Topic 7 is a practical-based topic — OCR GCSE Study Guide
Exam Board: OCR | Level: GCSE
Topic 7 is the ultimate toolkit for any aspiring chemist, transforming theoretical knowledge into hands-on practical skills. It covers essential techniques from safe handling to precise measurements and complex separation methods, directly contributing to 15% of your final exam grade.
## Overview

Welcome to Topic 7: Practical Skills! This topic is the heartbeat of Chemistry. It is where the theory you learn in the classroom comes to life on the laboratory bench. This topic is designed to equip you with the essential skills required for scientific enquiry, ensuring you can safely use apparatus, perform intricate techniques, and record accurate measurements.
Why does this matter? Because the skills you develop here are not just for the lab—they are directly assessed in your written examinations, accounting for 15% of your total marks. Examiners will test your ability to describe, explain, and evaluate experimental procedures. You will need to draw upon your knowledge of the eight Practical Activity Groups (PAGs) and apply it to new contexts.

## Key Concepts
### Concept 1: Safe Handling and Risk Assessment
Safety is the paramount concern in any laboratory. You must be able to identify hazards associated with specific chemicals and apparatus, and state appropriate precautions. A hazard is the potential for harm (e.g., concentrated acid is corrosive), while the risk is the likelihood of that harm occurring. Examiners look for specific links between the hazard and the precaution.
**Example**: When using toxic chlorine gas, the hazard is inhalation causing respiratory damage. The precaution is to carry out the experiment in a working fume cupboard.
### Concept 2: Accurate Measurement
Precision and accuracy in measurement are fundamental. You must select the correct apparatus for the task and read it correctly. For example, when measuring the volume of a liquid, a burette or pipette offers much higher precision than a measuring cylinder. Crucially, when reading a volume, you must read from the bottom of the meniscus (the curve of the liquid surface) at eye level to avoid parallax error.
**Example**: Recording a mass of 2.50 g on a two-decimal-place balance, rather than just 2.5 g, demonstrates appropriate precision.
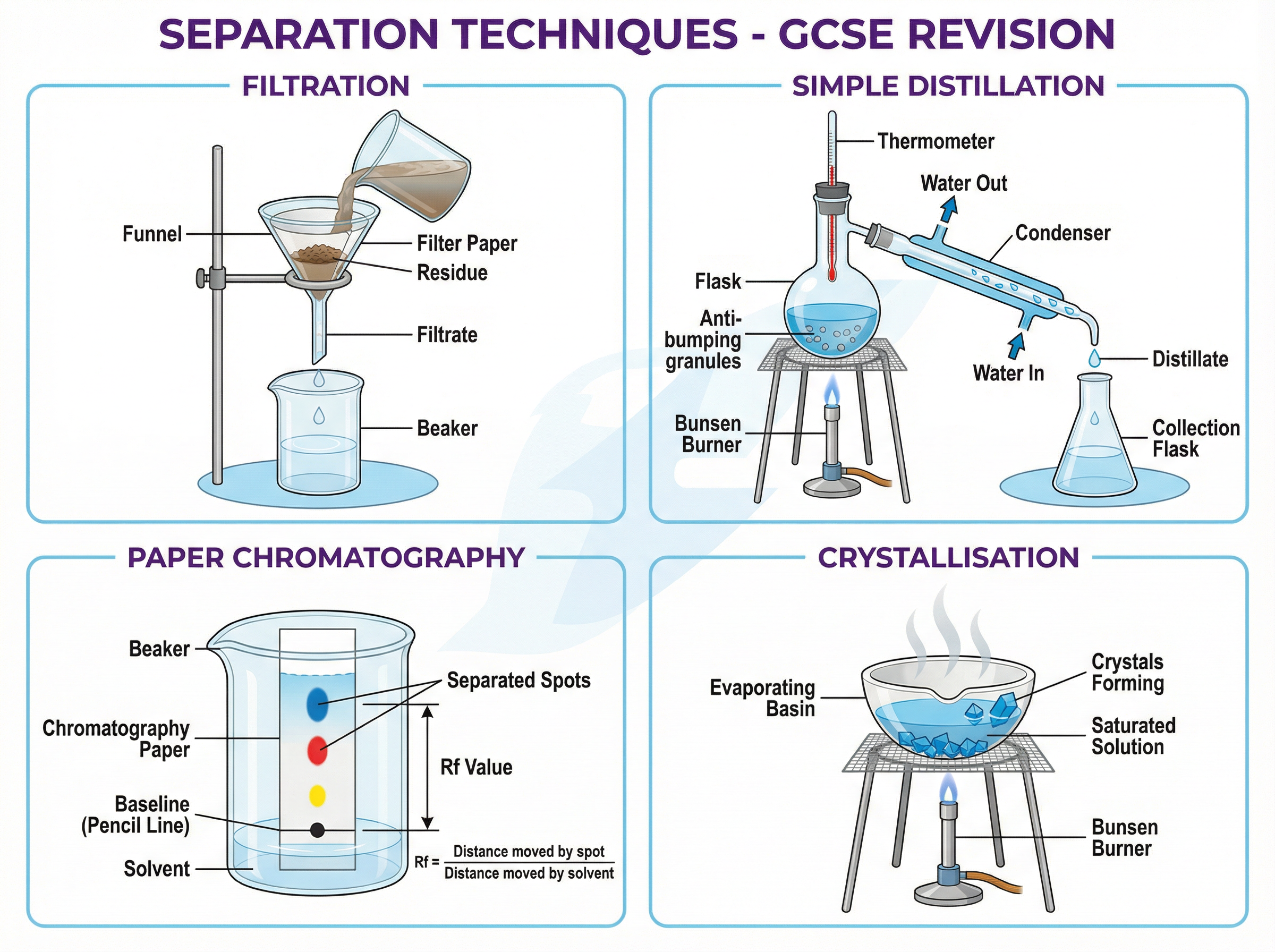
### Concept 3: Separation Techniques
Chemistry often involves isolating pure substances from mixtures. You must master four core techniques:
1. **Filtration**: Separates an insoluble solid from a liquid (e.g., sand from water).
2. **Crystallisation**: Obtains a soluble solid from a solution by evaporating the solvent.
3. **Simple Distillation**: Separates a liquid from a solution, relying on different boiling points.
4. **Chromatography**: Separates mixtures of soluble substances (like inks) based on their relative affinities for the stationary phase (paper) and mobile phase (solvent).
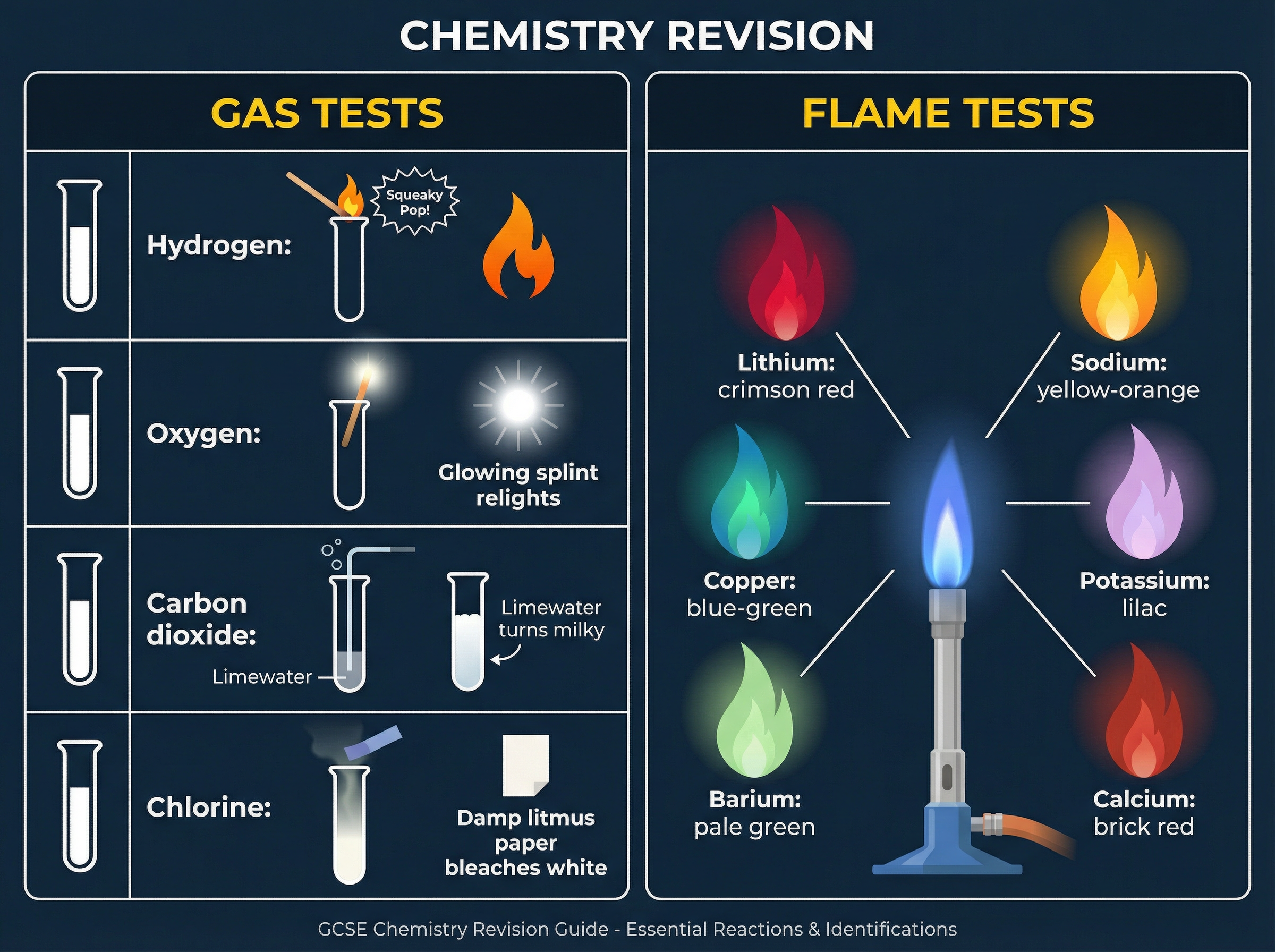
### Concept 4: Identification Tests
Chemists act as detectives, using specific reactions to identify unknown substances. You must memorize the standard tests for common gases (hydrogen, oxygen, carbon dioxide, chlorine) and the flame test colours for metal ions. These are high-frequency recall questions in exams.
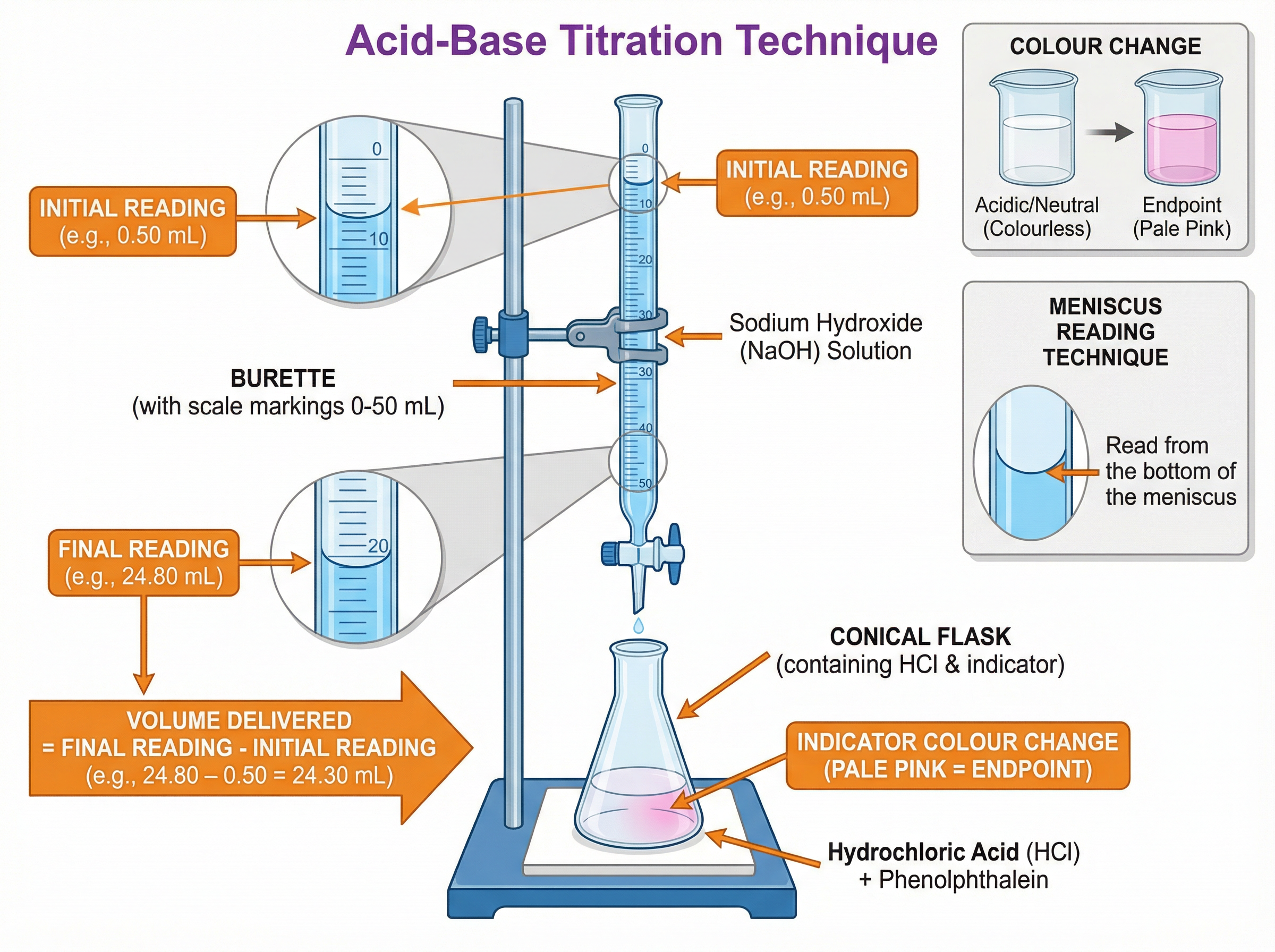
### Concept 5: Titration
Titration is a highly precise quantitative technique used to determine the concentration of an unknown solution by reacting it with a solution of known concentration. It requires meticulous technique, including the use of a volumetric pipette, a burette, and a suitable indicator (like phenolphthalein) to identify the exact endpoint of the reaction.
## Mathematical/Scientific Relationships
* **Rf Value (Chromatography)**: $R_f = \frac{\text{distance moved by substance}}{\text{distance moved by solvent}}$. This value is always between 0 and 1 and has no units. It helps identify substances by comparing them to known standards.
* **Titration Calculations**: $n = c \times V$, where $n$ is the number of moles, $c$ is the concentration (in mol/dm³), and $V$ is the volume (in dm³). Remember to convert volumes from cm³ to dm³ by dividing by 1000.
## Practical Applications
The skills learned here are applied across all scientific disciplines and industries. From pharmaceutical companies synthesising and purifying new drugs, to environmental agencies monitoring water quality using titration and chromatography, these practical techniques are the foundation of real-world scientific progress.