Separate chemistry 2 — Edexcel GCSE Study Guide
Exam Board: Edexcel | Level: GCSE
Master the art of identifying unknown ions! This topic covers the essential chemical tests—from colourful flame tests to characteristic precipitates—and introduces modern instrumental analysis, giving you guaranteed marks if you learn the rules.
## Overview
Chemical analysis is the detective work of chemistry. In this topic, you'll learn how to identify specific cations (positive ions) and anions (negative ions) in unknown salts using simple, qualitative laboratory tests.
This topic is heavily tested in exams because it assesses your ability to recall specific facts (like colours) and your understanding of practical methodology. It links closely to earlier topics on ionic bonding, reactivity series, and balancing equations. Examiners often test this via 'describe the test' questions, or by giving you a table of results and asking you to identify the unknown compound.

## Key Concepts
### Concept 1: Flame Tests for Metal Cations
When metal ions are heated in a Bunsen flame, their electrons absorb thermal energy and jump to higher energy levels. As they fall back to their ground state, they release this energy as visible light. The wavelength (and therefore colour) of this light is unique to each metal ion.
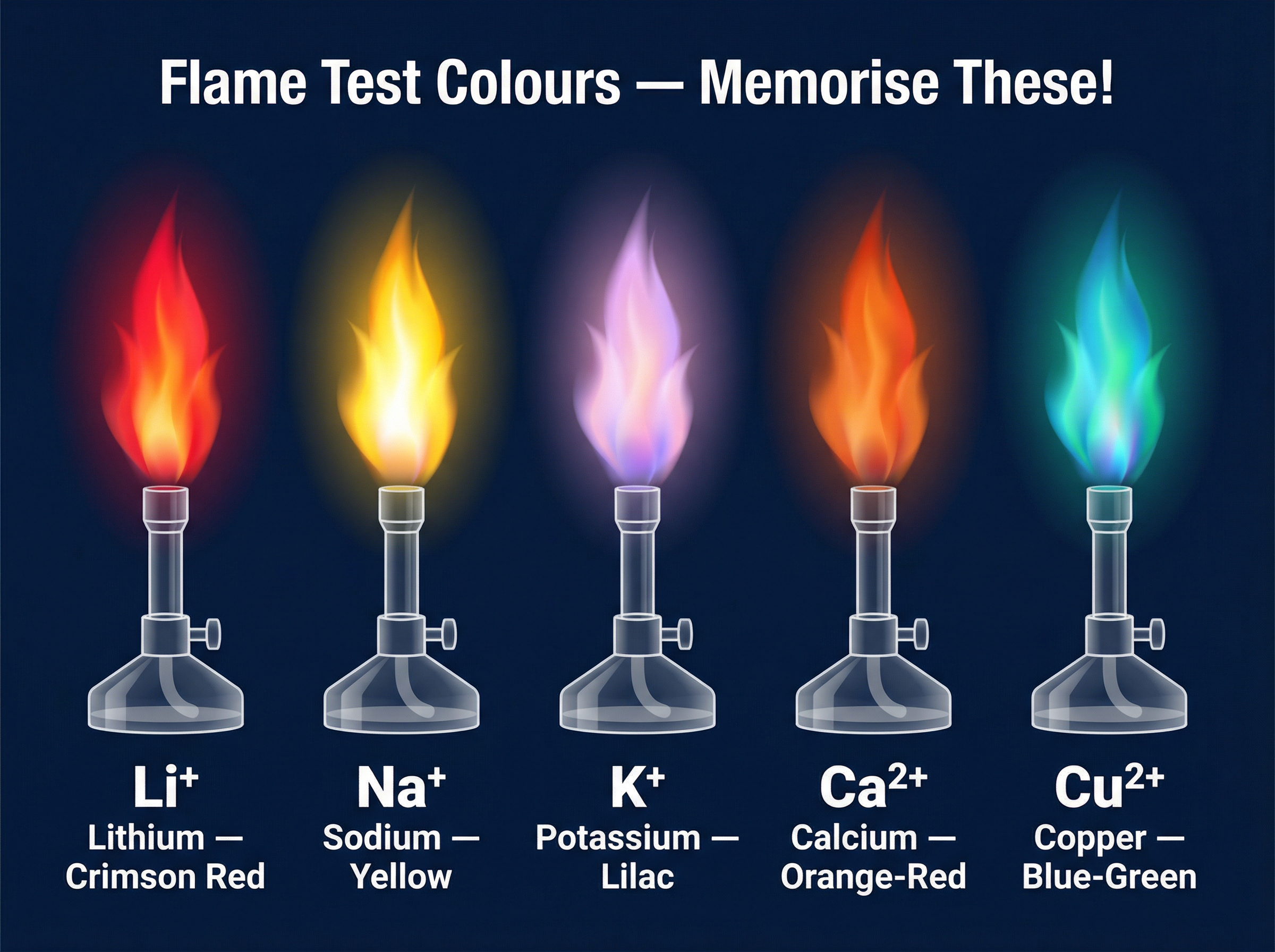
**The Core Colours to Memorise:**
- **Lithium (Li⁺):** Crimson red flame
- **Sodium (Na⁺):** Yellow flame
- **Potassium (K⁺):** Lilac flame
- **Calcium (Ca²⁺):** Orange-red flame
- **Copper (Cu²⁺):** Blue-green flame
*Examiner Tip:* Always clean the nichrome wire loop in concentrated hydrochloric acid before testing to avoid contamination, especially from sodium which has a very intense yellow flame that can mask other colours.
### Concept 2: Sodium Hydroxide Precipitation Tests
Many metal hydroxides are insoluble. When you add sodium hydroxide (NaOH) solution to a solution containing metal cations, an insoluble solid—a precipitate—forms. The colour of this precipitate tells you which ion is present.

**The Core Results:**
- **Aluminium (Al³⁺):** White precipitate. *Crucially, this dissolves in excess NaOH to form a colourless solution.* (Higher Tier focus)
- **Calcium (Ca²⁺):** White precipitate. (Does not dissolve in excess NaOH).
- **Copper(II) (Cu²⁺):** Pale blue precipitate.
- **Iron(II) (Fe²⁺):** Green precipitate.
- **Iron(III) (Fe³⁺):** Reddish-brown precipitate.
**Ammonium Ions (NH₄⁺):** Add NaOH and warm gently. No precipitate forms, but ammonia gas (NH₃) is released. Test the gas with damp red litmus paper—it turns blue because ammonia is alkaline.
### Concept 3: Testing for Anions (Negative Ions)
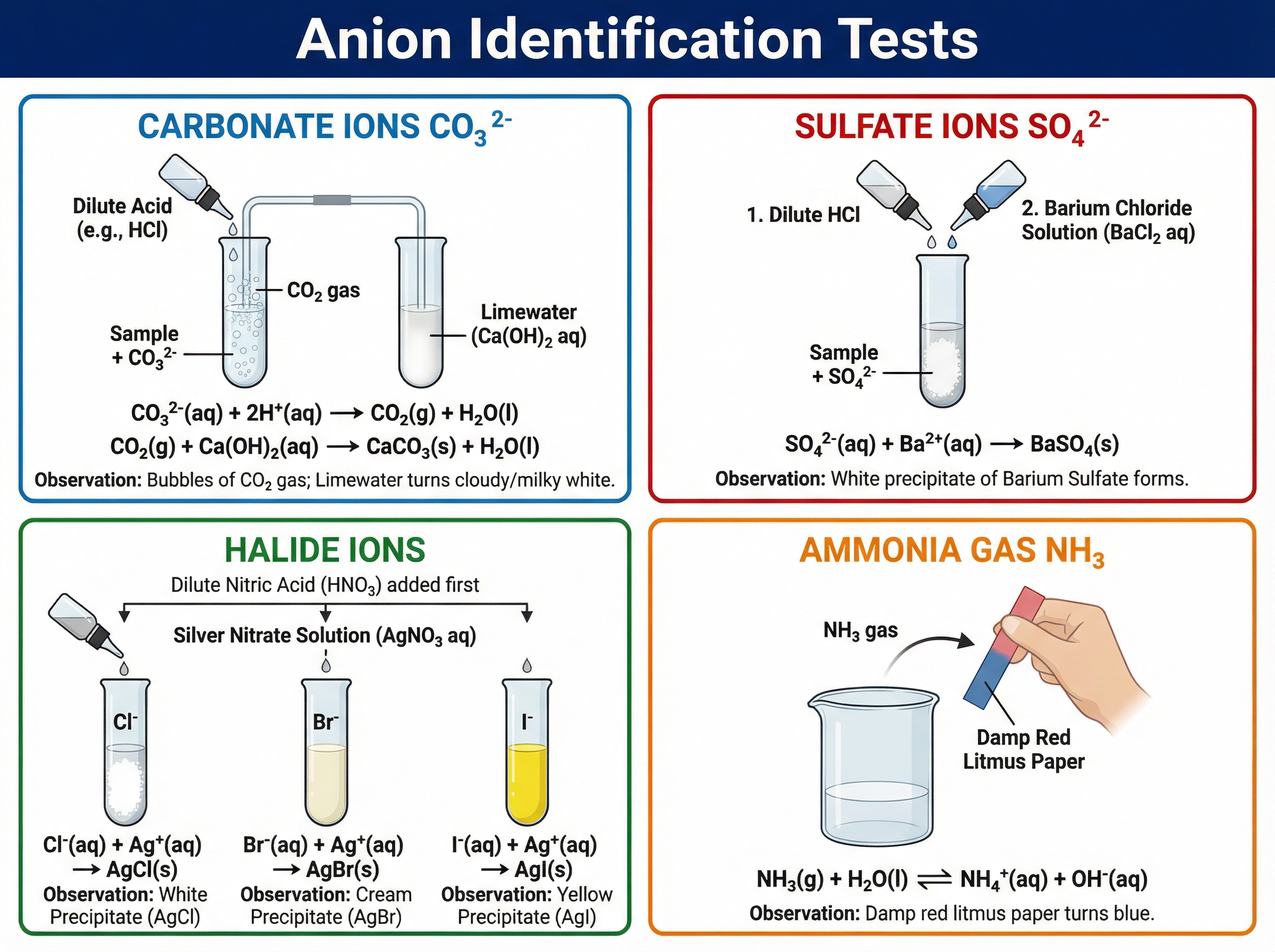
**Carbonate Ions (CO₃²⁻):**
Add dilute acid (e.g., HCl). You will observe effervescence (fizzing). Bubble the gas produced through limewater. If it turns cloudy/milky, the gas is carbon dioxide, confirming carbonate ions.
**Sulfate Ions (SO₄²⁻):**
1. Add dilute hydrochloric acid (HCl) to remove any carbonate impurities that would give a false positive.
2. Add barium chloride (BaCl₂) solution.
3. Result: A white precipitate of barium sulfate forms.
**Halide Ions (Cl⁻, Br⁻, I⁻):**
1. Add dilute nitric acid (HNO₃) to remove impurities.
2. Add silver nitrate (AgNO₃) solution.
3. Results:
- **Chloride (Cl⁻):** White precipitate (Silver chloride)
- **Bromide (Br⁻):** Cream precipitate (Silver bromide)
- **Iodide (I⁻):** Yellow precipitate (Silver iodide)
### Concept 4: Instrumental Methods (Flame Photometry)
Modern laboratories use instrumental methods like flame photometry instead of manual flame tests.
**Advantages of instrumental methods:**
- **Highly sensitive:** Can detect very small amounts of substances.
- **Highly accurate:** Removes human error in judging colours.
- **Rapid:** Gives results much faster than manual testing.
- **Quantitative:** Can tell you *how much* of an ion is present, not just that it is there.
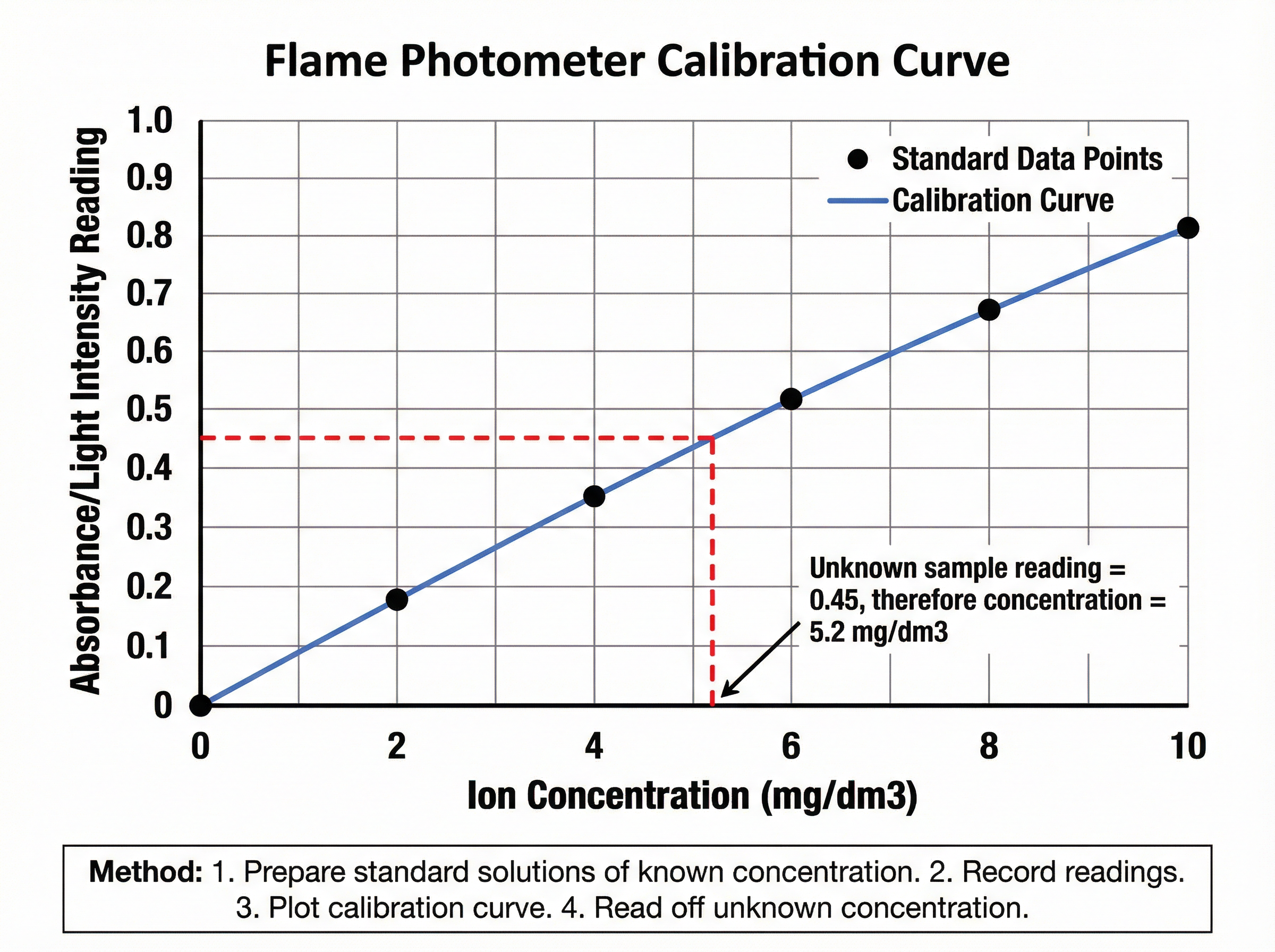
A flame photometer measures the light intensity of the flame. By comparing the reading of an unknown sample against a **calibration curve** (created using standard solutions of known concentrations), you can determine the exact concentration of the ion.
## Mathematical/Scientific Relationships
**Ionic Equations for Precipitation:**
Examiners often ask for the ionic equation for a precipitation reaction. It always follows this pattern:
Cation(aq) + Anion(aq) → Solid Precipitate(s)
*Example (Copper Hydroxide):*
Cu²⁺(aq) + 2OH⁻(aq) → Cu(OH)₂(s)
*Example (Barium Sulfate):*
Ba²⁺(aq) + SO₄²⁻(aq) → BaSO₄(s)
*Example (Silver Chloride):*
Ag⁺(aq) + Cl⁻(aq) → AgCl(s)
Ensure you include state symbols: (aq) for the ions and (s) for the precipitate.