States of matter and mixtures — Edexcel GCSE Study Guide
Exam Board: Edexcel | Level: GCSE
Master the difference between pure substances and mixtures, and discover how chemists use physical properties to separate them. This topic is foundational for your GCSE exams, featuring heavily in practical questions and calculations like Rf values.
## Overview
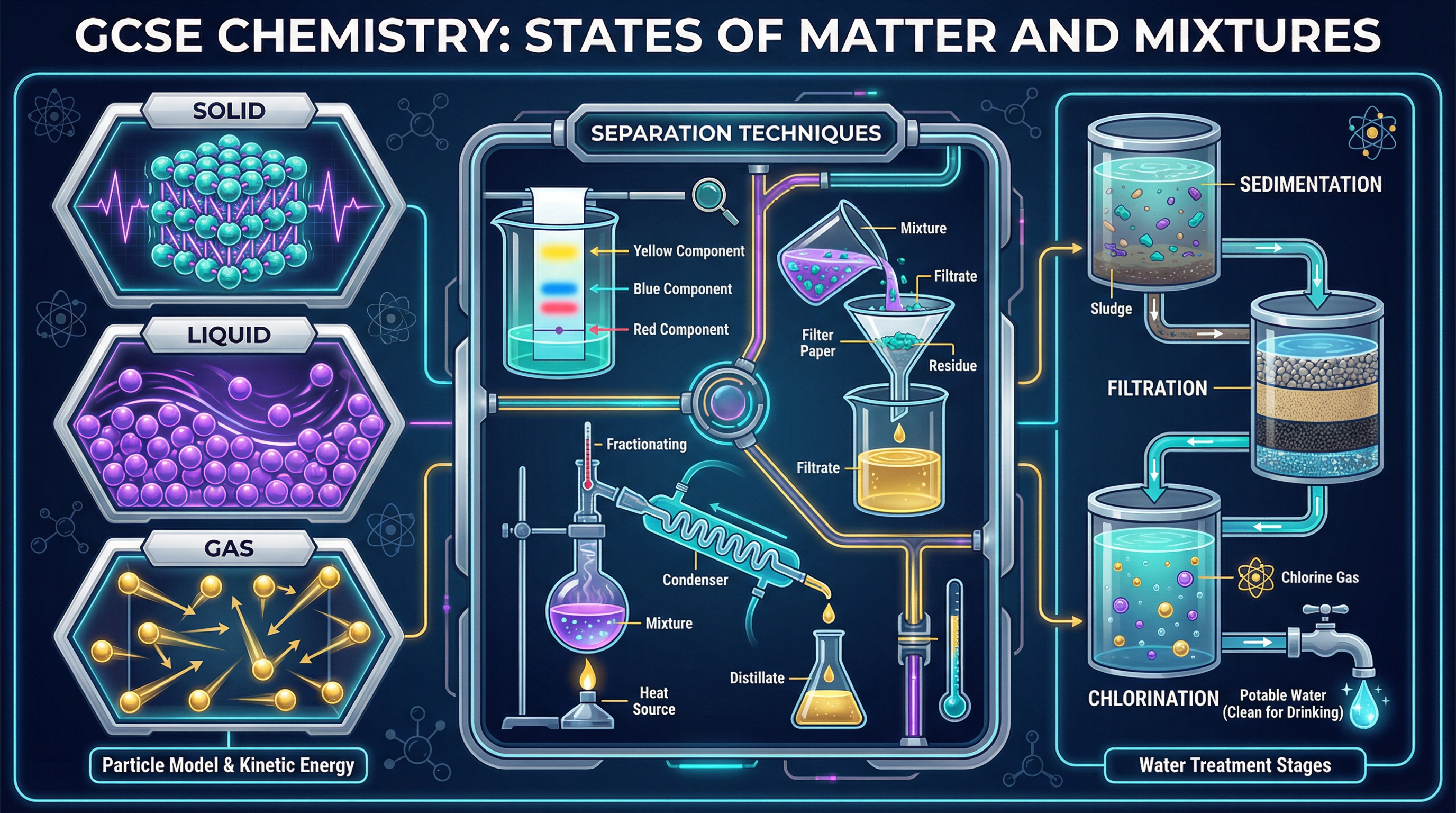
Welcome to one of the most practically important topics in your GCSE Chemistry specification: **States of Matter and Mixtures**. In everyday life, we might describe orange juice as 'pure', but a chemist would strongly disagree! This topic explores the precise scientific definition of purity and how we can use physical properties—like melting points and boiling points—to identify substances.
You will also master the four core separation techniques: **filtration**, **crystallisation**, **distillation**, and **paper chromatography**. These are not just theoretical concepts; they are essential laboratory skills that examiners frequently test through practical-based questions and data analysis. Understanding how to separate mixtures is crucial for everything from forensic science to providing safe drinking water for millions of people.
By the end of this guide, you will be able to interpret melting point data, calculate Rf values from chromatograms, and explain the stages of water purification with examiner-level precision.
## Key Concepts
### Concept 1: Pure Substances vs Mixtures
In chemistry, a **pure substance** contains only one type of element or compound. It is not mixed with anything else. A **mixture** consists of two or more elements or compounds that are not chemically combined together.
Examiners test your understanding of this by asking about melting and boiling points. A pure substance has a **sharp, specific melting and boiling point**. For example, pure water always boils at exactly 100°C and melts at exactly 0°C at standard pressure.
In contrast, mixtures melt and boil over a **range of temperatures**. Impurities lower the melting point and increase the boiling point of a substance.
**Example**: If a student tests a sample of water and finds it starts melting at -2°C and finishes at 1°C, the sample is a mixture, not pure water. The impurities disrupt the regular lattice structure of the solid, meaning less energy is required to break the intermolecular forces.
### Concept 2: Separation Techniques
Chemists use physical processes to separate mixtures without making new chemical substances. The technique you choose depends entirely on the physical properties of the mixture.
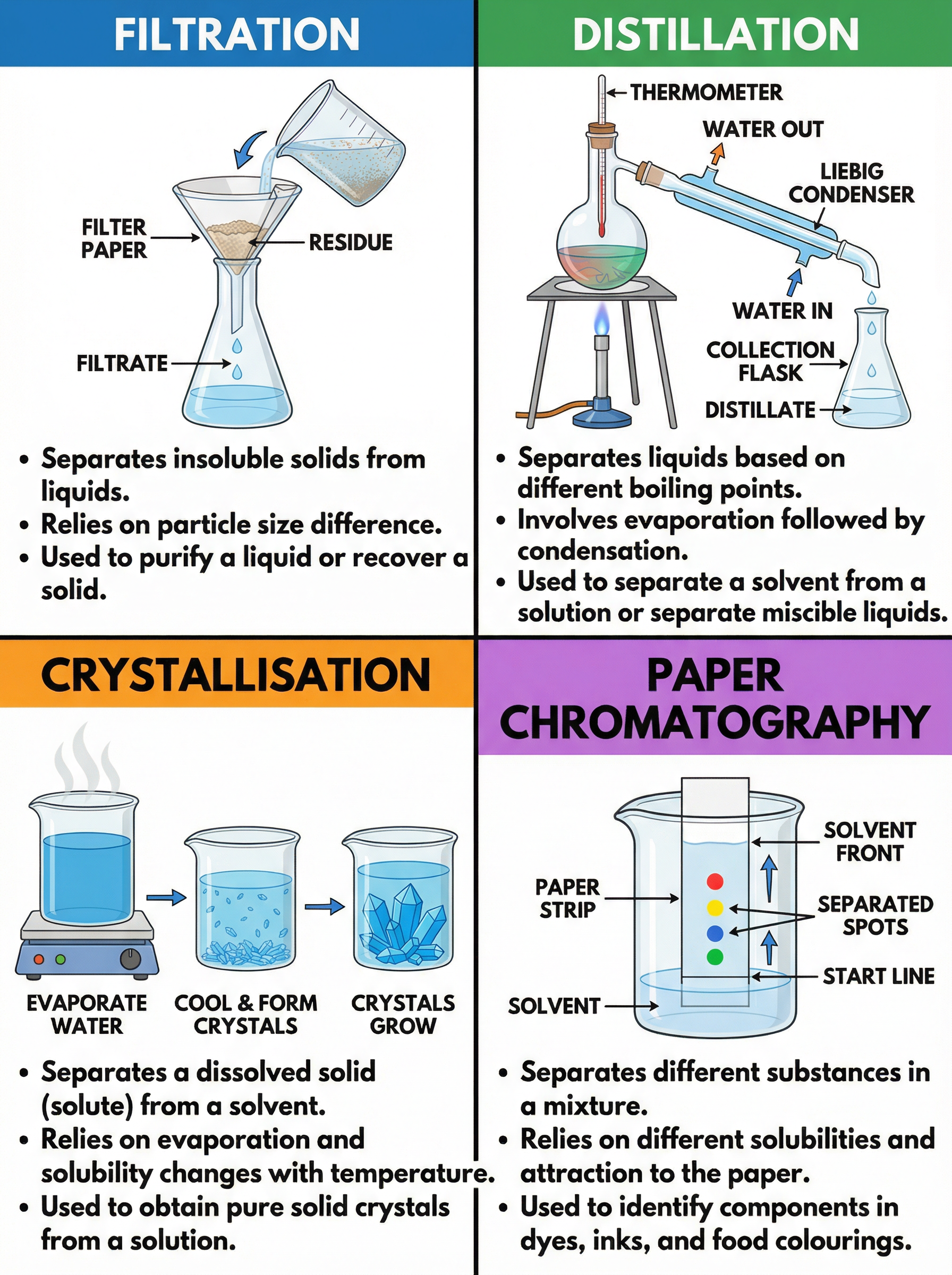
1. **Filtration**: Used to separate an insoluble solid from a liquid (e.g., sand and water). The mixture is poured through filter paper in a funnel. The liquid that passes through is the **filtrate**, and the solid left behind is the **residue**.
2. **Crystallisation**: Used to obtain a soluble solid from a solution (e.g., salt from saltwater). The solution is heated gently to evaporate some solvent, creating a saturated solution. As it cools, the solubility decreases, and solid crystals form.
3. **Distillation**: Used to separate a solvent from a solution or a mixture of miscible liquids (e.g., pure water from seawater). The mixture is heated until the substance with the lowest boiling point evaporates. The vapour then passes through a condenser, where it cools and condenses back into a liquid (the **distillate**).
4. **Paper Chromatography**: Used to separate mixtures of soluble substances, such as food colourings or inks.
### Concept 3: Paper Chromatography in Detail
Chromatography relies on two phases:
- The **stationary phase**: The paper itself, which does not move.
- The **mobile phase**: The solvent (e.g., water or ethanol), which moves up the paper by capillary action.
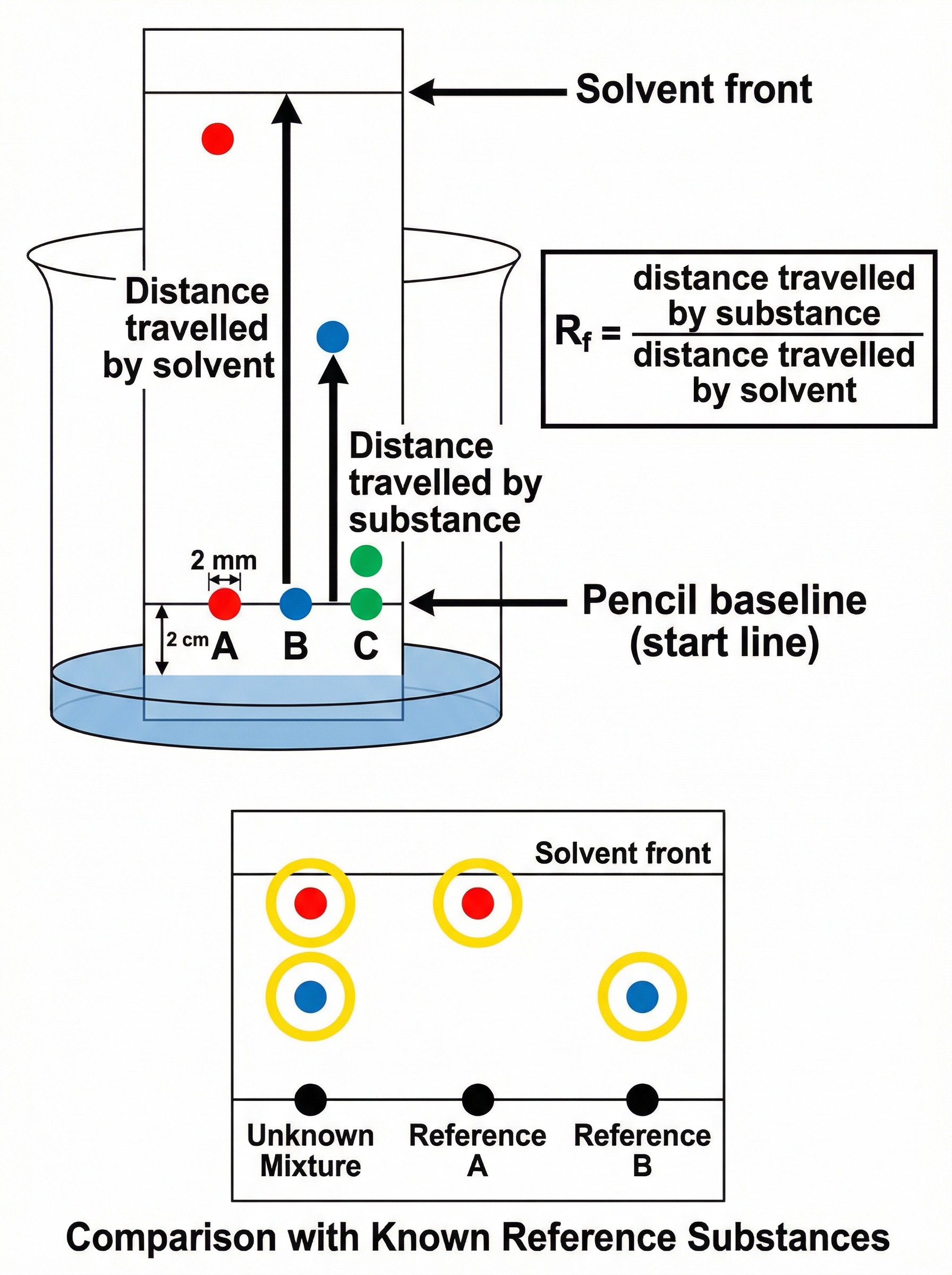
Substances separate because they have different solubilities in the mobile phase and different attractions to the stationary phase. A substance that is highly soluble in the solvent will travel further up the paper than a less soluble substance.
**Examiner Tip**: Always draw the start line (baseline) in **pencil**. Pen ink would dissolve in the solvent and interfere with your results.
### Concept 4: Water Purification
Providing potable (safe to drink) water involves removing solid particles and killing harmful microbes. The standard UK process has three stages:
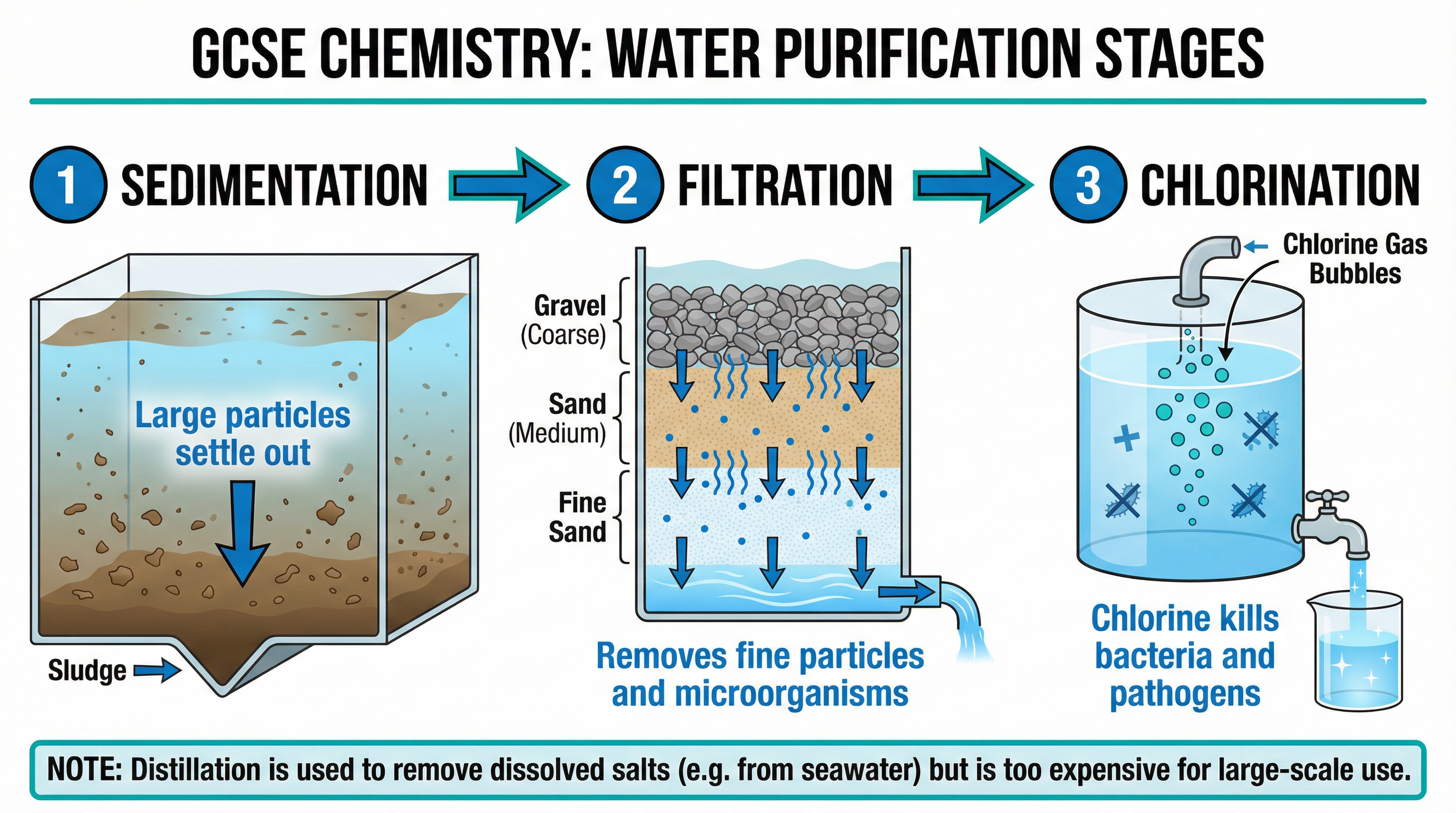
1. **Sedimentation**: Water sits in large tanks. Large insoluble particles settle to the bottom under gravity.
2. **Filtration**: Water passes through beds of sand and gravel to remove smaller insoluble particles.
3. **Chlorination**: Chlorine gas is added to kill bacteria and other harmful microorganisms.
If water contains high levels of dissolved salts (like seawater), it must be purified by **desalination** using distillation or reverse osmosis. However, these processes require huge amounts of energy, making them expensive.
## Mathematical/Scientific Relationships
### The Rf Value (Retention Factor)
In chromatography, we calculate the Rf value to help identify unknown substances.
**Formula**:
Rf = distance travelled by substance ÷ distance travelled by solvent
- Both distances must be measured from the pencil baseline.
- The Rf value has **no units**.
- The Rf value is always between 0 and 1.
## Podcast Episode
Listen to our 10-minute revision podcast covering all these concepts, common exam mistakes, and a quick-fire quiz!

## Practical Applications
Chromatography is widely used in forensic science to analyse ink samples from forged documents, or in sports medicine to test athletes' blood and urine for banned performance-enhancing drugs.