Topic C1: Particles — OCR GCSE Study Guide
Exam Board: OCR | Level: GCSE
Master the fundamentals of GCSE Chemistry with this comprehensive guide to particles and atomic structure. Discover how the particle model explains the states of matter, and trace the fascinating historical journey that led to our modern understanding of the atom.

## Overview
Welcome to Topic C1: Particles, the foundation of your entire GCSE Chemistry course. Whether you're exploring the properties of materials, balancing chemical equations, or understanding rates of reaction, it all starts here. This topic explores the particle model, explaining how the arrangement and movement of particles dictate whether a substance is a solid, liquid, or gas. You'll learn to distinguish between physical and chemical changes at a microscopic level.
Beyond states of matter, we dive into the very heart of the atom, examining its sub-atomic structure—protons, neutrons, and electrons. We will also trace the fascinating historical development of the atomic model, from Dalton's solid spheres to Bohr's energy levels. Examiners frequently test this topic through multiple-choice questions on atomic structure, calculations of sub-atomic particles, and longer 6-mark questions requiring you to explain changes of state or the evolution of atomic theories.

## Key Concepts
### Concept 1: The Particle Model and States of Matter
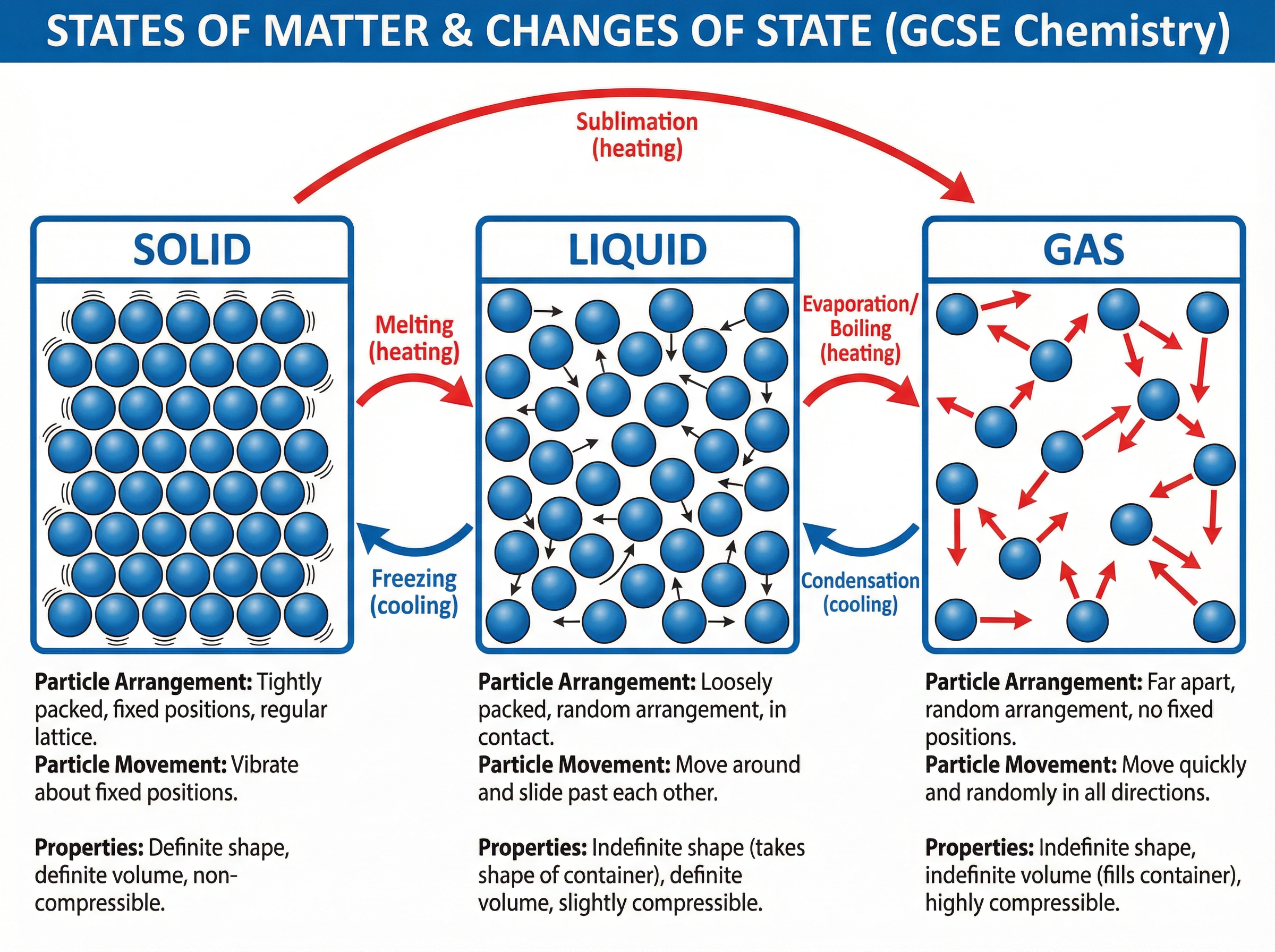
The particle model is a scientific theory stating that all matter is composed of tiny, indivisible particles in constant motion. The arrangement, energy, and forces of attraction between these particles determine the state of matter: solid, liquid, or gas.
* **Solids:** Particles are tightly packed in a regular, ordered lattice structure. They vibrate about fixed positions due to strong forces of attraction. This explains why solids have a definite shape, a fixed volume, and cannot be compressed.
* **Liquids:** Particles are close together but arranged randomly. They have sufficient energy to overcome some attractive forces, allowing them to move around and slide past one another. Consequently, liquids take the shape of their container, maintain a fixed volume, and are only very slightly compressible.
* **Gases:** Particles are widely spaced and move rapidly in random directions. The forces of attraction are negligible. The large empty spaces between particles mean gases have no definite shape or volume (they fill their container) and are highly compressible.
**Crucial Exam Point:** When a substance is heated, it expands. The particles themselves **do not** get bigger; rather, they gain kinetic energy, move faster, and the average distance between them increases.
### Concept 2: Changes of State
Changes of state occur when energy is transferred to or from a substance, altering the arrangement and movement of its particles. These are **physical changes**, meaning no new substances are formed and the process is reversible.
* **Melting (Solid to Liquid):** Heating transfers energy to the particles, causing them to vibrate more vigorously until they overcome the forces holding them in fixed positions.
* **Freezing (Liquid to Solid):** Cooling removes energy. Particles slow down and strong attractive forces pull them back into a regular lattice.
* **Boiling/Evaporation (Liquid to Gas):** Heating provides enough energy for particles to completely overcome attractive forces and escape as a gas.
* **Condensation (Gas to Liquid):** Cooling causes gas particles to lose energy, slow down, and clump together to form a liquid.
* **Sublimation (Solid to Gas):** Some substances, like solid carbon dioxide (dry ice), change directly from a solid to a gas when heated.
### Concept 3: Physical vs. Chemical Changes
Examiners frequently ask candidates to distinguish between physical and chemical changes using the particle model.
* **Physical Change:** The particles simply rearrange or change their spacing. The chemical identity of the substance remains identical. **No new substances are formed**, and the change is usually easily reversible (e.g., melting ice, dissolving sugar).
* **Chemical Change:** The atoms within the particles rearrange to form entirely **new substances** with different properties. These changes are typically irreversible (e.g., burning magnesium, rusting iron).
### Concept 4: Atomic Structure and Sub-atomic Particles
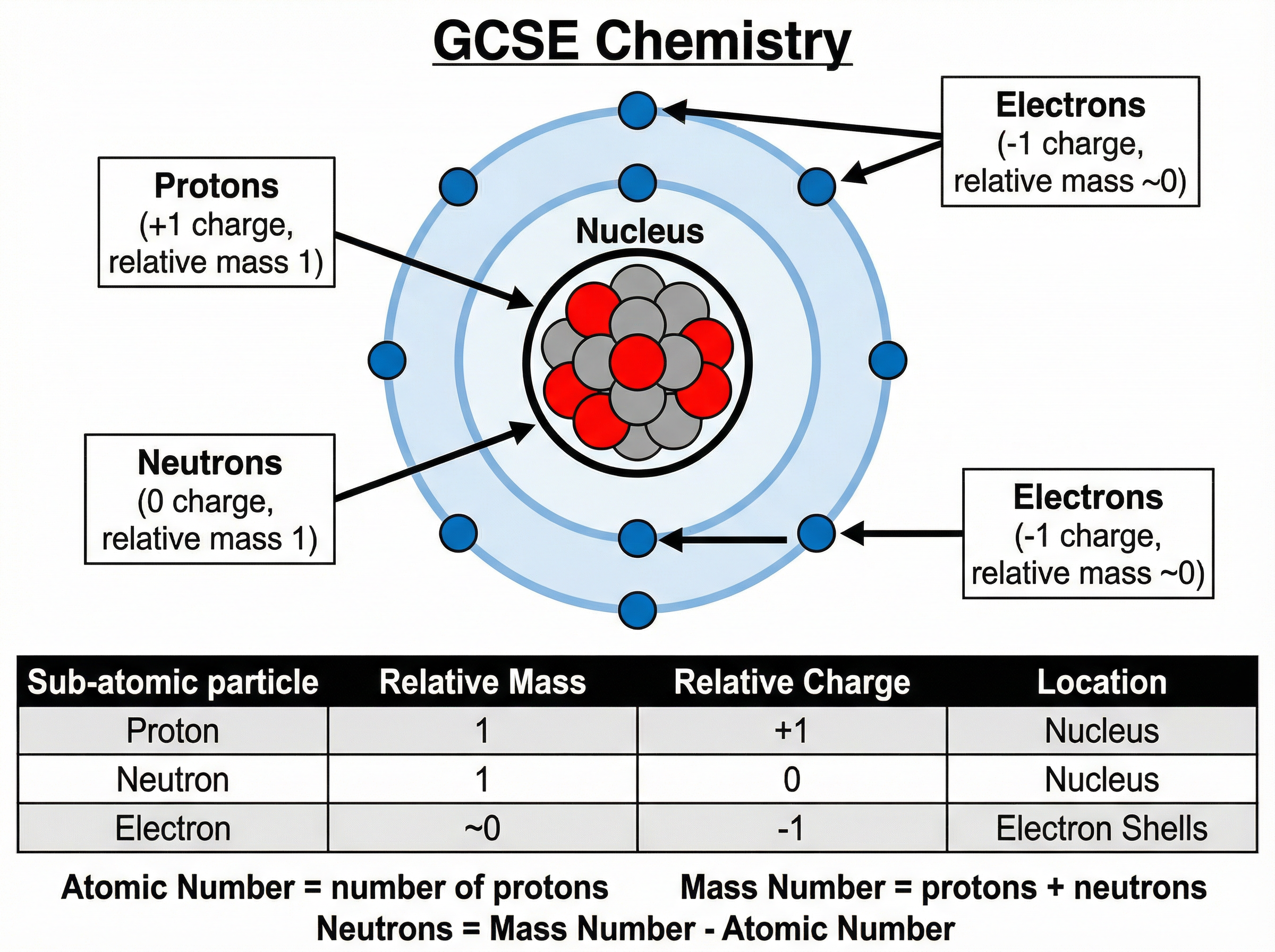
Atoms are the building blocks of all matter. They consist of a central nucleus surrounded by orbiting electrons.
* **Nucleus:** The tiny, dense centre of the atom containing protons and neutrons. It holds almost all the mass of the atom and has a positive charge.
* **Electron Shells:** Energy levels where electrons orbit the nucleus at high speeds. Most of the atom is empty space.
You must memorise the relative masses and charges of the sub-atomic particles:
* **Proton:** Relative Mass = 1, Relative Charge = +1
* **Neutron:** Relative Mass = 1, Relative Charge = 0
* **Electron:** Relative Mass = ~0 (very small, approx. 1/1836), Relative Charge = -1
In a neutral atom, the number of positive protons equals the number of negative electrons, so the overall charge cancels out.
### Concept 5: Isotopes and Ions
**Isotopes** are atoms of the same element that possess the same number of protons (same atomic number) but a different number of neutrons (different mass number). They have identical chemical properties but slightly different physical properties. For example, Carbon-12 has 6 neutrons, while Carbon-14 has 8 neutrons.
**Ions** are charged particles formed when atoms gain or lose electrons to achieve a full outer shell.
* Losing electrons forms a positive ion (cation).
* Gaining electrons forms a negative ion (anion).
* **Crucial Exam Point:** The number of protons *never* changes when an ion is formed.
### Concept 6: The Historical Development of the Atomic Model
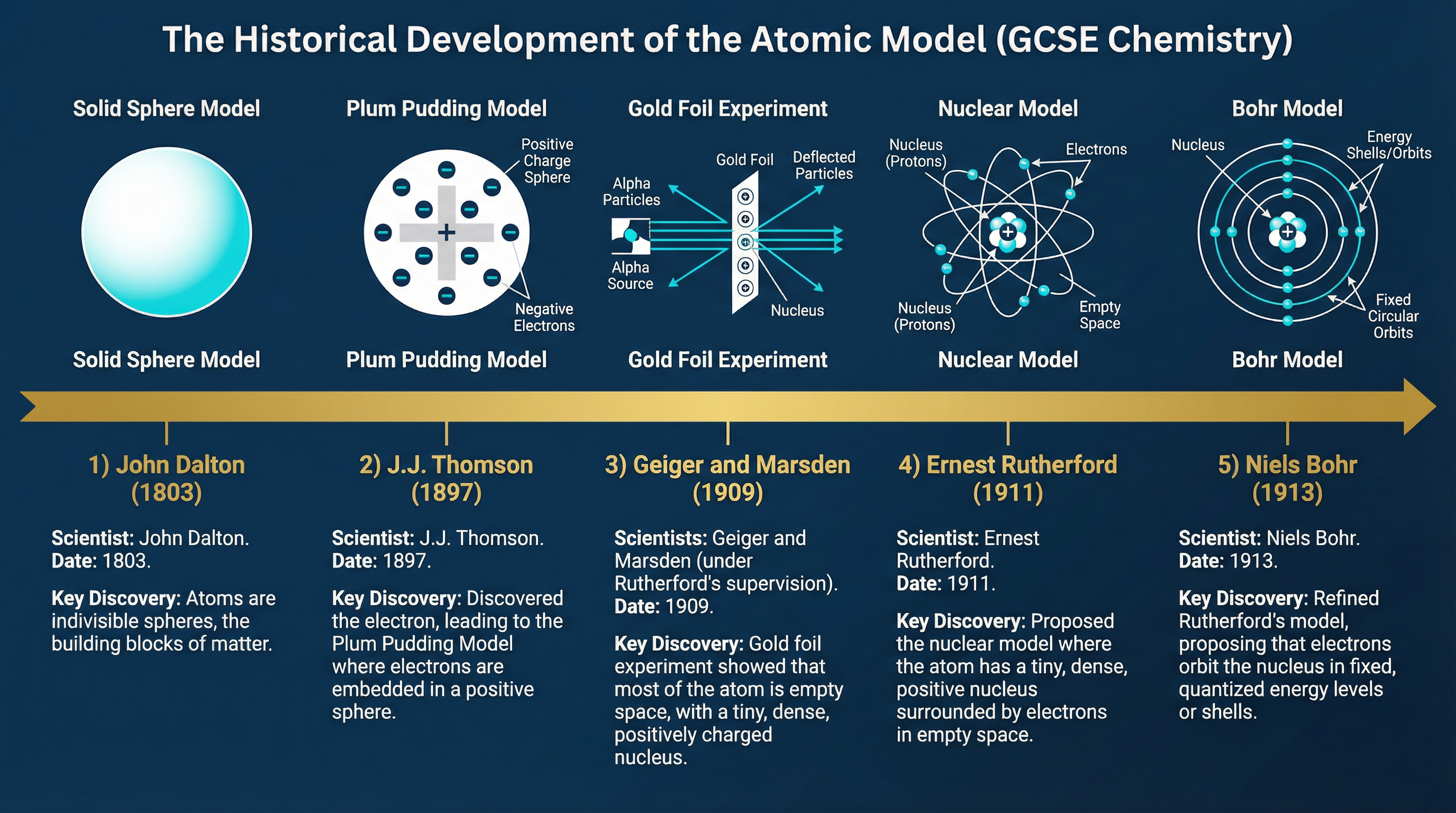
Scientific models change when new experimental evidence emerges that cannot be explained by the existing model. You must know the timeline:
1. **John Dalton (1803):** Proposed that atoms are solid, indivisible spheres.
2. **J.J. Thomson (1897):** Discovered the electron. Proposed the **Plum Pudding Model**: a ball of positive charge with negative electrons embedded within it.
3. **Ernest Rutherford (1911):** Directed the **Alpha Particle Scattering Experiment** (conducted by Geiger and Marsden). They fired positive alpha particles at thin gold foil. Most passed straight through (proving atoms are mostly empty space), but a few deflected at large angles (proving the existence of a small, dense, positive nucleus). This led to the **Nuclear Model**.
4. **Niels Bohr (1913):** Adapted the nuclear model by proposing that electrons orbit the nucleus at specific distances in fixed energy levels (shells).
## Mathematical/Scientific Relationships
You must be able to calculate the number of sub-atomic particles using the Periodic Table.
* **Atomic Number (Proton Number):** The smaller number. It equals the number of protons. (And the number of electrons in a neutral atom).
* **Mass Number (Nucleon Number):** The larger number. It equals the total number of protons + neutrons.
**Formula to memorise:**
Number of Neutrons = Mass Number - Atomic Number
**Example Calculation:**
Find the sub-atomic particles for Sodium (Na). The Periodic Table shows a mass number of 23 and an atomic number of 11.
* Protons = 11 (from atomic number)
* Electrons = 11 (same as protons because it's a neutral atom)
* Neutrons = 23 - 11 = 12
## Practical Applications
Understanding the particle model is essential for explaining everyday phenomena. For example, why do bridges have expansion joints? Because the metal particles vibrate more and move slightly further apart on hot days, causing the metal to expand. Understanding atomic structure is the basis for all modern chemistry, including nuclear energy (which relies on splitting the nucleus) and medical imaging using radioactive isotopes.