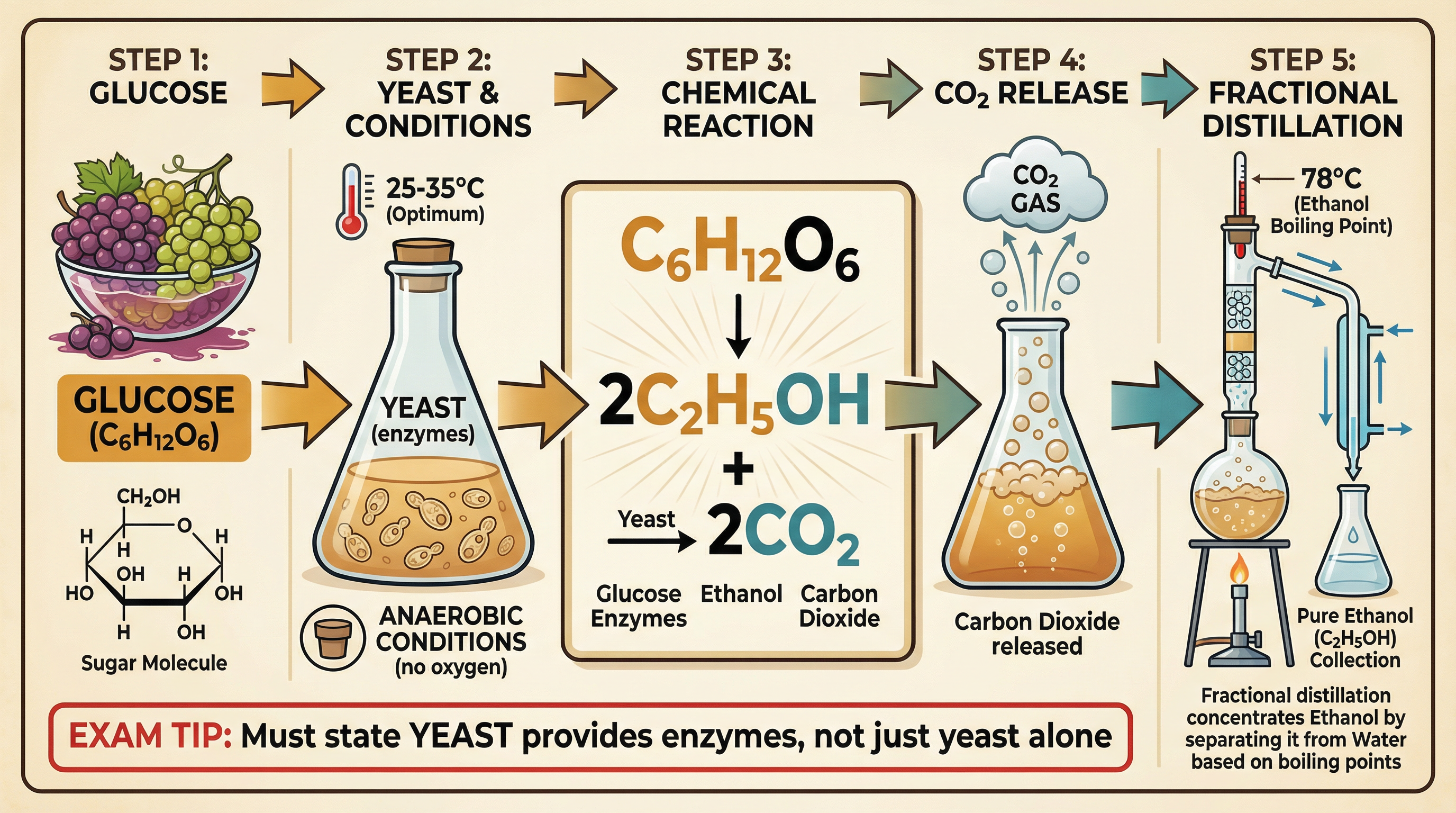
The conditions for fermentation are highly specific and frequently tested:
1. **Aqueous solution of glucose**: The raw material.
2. **Yeast**: This provides the enzymes that catalyse the reaction.
3. **Anaerobic conditions (no oxygen)**: If oxygen is present, the yeast will respire aerobically, producing carbon dioxide and water instead of ethanol.
4. **Temperature of 25°C to 35°C**: This is the optimum temperature for the yeast enzymes. If it is too cold, the reaction is too slow. If it is too hot, the enzymes denature and the reaction stops.
Fermentation produces a dilute aqueous solution of ethanol (up to about 15%). To produce pure ethanol, this mixture must be separated using **fractional distillation**, which works because ethanol has a lower boiling point (78°C) than water (100°C).
### Concept 3: Oxidation of Alcohols
Alcohols can be oxidised to form carboxylic acids. This requires an oxidising agent, or it can happen naturally through the action of microbes in the air.
When ethanol is oxidised, it forms ethanoic acid. This is the chemical reaction that causes wine to turn into vinegar when left open to the air.
**Equation**: Ethanol + Oxygen (from oxidising agent) → Ethanoic acid + Water
### Concept 4: Reactions of Carboxylic Acids
Carboxylic acids are weak acids. They exhibit the typical reactions of acids, but they only partially ionise in water, meaning they have a higher pH (around 3-5) than strong acids like hydrochloric acid at the same concentration.
They react with:
- **Metals** to form a salt and hydrogen gas.
- **Bases (alkalis)** to form a salt and water.
- **Carbonates** to form a salt, water, and carbon dioxide gas.
**Example**: Ethanoic acid reacts with sodium carbonate to produce sodium ethanoate (the salt), water, and carbon dioxide. You would observe fizzing (effervescence) as the carbon dioxide gas is released.
## Mathematical/Scientific Relationships
- **General formula for Alcohols**: $C_nH_{2n+1}OH$
- **General formula for Carboxylic Acids**: $C_nH_{2n+1}COOH$ (where n is the number of carbon atoms in the alkyl chain, not including the carboxyl carbon)
## Practical Applications
- **Biofuels**: Ethanol produced by fermentation can be used as a renewable fuel for vehicles.
- **Solvents**: Alcohols are excellent solvents for substances that do not dissolve in water, such as perfumes and cosmetics.
- **Vinegar**: A dilute solution of ethanoic acid is used as vinegar for flavouring and preserving food.
## Podcast Summary
Listen to the audio guide for a complete overview of this topic, including exam tips and a quick-fire recall quiz.
