Topic C2: Elements, compounds and mixtures — OCR GCSE Study Guide
Exam Board: OCR | Level: GCSE
Master the fundamental building blocks of Chemistry. This topic covers the distinction between elements, compounds, and mixtures, explores how substances bond, and details the separation techniques essential for laboratory analysis.
## Overview
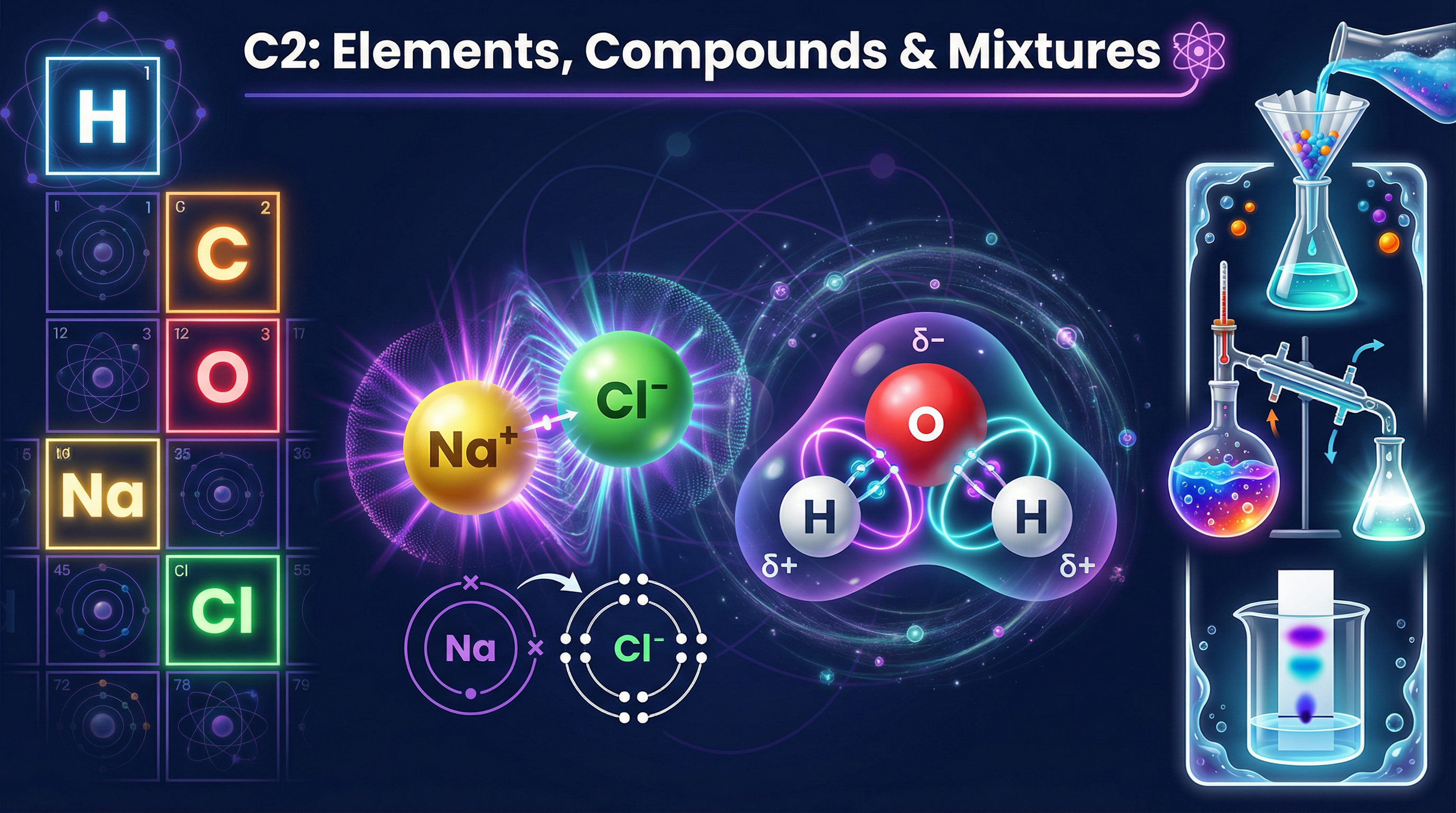
Welcome to Topic C2: Elements, Compounds and Mixtures. This foundational topic is the bedrock of your GCSE Chemistry course. It explores the very nature of matter—how atoms interact to form the myriad of substances around us, and how we can separate them when they are mixed.
Understanding the difference between a pure substance and a mixture is crucial, not just in the laboratory, but for the exam, where the scientific definition of 'pure' is rigorously tested. This topic connects heavily with quantitative chemistry (Topic C3) when calculating relative formula masses, and underpins all organic chemistry (Topic C7) when discussing covalent bonding in carbon compounds.
Typical exam questions range from simple multiple-choice questions identifying separation techniques, to complex six-mark extended response questions requiring you to relate the structure and bonding of a substance to its bulk physical properties.
Listen to our comprehensive revision podcast to solidify these concepts:

## Key Concepts
### Concept 1: Pure Substances and Mixtures
In everyday language, 'pure' might mean 'natural' or 'unprocessed' (like 'pure orange juice'). However, in chemistry, a pure substance contains only **one type of element or one type of compound**.
Examiners test this by asking how you can distinguish a pure substance from a mixture. The answer lies in melting and boiling points. A pure substance melts at a specific, sharp temperature. A mixture melts over a range of temperatures.
**Example**: Pure water boils at exactly 100°C. If salt is added (creating a mixture), the boiling point increases and occurs over a range of temperatures.
### Concept 2: Separation Techniques
When substances are physically mixed (not chemically bonded), they can be separated using physical processes.
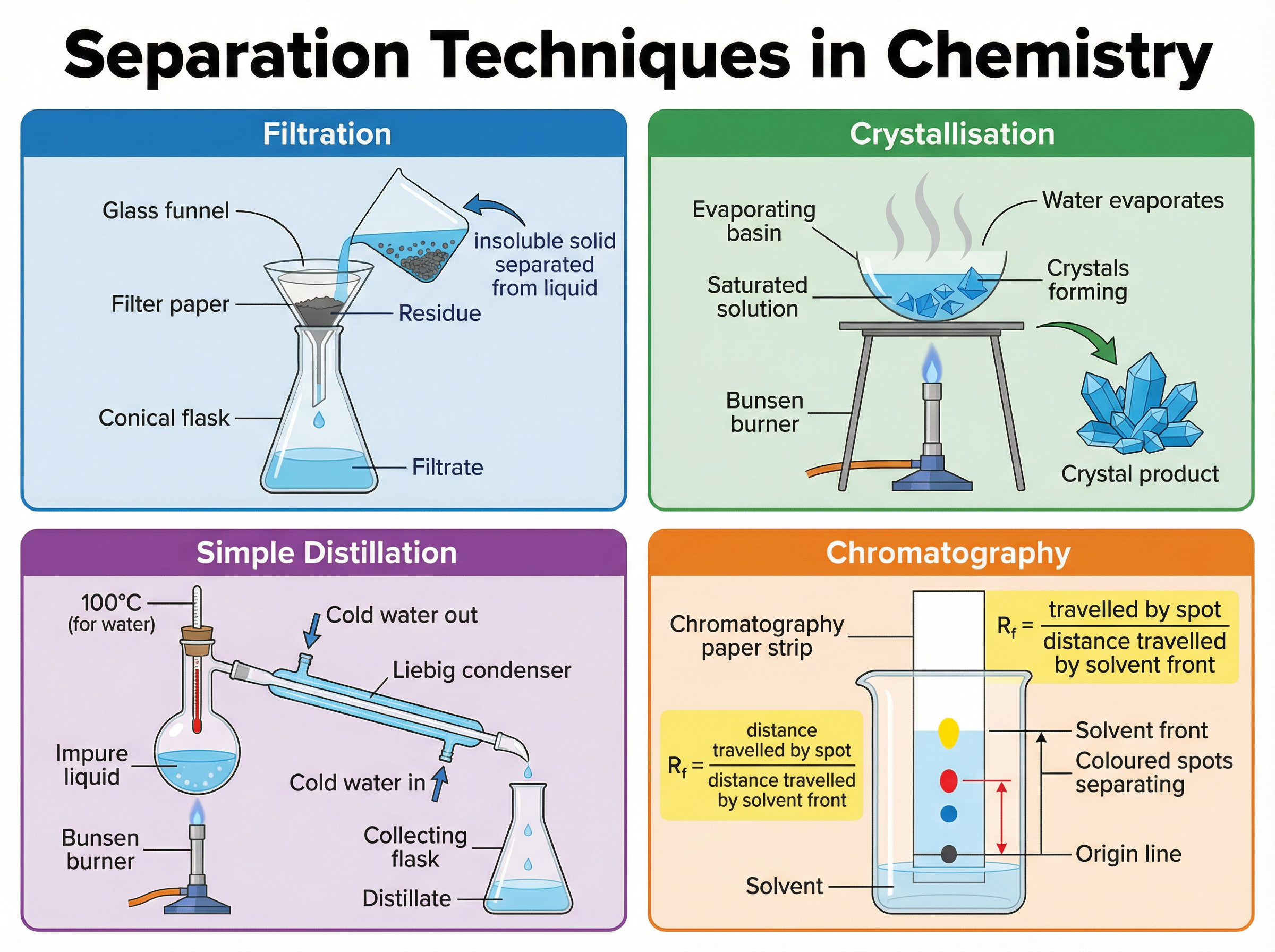
- **Filtration**: Separates an insoluble solid from a liquid. The liquid that passes through the filter paper is the filtrate; the solid left behind is the residue.
- **Crystallisation**: Separates a soluble solid from a solvent. The solution is heated to evaporate the solvent, leaving crystals behind.
- **Simple Distillation**: Separates a liquid from a solution (e.g., pure water from seawater). It relies on the liquid having a much lower boiling point than the dissolved solid.
- **Chromatography**: Separates mixtures of soluble substances, such as inks or dyes. The mobile phase (solvent) moves through the stationary phase (paper), carrying different substances at different rates.
### Concept 3: Chemical Bonding
When elements react, their atoms join with other atoms to form compounds. This involves giving, taking, or sharing electrons to achieve a full outer electron shell.
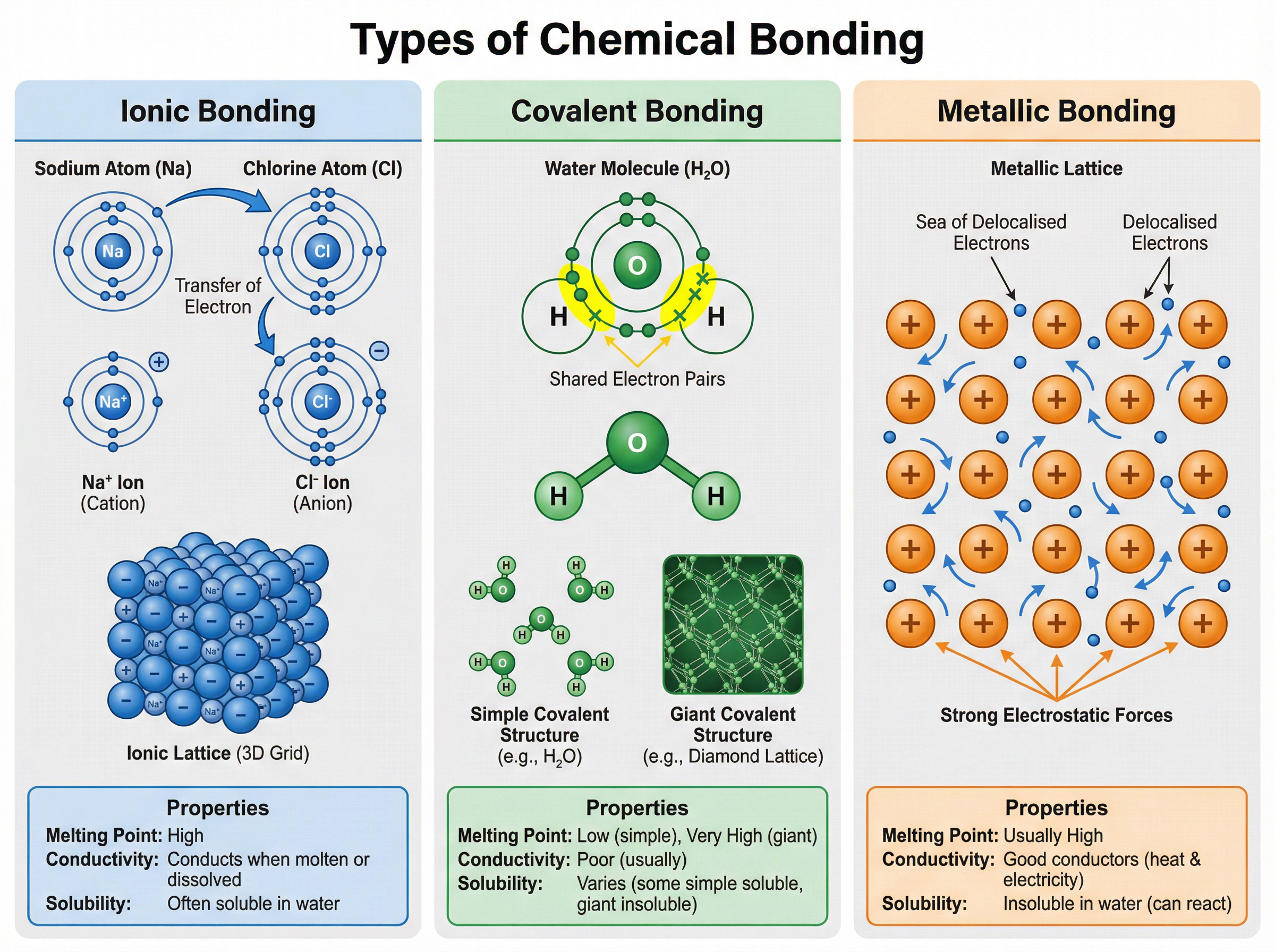
- **Ionic Bonding**: Occurs between metals and non-metals. Electrons are transferred. Metals lose electrons to form positive ions (cations); non-metals gain electrons to form negative ions (anions). The strong electrostatic forces between these oppositely charged ions form a giant ionic lattice.
- **Covalent Bonding**: Occurs between non-metals. Atoms share pairs of electrons. Simple covalent molecules (like H2O) have strong bonds within the molecule but weak intermolecular forces between molecules. Giant covalent structures (like diamond) have strong covalent bonds throughout the entire lattice.
- **Metallic Bonding**: Occurs in metals. The outer electrons become delocalised, creating a lattice of positive metal ions in a 'sea' of electrons.
### Concept 4: Carbon Allotropes
Carbon is unique because it can form several different structures (allotropes) with entirely different properties, despite all being made of just carbon atoms.

- **Diamond**: Each carbon atom forms four covalent bonds in a rigid 3D tetrahedral structure. It is extremely hard and does not conduct electricity.
- **Graphite**: Each carbon atom forms three covalent bonds in flat hexagonal layers. The fourth electron is delocalised, allowing graphite to conduct electricity. The layers can slide over each other, making it soft and slippery.
- **Graphene**: A single layer of graphite. It is incredibly strong, light, and an excellent conductor.
- **Fullerenes**: Molecules of carbon atoms with hollow shapes, such as Buckminsterfullerene (C60).
## Mathematical/Scientific Relationships
### Calculating Rf Values (Chromatography)
The Retention Factor ($R_f$) is used to identify substances in chromatography.
$$R_f = \frac{\text{distance moved by substance}}{\text{distance moved by solvent}}$$
- The $R_f$ value is always between 0 and 1.
- It has no units.
- You must measure from the pencil baseline to the centre of the spot.
## Practical Applications
**Required Practical: Chromatography**
Students must investigate how paper chromatography can be used to separate and tell the difference between coloured substances. Examiners often ask why the baseline must be drawn in pencil (ink would dissolve and run up the paper) and why the solvent level must be below the baseline (so the spots don't wash off into the solvent).
**Nanoparticles**
Nanoparticles (1-100 nm in size) have a massive surface area to volume ratio. This makes them highly effective as catalysts, in suncreams, and for targeted drug delivery. However, their small size means they can easily pass through cell membranes, raising concerns about their long-term health and environmental impacts.