Topic C5: Monitoring and controlling chemical reactions — OCR GCSE Study Guide
Exam Board: OCR | Level: GCSE
Master the mathematics and mechanics of chemical reactions with this comprehensive guide to C5. From mole calculations to dynamic equilibrium, you'll learn how to predict, control, and measure chemical changes to secure top marks in your GCSE exams.
## Overview

Topic C5 is the cornerstone of quantitative chemistry. It bridges the gap between theoretical concepts and practical laboratory work, teaching you how to measure, monitor, and control chemical reactions. This topic is essential because it introduces the mathematical tools required to calculate reacting masses, concentrations, and gas volumes—skills that are heavily assessed in both structured and unstructured exam questions.
You will also explore collision theory and dynamic equilibrium, concepts that connect directly to industrial applications like the Haber process. Examiners frequently test this topic through multi-step calculation questions and extended response items requiring you to apply Le Chatelier's principle. Mastery of C5 not only secures significant marks but also provides the foundational understanding necessary for advanced study in Chemistry.
## Key Concepts
### Concept 1: The Mole and Avogadro's Constant
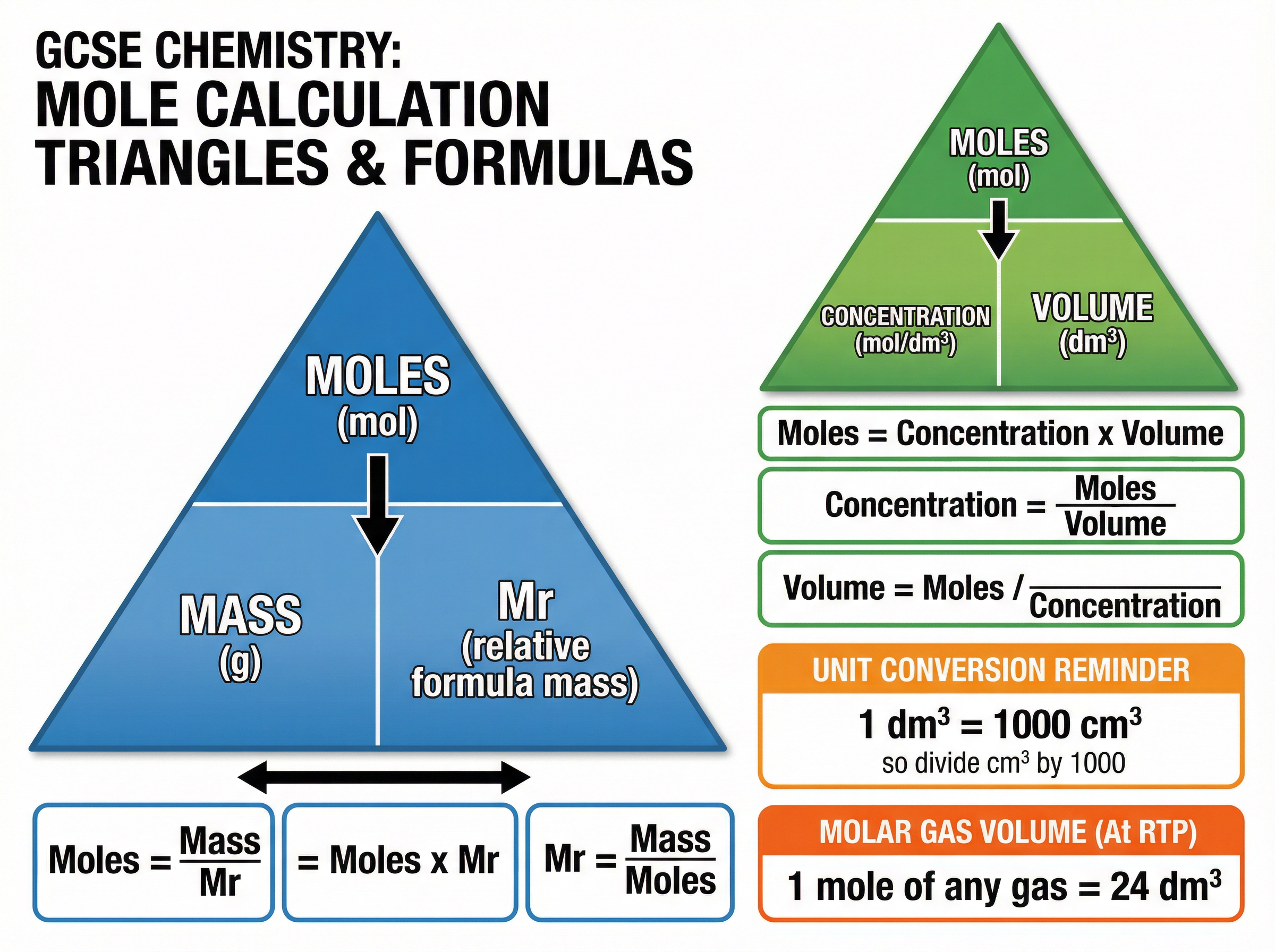
The mole is the standard unit for the amount of substance in chemistry. Because atoms are too small to count individually, we group them into 'moles'. One mole contains $6.02 \times 10^{23}$ particles (Avogadro's constant). The mass of one mole of any substance is equal to its relative formula mass ($M_r$) in grams.
Understanding this allows you to convert between mass and the number of particles. Examiners often test this by asking you to calculate the number of moles in a given mass, or vice versa.
**Example**: Calculate the number of moles in 88g of carbon dioxide ($CO_2$).
$M_r$ of $CO_2 = 12 + (16 \times 2) = 44$.
Moles = Mass / $M_r = 88 / 44 = 2$ moles.
### Concept 2: Concentration of Solutions
Concentration measures how much solute is dissolved in a given volume of solvent, typically expressed in $mol/dm^3$. This is a critical concept for titrations and required practicals. The most common error candidates make is failing to convert volumes from $cm^3$ to $dm^3$ before calculating. Always divide $cm^3$ by 1000 to get $dm^3$.
**Example**: Calculate the concentration of a solution containing 0.5 moles of NaCl dissolved in $250 cm^3$ of water.
Volume in $dm^3 = 250 / 1000 = 0.25 dm^3$.
Concentration = Moles / Volume = $0.5 / 0.25 = 2.0 mol/dm^3$.
### Concept 3: Rates of Reaction and Collision Theory
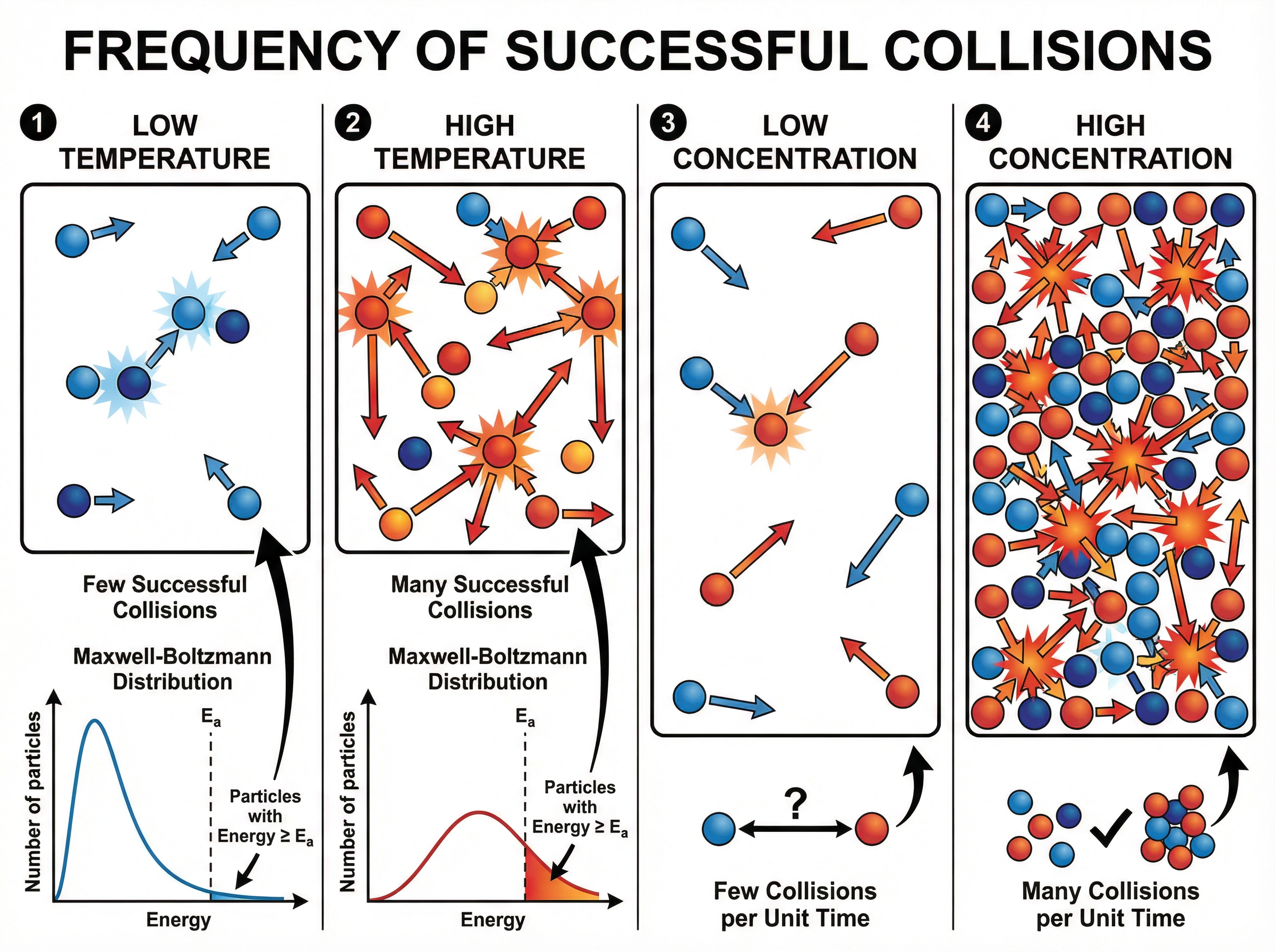
For a chemical reaction to occur, reactant particles must collide with sufficient energy to break existing bonds. This minimum energy threshold is called the **activation energy**. Collision theory states that the rate of a reaction depends on the **frequency of successful collisions** between reacting particles.
Factors affecting the rate include temperature, concentration, pressure (for gases), and surface area. Increasing any of these increases the frequency of collisions. Increasing temperature also increases the proportion of particles with energy greater than the activation energy, leading to more successful collisions per unit time.
### Concept 4: Dynamic Equilibrium and Le Chatelier's Principle
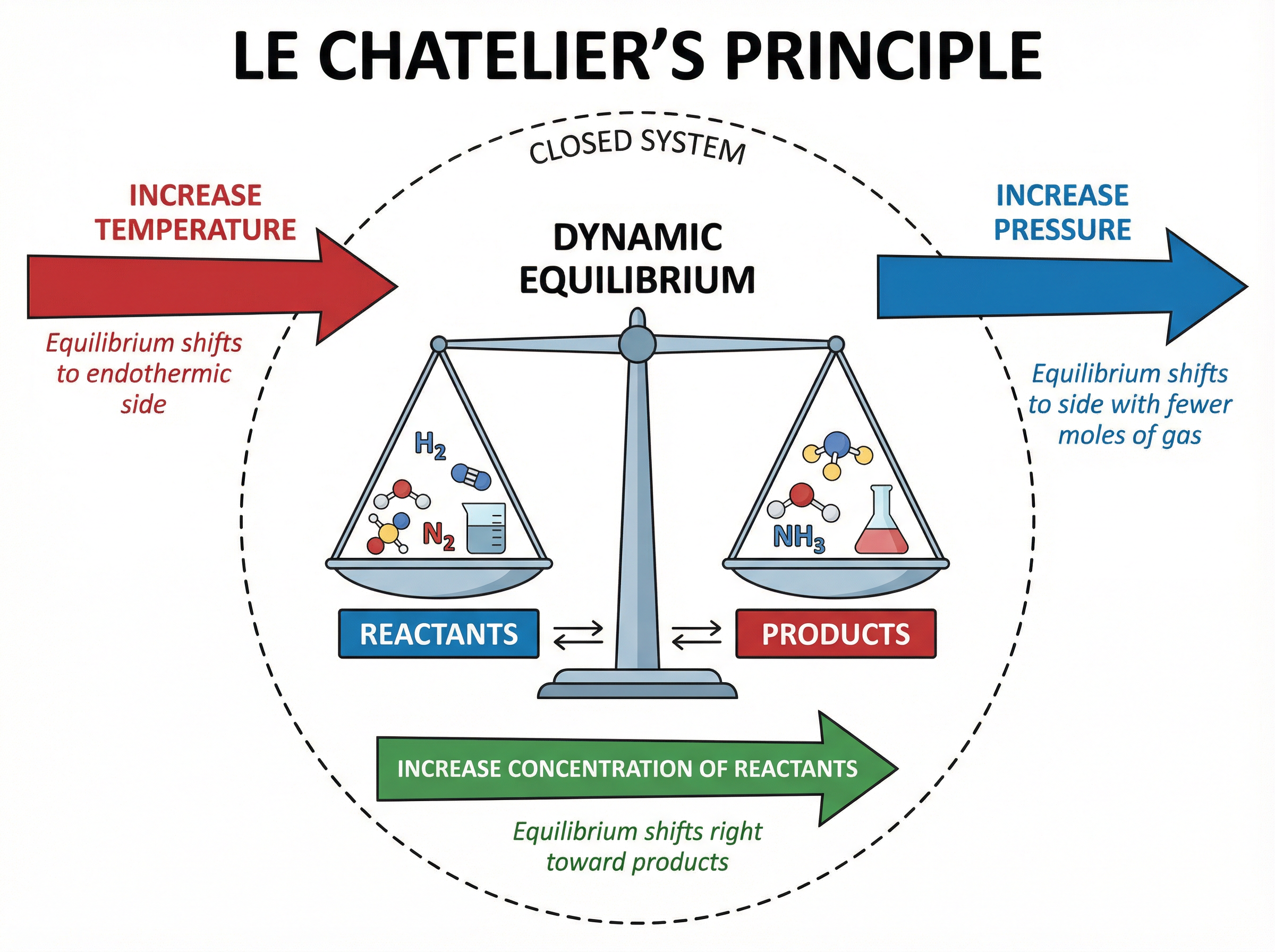
In a closed system, a reversible reaction will eventually reach dynamic equilibrium. At this point, the rate of the forward reaction equals the rate of the reverse reaction, and the concentrations of reactants and products remain constant (though not necessarily equal).
Le Chatelier's Principle states that if a change is made to the conditions of a system at equilibrium, the system will respond to counteract that change. This principle is vital for predicting how changes in temperature, pressure, or concentration will affect the yield of a reversible reaction.
## Mathematical/Scientific Relationships
1. **Moles = Mass / $M_r$** (Must memorise)
2. **Concentration ($mol/dm^3$) = Moles / Volume ($dm^3$)** (Must memorise)
3. **Volume of Gas ($dm^3$) = Moles $\times 24$** (At room temperature and pressure; Must memorise)
4. **Percentage Yield = (Actual Yield / Theoretical Yield) $\times 100$** (Must memorise)
5. **Atom Economy = ($M_r$ of desired product / Sum of $M_r$ of all products) $\times 100$** (Must memorise)
## Practical Applications
**Required Practical: Titration**
Titrations are used to determine the exact concentration of an acid or alkali. You must know how to use a pipette to measure a precise volume of one solution into a conical flask, add an indicator (like phenolphthalein), and use a burette to add the other solution until the indicator changes colour (the end-point).
**Industrial Application: The Haber Process**
The production of ammonia ($NH_3$) from nitrogen and hydrogen is a classic example of dynamic equilibrium. The forward reaction is exothermic, and there are fewer moles of gas on the product side. Therefore, high pressure and low temperature favour the yield of ammonia. However, a low temperature would make the reaction too slow, so a compromise temperature of $450^\circ C$ and an iron catalyst are used to achieve an acceptable yield at a reasonable rate.
## Audio Revision
Listen to the comprehensive podcast episode covering all these topics, perfect for revising on the go:
