States of matter and mixtures — Edexcel GCSE Study Guide
Exam Board: Edexcel | Level: GCSE
Master the fundamentals of how particles behave in solids, liquids, and gases, and learn how to separate complex mixtures into pure substances. This topic is heavily tested in exams, particularly your ability to choose and justify separation techniques and calculate Rf values.
## Overview
Welcome to one of the most foundational topics in your GCSE Chemistry specification: **States of Matter and Mixtures**. This topic explores the microscopic world of particles and how their arrangement, movement, and energy determine the physical properties of everything around us. It's a critical topic because these concepts form the building blocks for almost everything else you'll study in Chemistry, from chemical bonding to rates of reaction.
Examiners love this topic because it allows them to test your understanding of models (like the particle model) and your practical skills (like choosing the right separation technique). You'll frequently encounter questions asking you to describe particle behaviour, interpret melting point graphs to determine purity, or calculate Rf values from chromatograms. By mastering this topic, you'll secure marks that many candidates lose through imprecise language.
## Key Concepts
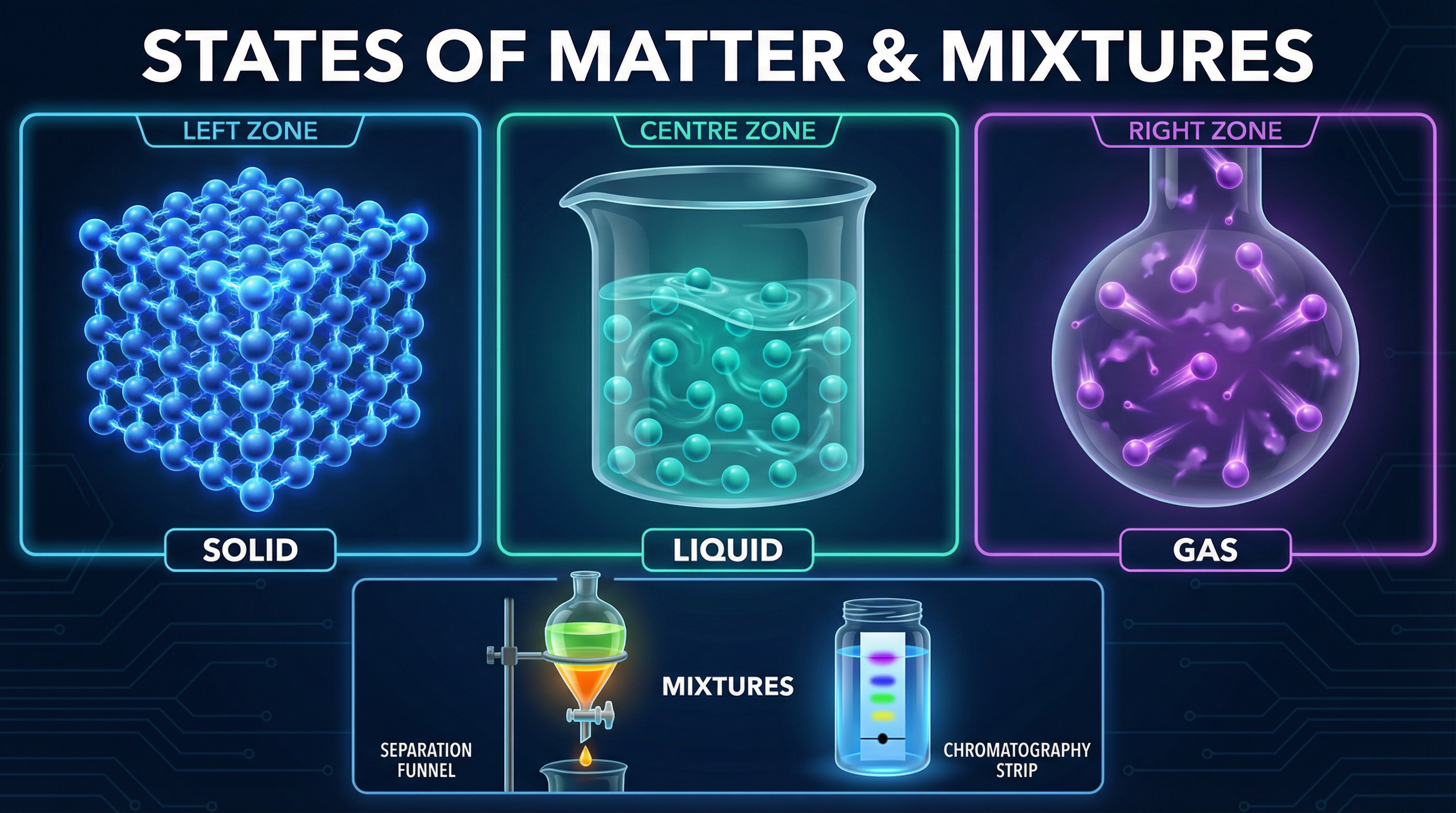
### Concept 1: The Three States of Matter
All matter is made of particles (atoms, molecules, or ions). The behaviour of these particles determines whether a substance is a solid, a liquid, or a gas. Examiners require you to describe these states using three specific criteria: **arrangement**, **movement**, and **energy**.
* **Solids**: Particles are arranged in a regular, closely packed lattice. They do not move from place to place but **vibrate in fixed positions**. They have the lowest energy of the three states.
* **Liquids**: Particles are still mostly touching but are randomly arranged. They can move around and flow past each other. They have more energy than in a solid.
* **Gases**: Particles are widely spaced and randomly arranged. They move rapidly in all directions. The forces of attraction between them are negligible (almost zero). They have the highest energy.
**Example**: When answering a 3-mark question to describe a solid, write: "Particles are in a regular arrangement (1 mark), vibrate in fixed positions (1 mark), and have low energy (1 mark)."
### Concept 2: Changes of State
Changes of state are physical changes, not chemical reactions. This is because no new substances are formed; the particles themselves remain exactly the same, only their arrangement and energy change. State changes can be reversed by heating or cooling.
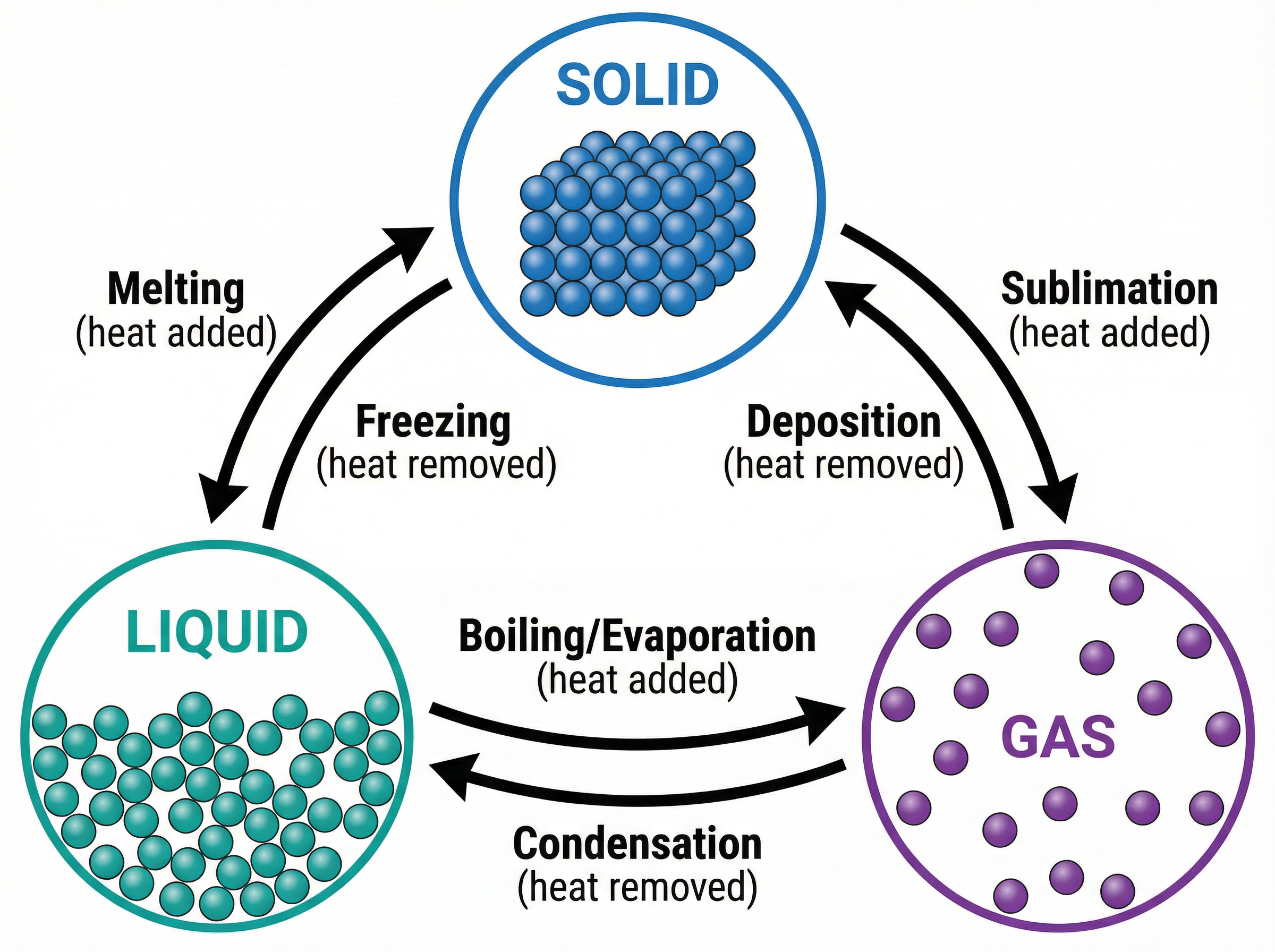
When a substance is heated, its particles gain kinetic energy. For example, during **melting** (solid to liquid), the particles vibrate more vigorously until they have enough energy to overcome the forces holding them in their fixed positions. Conversely, during **condensation** (gas to liquid), particles lose energy and move more slowly until the forces of attraction pull them close together again.
* **Melting**: Solid $\rightarrow$ Liquid
* **Freezing**: Liquid $\rightarrow$ Solid
* **Boiling / Evaporation**: Liquid $\rightarrow$ Gas
* **Condensation**: Gas $\rightarrow$ Liquid
* **Sublimation**: Solid $\rightarrow$ Gas (direct, e.g., iodine or dry ice)
* **Deposition**: Gas $\rightarrow$ Solid
### Concept 3: Pure Substances vs Mixtures
In everyday language, "pure" might mean clean or natural. In chemistry, a **pure substance** contains only one type of element or one type of compound. A **mixture** consists of two or more elements or compounds that are not chemically combined together. The chemical properties of each substance in the mixture are unchanged.
Examiners frequently test your ability to distinguish between the two using melting point data. A pure substance will melt at a **sharp, specific temperature** (e.g., pure water melts exactly at 0°C). A mixture will melt over a **range of temperatures**.
### Concept 4: Separation Techniques
Because the components of a mixture are not chemically combined, they can be separated using physical processes. You must be able to select the correct technique based on the properties of the mixture.
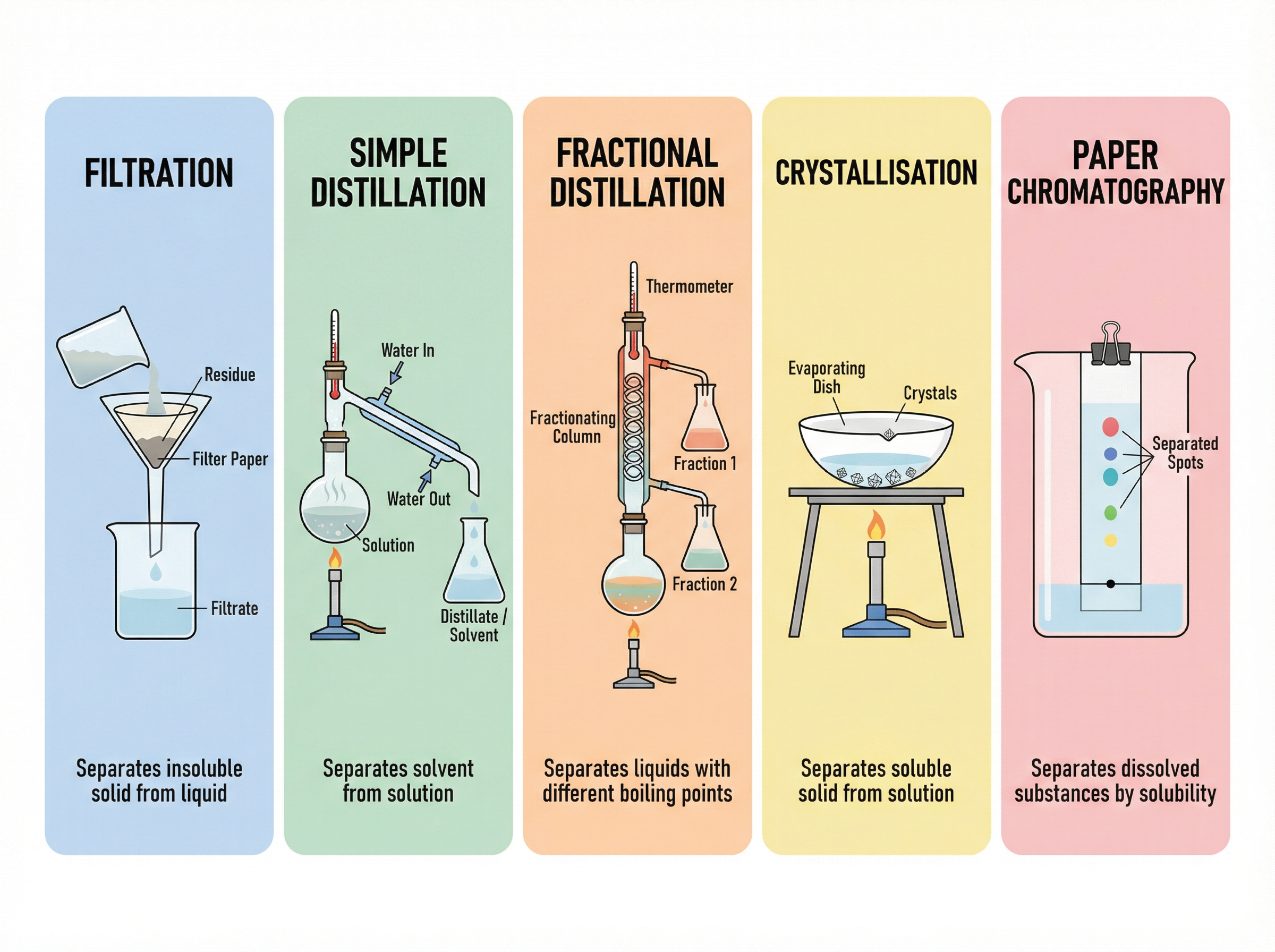
* **Filtration**: Separates an insoluble solid from a liquid (e.g., sand from water).
* **Crystallisation**: Separates a soluble solid from a solution (e.g., obtaining copper sulfate crystals from copper sulfate solution).
* **Simple Distillation**: Separates a liquid (solvent) from a solution (e.g., obtaining pure water from salt water). It relies on a significant difference in boiling points.
* **Fractional Distillation**: Separates a mixture of liquids with different boiling points (e.g., separating crude oil or a mixture of ethanol and water).
* **Paper Chromatography**: Separates mixtures of dissolved substances (e.g., different dyes in an ink). It relies on differences in solubility in the mobile phase and attraction to the stationary phase.
### Concept 5: Making Water Potable
Potable water is water that is safe to drink. It is not necessarily "pure" water, as it still contains dissolved salts, but the levels of dissolved salts are low and it is free of harmful microbes. The process typically involves:
1. **Sedimentation**: Large solid particles settle to the bottom.
2. **Filtration**: Water is passed through beds of sand and gravel to remove smaller insoluble particles.
3. **Chlorination**: Chlorine gas is bubbled through to kill harmful bacteria and microbes.
If completely pure water is required (e.g., for chemical analysis in a lab), it must be obtained through distillation, which removes all dissolved ions that could interfere with chemical tests.

## Mathematical/Scientific Relationships
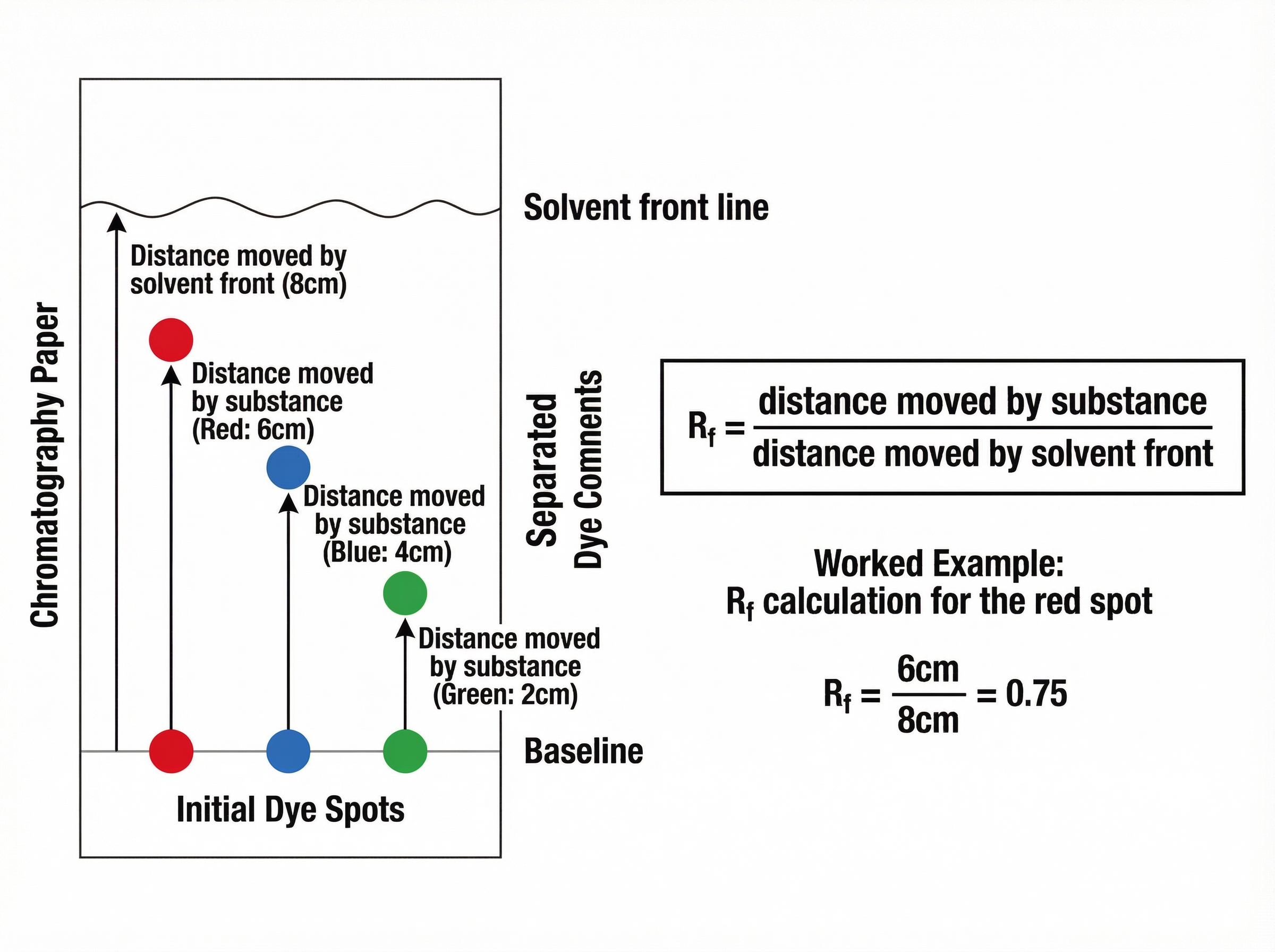
### Retention Factor ($R_f$)
In paper chromatography, the $R_f$ value is used to identify unknown substances by comparing them with known reference values. It is calculated using the formula:
$$R_f = \frac{\text{distance moved by substance}}{\text{distance moved by solvent front}}$$
* **Distance moved by substance**: Measured from the pencil baseline to the centre of the spot.
* **Distance moved by solvent front**: Measured from the pencil baseline to the line reached by the solvent.
* **Key Rule**: The $R_f$ value is a ratio and therefore has no units. It must **always be less than 1**. If your calculation gives a value greater than 1, you have divided the numbers the wrong way around.
## Practical Applications
**Required Practical: Chromatography**
* **Method**: Draw a pencil line (baseline) near the bottom of chromatography paper. Place a spot of the mixture on the line. Place the paper in a beaker with a small volume of solvent (e.g., water or ethanol). The solvent level must be **below** the pencil line. Put a lid on the beaker to prevent solvent evaporation. Wait for the solvent to travel near the top, then remove the paper and mark the solvent front.
* **Common Examiner Question**: "Why must the baseline be drawn in pencil?"
* *Answer*: Because pencil lead (graphite) is insoluble in the solvent and will not run up the paper and interfere with the results. Pen ink would dissolve and separate.
* **Common Examiner Question**: "Why must the solvent level be below the baseline?"
* *Answer*: So that the spots of mixture do not simply dissolve away into the bulk of the solvent in the beaker.