Separate chemistry 2 — Edexcel GCSE Study Guide
Exam Board: Edexcel | Level: GCSE
Master the fundamentals of addition and condensation polymerisation, a high-yield topic that appears in almost every Separate Chemistry exam. Learn to draw repeating units, distinguish between synthetic and natural polymers, and evaluate the environmental impacts of plastic disposal to secure top marks.

## Overview
Welcome to Topic 9: Polymers. This is a core component of the Separate Chemistry specification and a topic where examiners frequently set high-mark, synoptic questions. Understanding polymers is not just about memorising chemical structures; it's about connecting molecular-level changes to macro-level properties and environmental consequences.
Polymers are ubiquitous in modern life, from the DNA in your cells to the plastic bottle on your desk. In the exam, you will be tested on your ability to distinguish between the two fundamental mechanisms of polymer formation: addition and condensation. You will also need to demonstrate spatial reasoning by drawing monomers from polymers and vice versa. Finally, this topic provides a classic context for 'evaluate' questions, requiring you to weigh the economic and environmental impacts of different disposal methods.
Listen to the audio guide below for a comprehensive walk-through of the key concepts:

## Key Concepts
### Concept 1: What is a Polymer?
A polymer is defined as a substance with a high average relative molecular mass, consisting of many small repeating units. These small units are called monomers. The process of linking these monomers together is called polymerisation.
Examiners often award marks for the precise use of terminology. Ensure you use the term 'repeating unit' rather than 'molecule' when referring to the segment inside the brackets of a polymer structure.
### Concept 2: Addition Polymerisation
Addition polymerisation occurs when monomers containing a carbon-carbon double bond (C=C) join together. During this process, the double bond opens up, allowing the carbon atoms to form single bonds with adjacent monomers, creating a long chain.
Crucially, in addition polymerisation, the polymer is the *only* product. No atoms are lost; all atoms present in the original monomers are incorporated into the polymer chain.
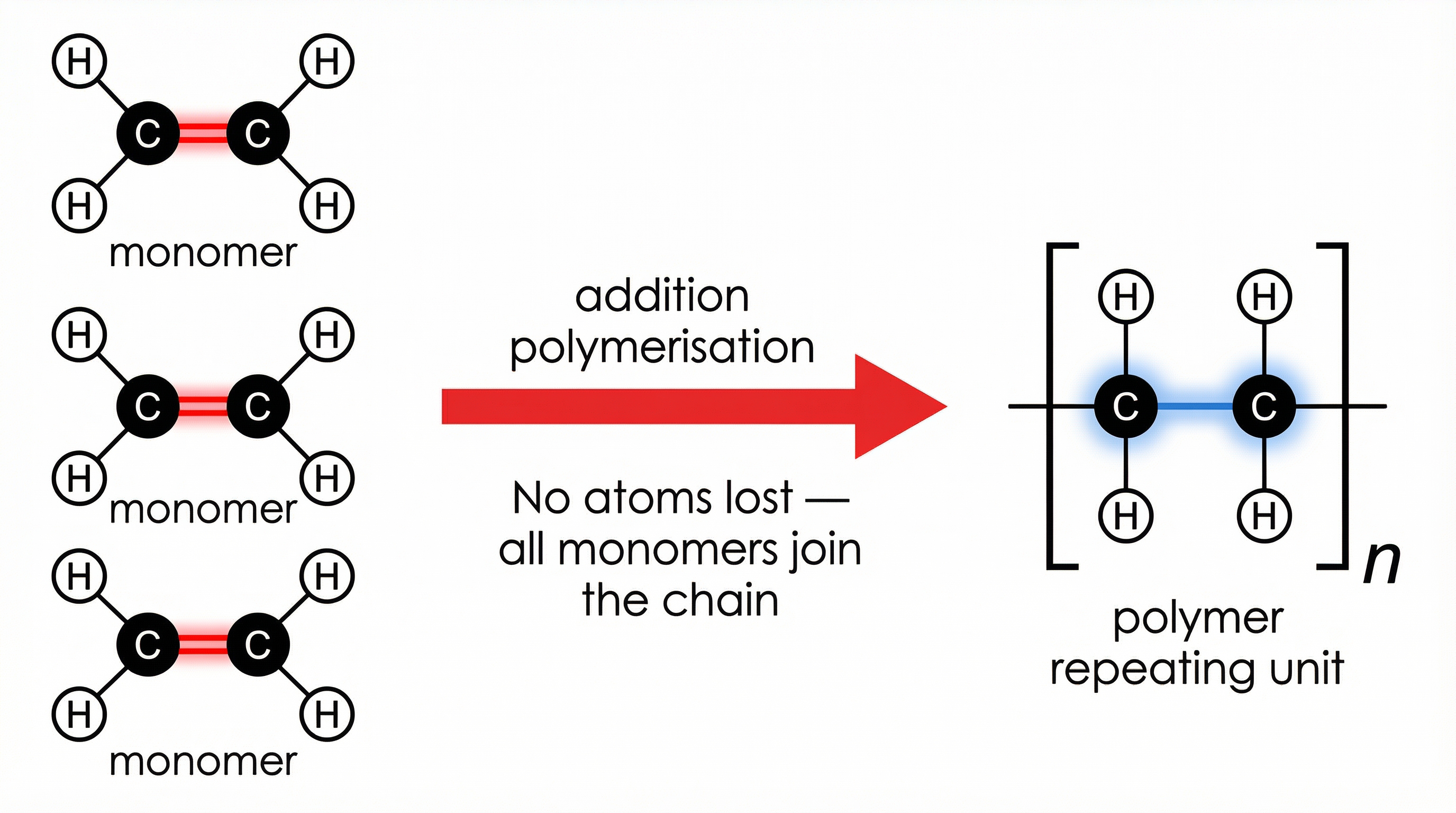
**Example**: The polymerisation of ethene (C₂H₄) forms poly(ethene). The monomer ethene has a C=C double bond. In poly(ethene), the repeating unit has only C-C single bonds. The naming convention is straightforward: place the name of the monomer in brackets and add the prefix 'poly'.
### Concept 3: Condensation Polymerisation
Condensation polymerisation involves monomers with two functional groups. When these monomers react, they join together, usually losing small molecules such as water. This is why the process is called a condensation reaction.
Unlike addition polymerisation, which typically involves one type of monomer with a C=C bond, condensation polymerisation often involves two different types of monomers, each with two functional groups at either end of the molecule.
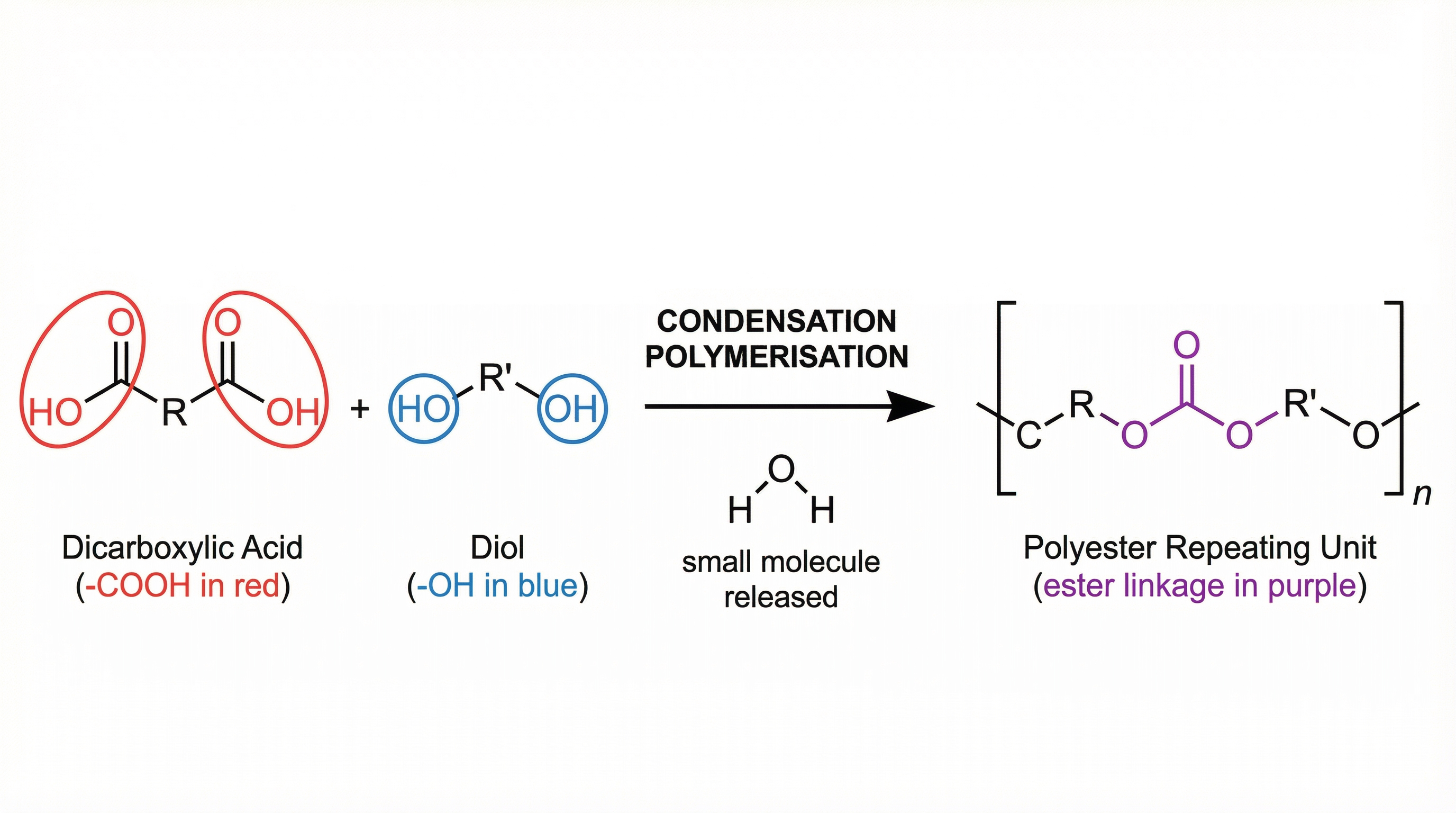
**Example: Formation of Polyesters**
Polyesters are formed from the condensation polymerisation of a dicarboxylic acid (a molecule with two -COOH groups) and a diol (a molecule with two -OH groups). When the carboxyl group of the acid reacts with the hydroxyl group of the diol, an ester link (-COO-) is formed, and a molecule of water (H₂O) is eliminated.
### Concept 4: Natural Polymers
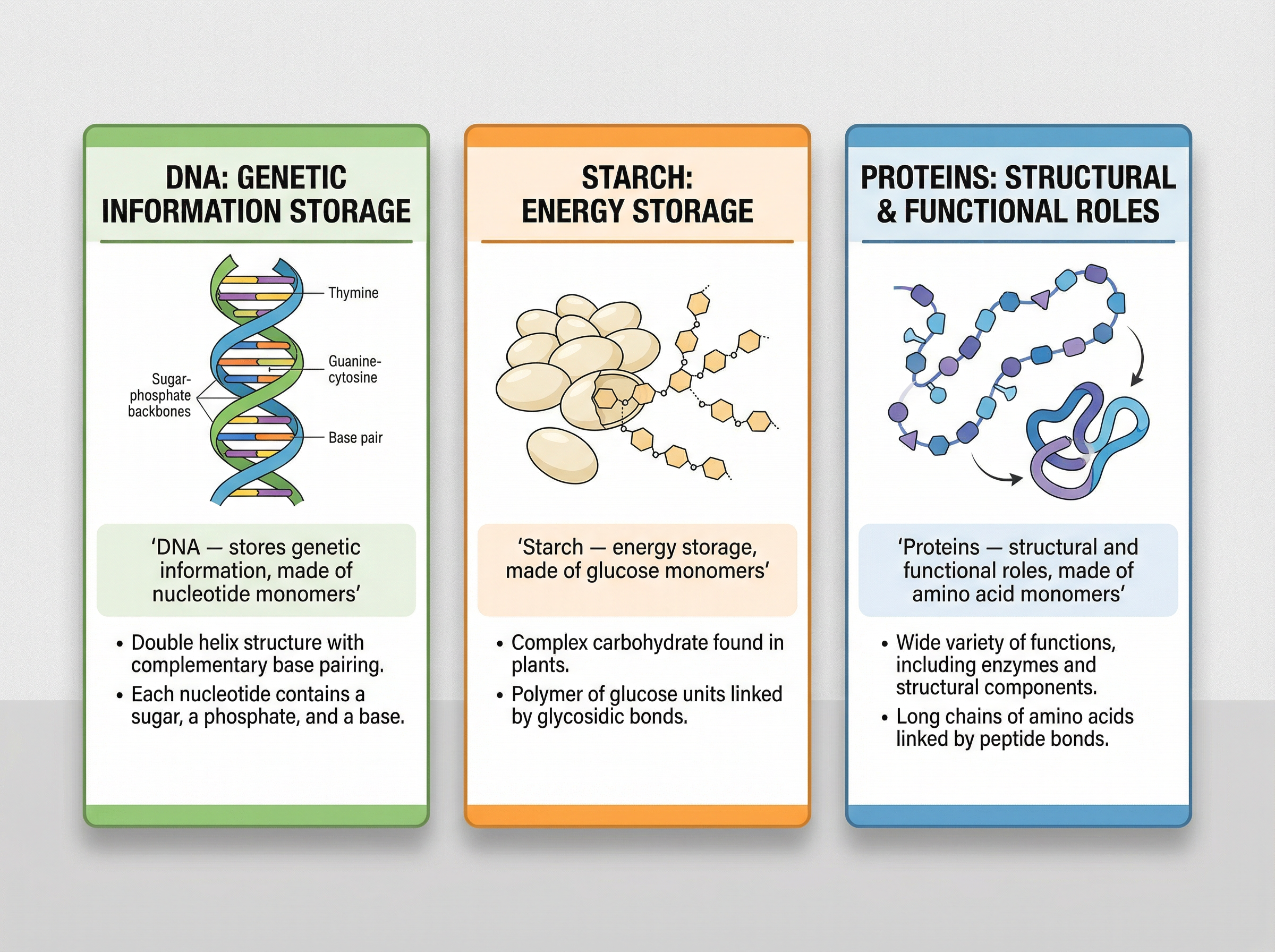
Not all polymers are synthetic. Nature has been utilising polymerisation for billions of years. You must be able to identify the following natural polymers and their corresponding monomers:
1. **DNA (Deoxyribonucleic acid)**: A crucial polymer essential for life. It consists of two polymer chains made from monomers called nucleotides, forming a double helix structure.
2. **Proteins**: Formed from amino acid monomers via condensation polymerisation. Proteins have diverse structural and functional roles in living organisms.
3. **Starch and Cellulose**: Both are polymers of glucose monomers. They are carbohydrates; starch acts as an energy store, while cellulose provides structural support in plant cell walls.
### Concept 5: Environmental and Economic Impacts
The widespread use of synthetic polymers, particularly plastics, presents significant environmental challenges. Most addition polymers are unreactive and non-biodegradable, meaning they persist in the environment for hundreds of years, accumulating in landfill sites and oceans.
Examiners frequently ask candidates to evaluate the methods of disposing of polymers. You must be able to discuss the advantages and disadvantages of:
* **Landfill**: Cheap and easy, but requires space and materials do not degrade.
* **Combustion (Incineration)**: Releases energy, but produces greenhouse gases (CO₂) and potentially toxic gases (e.g., HCl from PVC).
* **Recycling**: Conserves finite crude oil resources and reduces landfill waste, but requires sorting (which is expensive and labour-intensive) and the quality of the plastic may degrade over time.
## Mathematical/Scientific Relationships
While there are no complex mathematical formulas in this specific topic, you must master the structural representation of polymers:
* **Drawing the repeating unit**: The repeating unit must be drawn inside square brackets, with the bonds extending *through* the brackets to indicate the continuous chain. A subscript 'n' is placed outside the bracket to represent a large number of repeating units.
* **Finding the monomer**: To find the monomer from an addition polymer, isolate the repeating unit, remove the extension bonds, and insert a C=C double bond between the two carbon atoms.
## Practical Applications
Understanding polymerisation is vital in materials science. The properties of a polymer (e.g., flexibility, melting point) depend on the monomers used and the conditions under which it was made. For instance, poly(ethene) can be manufactured as low-density poly(ethene) (LDPE) for flexible carrier bags, or high-density poly(ethene) (HDPE) for rigid plastic bottles, simply by altering the reaction conditions (temperature, pressure, and catalyst).