Topic C3: Chemical reactions — OCR GCSE Study Guide
Exam Board: OCR | Level: GCSE
Master the fundamental principles of chemical reactions, from balancing equations to predicting electrolysis products. This high-yield topic connects mass conservation, energetics, and redox processes, forming the foundation for almost every calculation question in your GCSE Chemistry exam.
## Overview
Topic C3: Chemical Reactions is the engine room of GCSE Chemistry. This topic bridges the gap between theoretical atomic structure and practical chemical processes. It is fundamentally about how and why substances change into new substances, and how we quantify those changes.
Understanding this topic is critical because it introduces the **Law of Conservation of Mass** and the concept of the **mole**—the foundational tools for all chemical calculations (stoichiometry). You will also explore **energetics** (exothermic and endothermic reactions), **redox** (oxidation and reduction), and **electrolysis**.
Examiners love to test C3 because it allows them to assess your mathematical skills alongside your conceptual understanding. Typical exam questions will ask you to balance equations, calculate reacting masses, draw reaction profiles, or predict the products of electrolysis. Mastery here guarantees you can access the high-tariff calculation marks that often determine the boundary between a Grade 6 and a Grade 8/9.
---
## Key Concepts
### Concept 1: The Law of Conservation of Mass & Balanced Equations
The fundamental rule of chemistry is that **no atoms are created or destroyed during a chemical reaction**. They are simply rearranged to form new products. Therefore, the total mass of the reactants must exactly equal the total mass of the products.
To represent this on paper, we use balanced chemical equations. When balancing an equation, you must ensure there is the same number of atoms of each element on both sides of the equation.
**Crucial Examiner Point**: You can only add large numbers (coefficients) *in front* of formulas to balance an equation. You must **never** change the small subscript numbers within a formula, as this changes the chemical identity of the substance.
**Example**: The combustion of methane.
Unbalanced: CH₄ + O₂ → CO₂ + H₂O
Balanced: CH₄ + 2O₂ → CO₂ + 2H₂O
**Mass Changes in Non-Enclosed Systems**
Examiners frequently try to trick candidates with questions about mass "loss" or "gain". If a reaction takes place in an open flask (a non-enclosed system) and a gas is produced, the gas will escape into the atmosphere. The mass of the flask and its contents will appear to decrease. Conversely, if a solid reacts with a gas from the air (like burning magnesium in oxygen), the mass will appear to increase because the gas has been incorporated into the solid product. The mass is still conserved overall; you just haven't weighed the gas.
### Concept 2: The Mole and Avogadro's Constant
Atoms are too small to count individually, so chemists count them in "moles". One mole of any substance contains exactly $6.02 \times 10^{23}$ particles (atoms, molecules, or ions). This number is known as **Avogadro's constant**.
The mass of one mole of a substance in grams is numerically equal to its relative formula mass ($M_r$). For example, carbon has an $A_r$ of 12, so one mole of carbon weighs 12g. Water ($H_2O$) has an $M_r$ of 18 ($16 + 1 + 1$), so one mole of water weighs 18g.
This concept allows us to calculate the masses of reactants and products in a reaction using the formula:
**Mass = Moles × Relative Formula Mass**
### Concept 3: Energetics (Exothermic and Endothermic Reactions)
Chemical reactions involve energy changes.
* **Exothermic reactions** transfer energy *to* the surroundings. The temperature of the surroundings increases. Examples include combustion, neutralisation, and many oxidation reactions. Hand warmers use exothermic reactions.
* **Endothermic reactions** take in energy *from* the surroundings. The temperature of the surroundings decreases. Examples include thermal decomposition and the reaction of citric acid and sodium hydrogencarbonate. Sports injury cold packs use endothermic reactions.
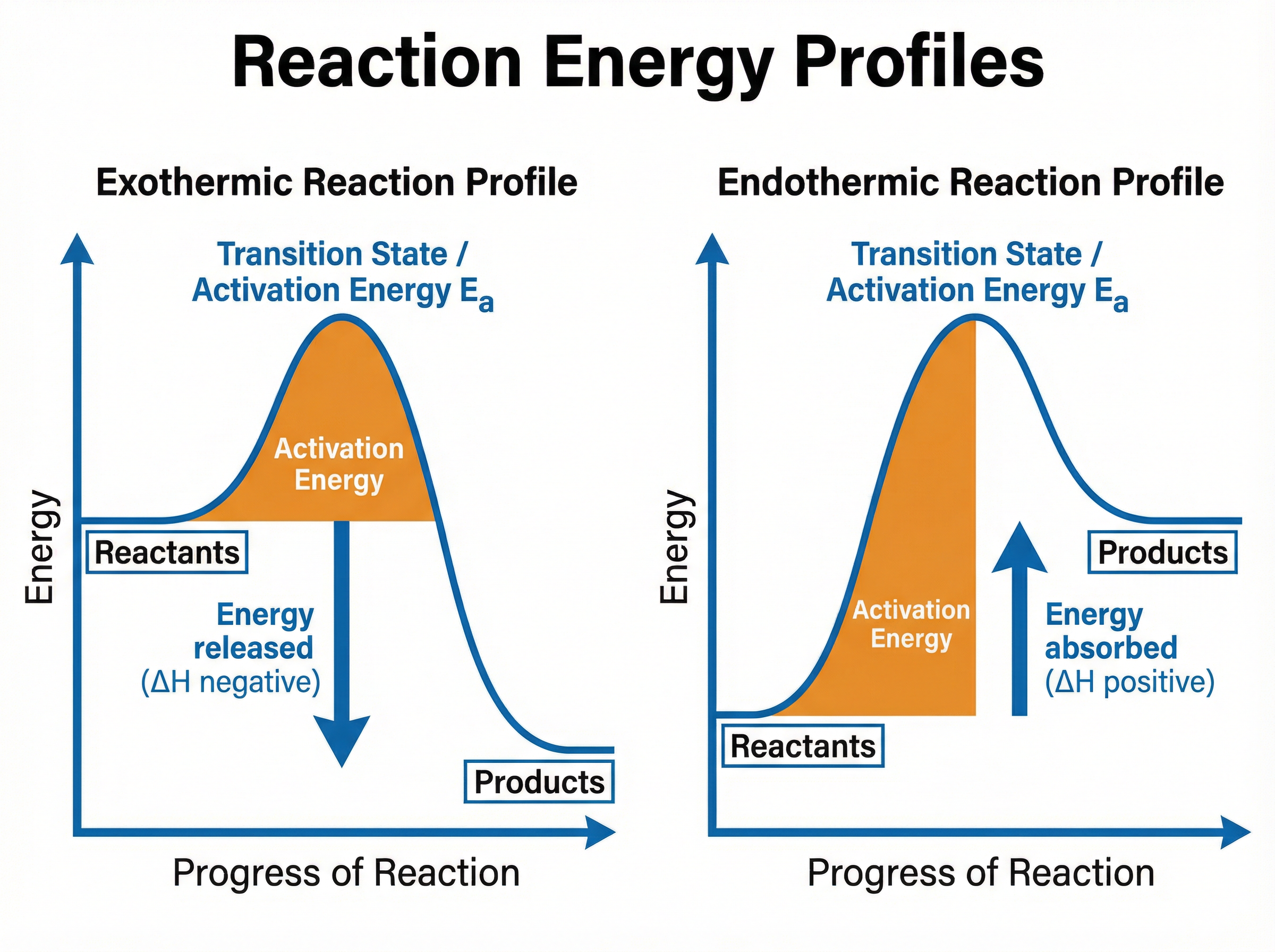
**Reaction Profiles**
These diagrams show the relative energies of reactants and products as the reaction progresses.
* The **activation energy ($E_a$)** is the minimum amount of energy that particles must have to react when they collide. It is shown as the energy "hump" from the reactants to the peak of the curve.
* In an exothermic profile, products are lower in energy than reactants.
* In an endothermic profile, products are higher in energy than reactants.
### Concept 4: Oxidation, Reduction, and Neutralisation
**Redox Reactions**
Historically, oxidation meant gaining oxygen, and reduction meant losing oxygen.
* *Example*: $2Cu + O_2 \rightarrow 2CuO$ (Copper is oxidised).
However, the more comprehensive definition (especially for Higher Tier) is based on electron transfer.
* **Oxidation is the loss of electrons.**
* **Reduction is the gain of electrons.**
These processes always occur together in a **redox** reaction.
**Acids, Bases, and Neutralisation**
Acids produce hydrogen ions ($H^+$) in aqueous solutions. Alkalis (soluble bases) produce hydroxide ions ($OH^-$).
When an acid reacts with a base, a **neutralisation** reaction occurs, producing a salt and water.
* General equation: $Acid + Base \rightarrow Salt + Water$
* Ionic equation for neutralisation: $H^+_{(aq)} + OH^-_{(aq)} \rightarrow H_2O_{(l)}$
### Concept 5: Electrolysis
Electrolysis is the process of passing a direct current (DC) through an ionic substance that is molten or in solution to break it down into its elements.
For electrolysis to work, the ions must be free to move. Therefore, solid ionic compounds cannot be electrolysed. They must be melted (molten) or dissolved in water (aqueous).
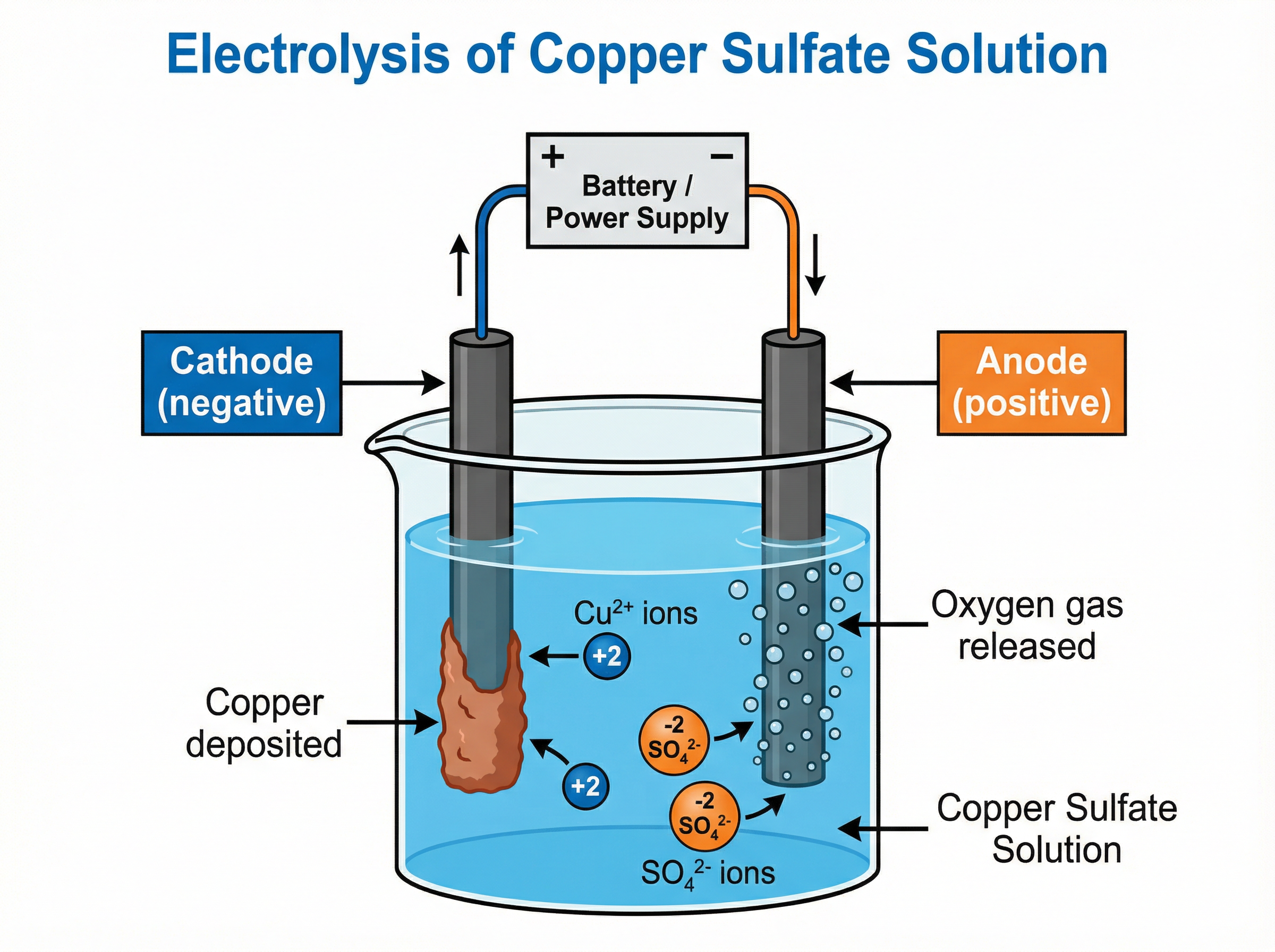
During electrolysis:
1. Positive ions (cations) move to the negative electrode (cathode). Here, they gain electrons (reduction).
2. Negative ions (anions) move to the positive electrode (anode). Here, they lose electrons (oxidation).
**Aqueous Electrolysis Rules (Higher Tier)**
When electrolysing aqueous solutions, water molecules also break down to form $H^+$ and $OH^-$ ions, complicating the products.
* **At the cathode (-):** Hydrogen gas is produced *unless* the metal is less reactive than hydrogen (e.g., copper, silver, gold), in which case the metal is produced.
* **At the anode (+):** Oxygen gas is produced (from $OH^-$ ions) *unless* a halide ion (chloride, bromide, iodide) is present, in which case the halogen is produced.
---
## Mathematical/Scientific Relationships
* **Moles Formula**: $\text{Moles} = \frac{\text{Mass (g)}}{\text{Relative Formula Mass } (M_r)}$
* *Must memorise.*
* **Concentration Formula (g/dm³)**: $\text{Concentration} = \frac{\text{Mass of solute (g)}}{\text{Volume of solvent (dm}^3\text{)}}$
* *Must memorise.* Remember to convert cm³ to dm³ by dividing by 1000.
* **Concentration Formula (mol/dm³)** (Higher Tier): $\text{Concentration} = \frac{\text{Moles}}{\text{Volume (dm}^3\text{)}}$
* *Must memorise.*
* **Energy Change (Bond Energies)** (Higher Tier): $\text{Energy Change} = \text{Total Energy to Break Bonds} - \text{Total Energy Released Forming Bonds}$
* *Given in question context, but must know how to apply.* A negative result means exothermic; positive means endothermic.
---
## Practical Applications
**Required Practical: Temperature Changes (Energetics)**
This practical investigates the variables that affect temperature changes in reacting solutions, typically the neutralisation of hydrochloric acid with sodium hydroxide.
* **Apparatus**: Polystyrene cup (for insulation), beaker (to hold the cup), thermometer, measuring cylinders.
* **Method**:
1. Measure a set volume of dilute HCl into the polystyrene cup.
2. Record the initial temperature.
3. Add a set volume of NaOH solution.
4. Put a lid on the cup (to reduce heat loss) and stir gently with the thermometer.
5. Record the maximum temperature reached.
6. Repeat with increasing volumes of NaOH.
* **Expected Results**: The maximum temperature will increase as more NaOH is added because more neutralisation occurs (exothermic). Eventually, the temperature will start to decrease because the HCl is fully neutralised, and adding more NaOH just cools the mixture down.
* **Examiner Focus**: Examiners frequently ask why a polystyrene cup and lid are used (to reduce heat loss to the surroundings, making the temperature reading more accurate).

Listen to the podcast for a comprehensive review of the topic, including a quick-fire recall quiz!