Topic C6: Global challenges — OCR GCSE Study Guide
Exam Board: OCR | Level: GCSE
Topic C6 connects your foundational chemistry knowledge to real-world global challenges, from industrial fertiliser production to tackling climate change. Examiners love this topic because it tests your ability to apply chemical principles—like equilibrium and reactivity—to evaluate sustainable solutions for our planet's future.
## Overview

Topic C6: Global Challenges is where theoretical chemistry meets the real world. This topic explores how chemical processes are used to extract resources, manufacture essential products like fertilisers, and develop sustainable materials, while also addressing the environmental consequences of these activities.
It is a highly synoptic topic, meaning it draws heavily on concepts you have learned previously, such as the reactivity series, bonding, and quantitative chemistry. Examiners frequently use C6 to test Assessment Objective 3 (AO3) – your ability to analyze information, interpret data (like Life Cycle Assessments), and evaluate competing industrial methods.
Typical exam questions in this area will ask you to balance the trade-offs between the rate of a reaction and its yield, or to justify the use of a specific extraction method based on environmental and economic factors. You will also need to demonstrate a solid grasp of organic chemistry, identifying functional groups and explaining polymerisation.
## Key Concepts
### Concept 1: Metal Extraction and the Reactivity Series
The method chosen to extract a metal from its ore depends entirely on its position in the reactivity series. Metals that are less reactive than carbon (such as iron, zinc, and copper) can be extracted by reduction with carbon. In this process, carbon displaces the metal from its oxide because carbon is more reactive. For example, in a blast furnace, iron(III) oxide is reduced by carbon monoxide to form molten iron.
However, metals that are more reactive than carbon (such as aluminium, magnesium, and sodium) hold onto their oxygen too strongly for carbon to remove it. These metals must be extracted using electrolysis of the molten compound. Electrolysis requires massive amounts of electrical energy, making the extraction of metals like aluminium significantly more expensive than iron.
**Example**: Extracting aluminium from bauxite (aluminium oxide, Al₂O₃) requires melting the ore (often mixed with cryolite to lower the melting point) and passing a large direct current through it. Aluminium ions (Al³⁺) gain electrons at the cathode to form aluminium atoms: Al³⁺ + 3e⁻ → Al.
### Concept 2: The Haber Process and Equilibrium Trade-offs
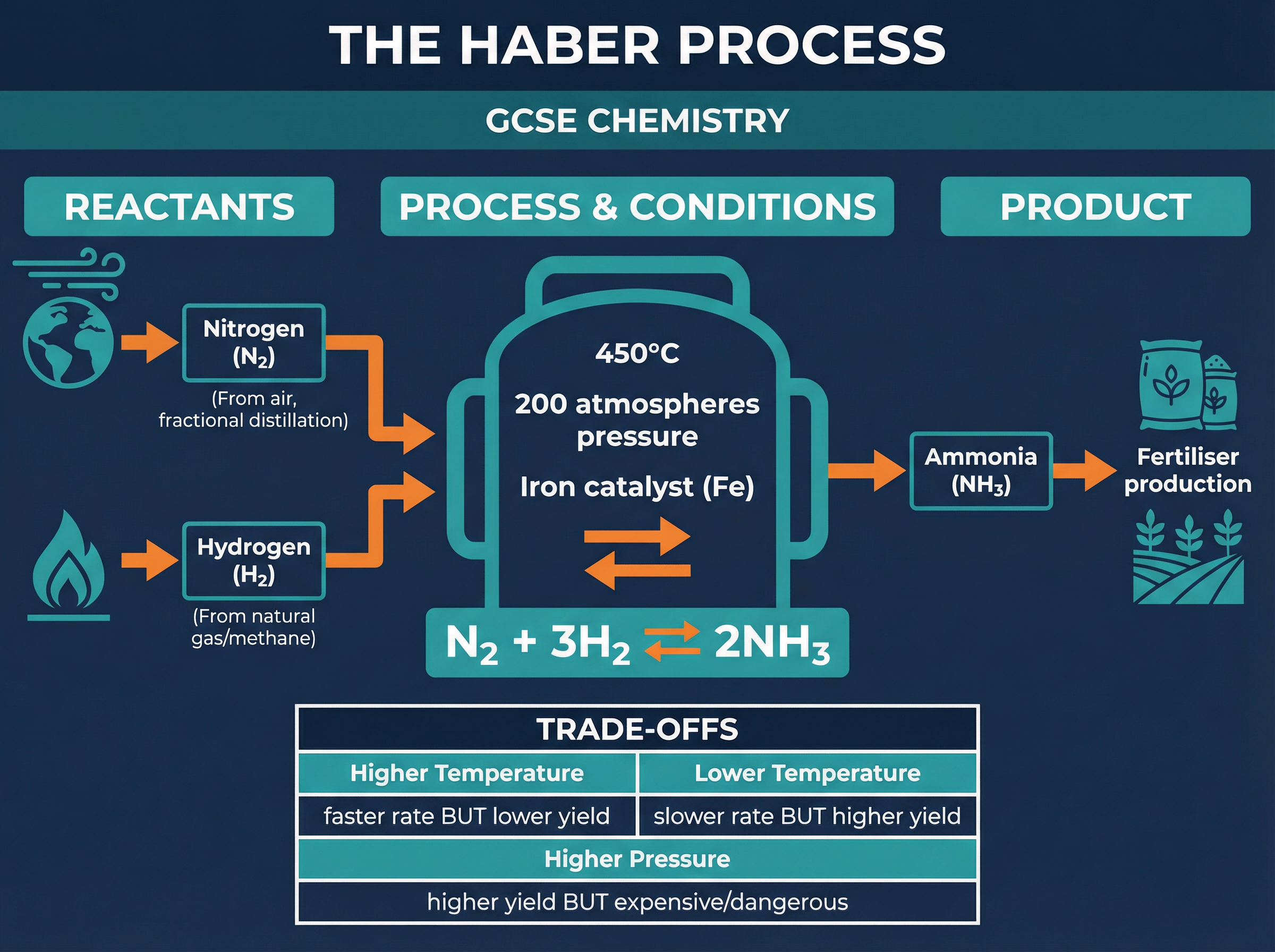
The Haber process is the industrial method for producing ammonia (NH₃), which is vital for manufacturing nitrogen-based fertilisers to sustain global food production. The reaction involves nitrogen (extracted from the air) and hydrogen (obtained from natural gas) reacting over an iron catalyst.
The reaction is reversible and reaches a dynamic equilibrium: N₂(g) + 3H₂(g) ⇌ 2NH₃(g). The forward reaction is exothermic. According to Le Chatelier's principle, lowering the temperature would shift the equilibrium to the right, increasing the yield of ammonia. However, a low temperature would make the reaction rate too slow to be economically viable. Therefore, a compromise temperature of approximately 450°C is used to balance a reasonable yield with an acceptable rate.
Similarly, there are four moles of reactant gas and only two moles of product gas. Increasing the pressure shifts the equilibrium to the right, increasing the yield. However, generating and containing very high pressures is expensive and dangerous. A compromise pressure of 200 atmospheres is typically used.
### Concept 3: Organic Chemistry and Functional Groups
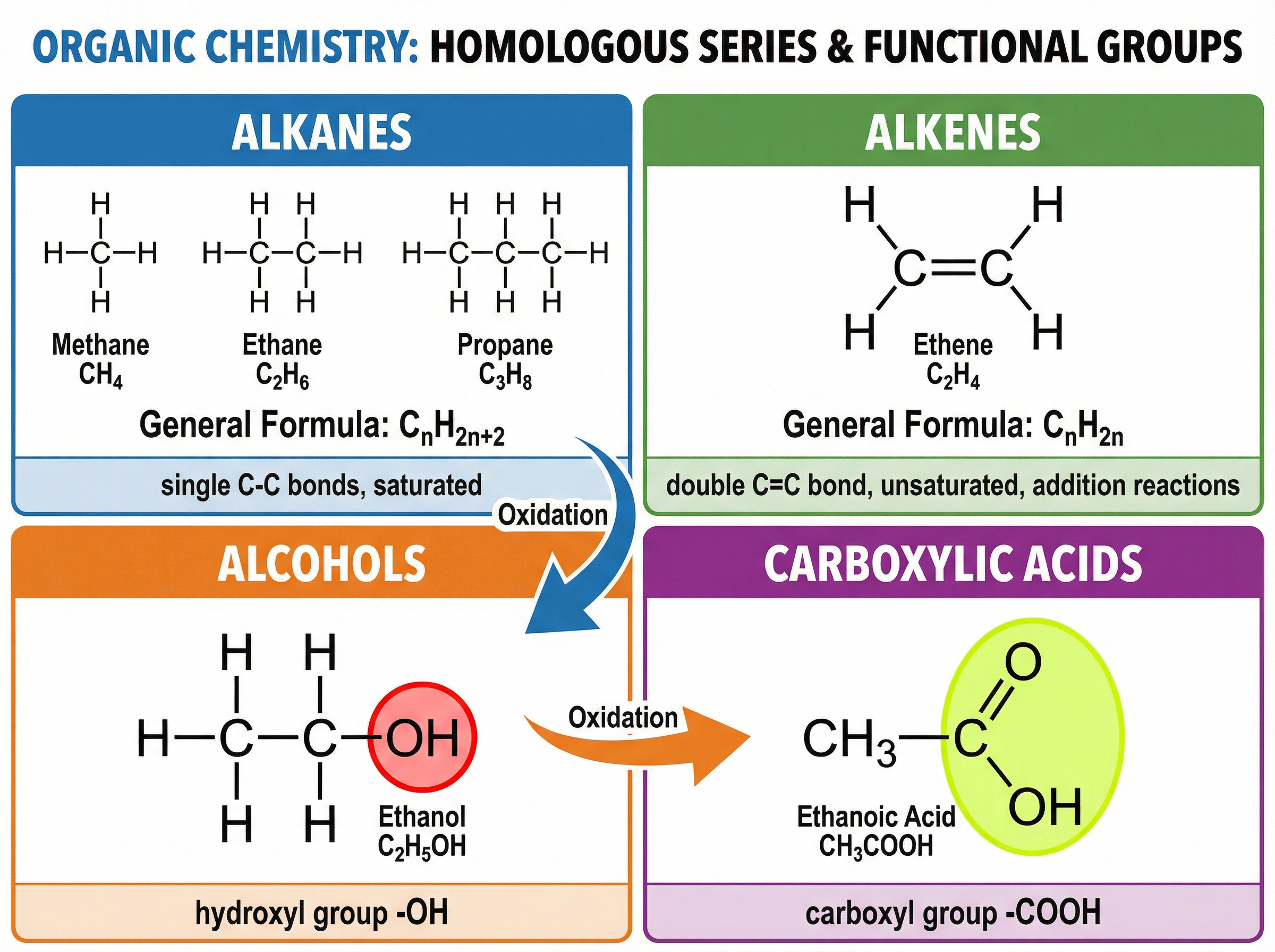
Organic chemistry focuses on carbon-based compounds, which are organized into homologous series—families of compounds with the same general formula and similar chemical properties due to the presence of a specific functional group.
- **Alkanes** (e.g., methane, ethane) are saturated hydrocarbons with only single carbon-carbon bonds. They are primarily used as fuels.
- **Alkenes** (e.g., ethene, propene) are unsaturated hydrocarbons containing at least one carbon-carbon double bond (C=C). This double bond makes them highly reactive, allowing them to undergo addition reactions, such as decolourising bromine water.
- **Alcohols** contain the hydroxyl functional group (-OH). Ethanol is widely used as a solvent, in alcoholic beverages, and as a renewable biofuel.
- **Carboxylic Acids** contain the carboxyl functional group (-COOH). They are weak acids, meaning they only partially ionise in water.
### Concept 4: The Earth's Atmosphere and Climate Change
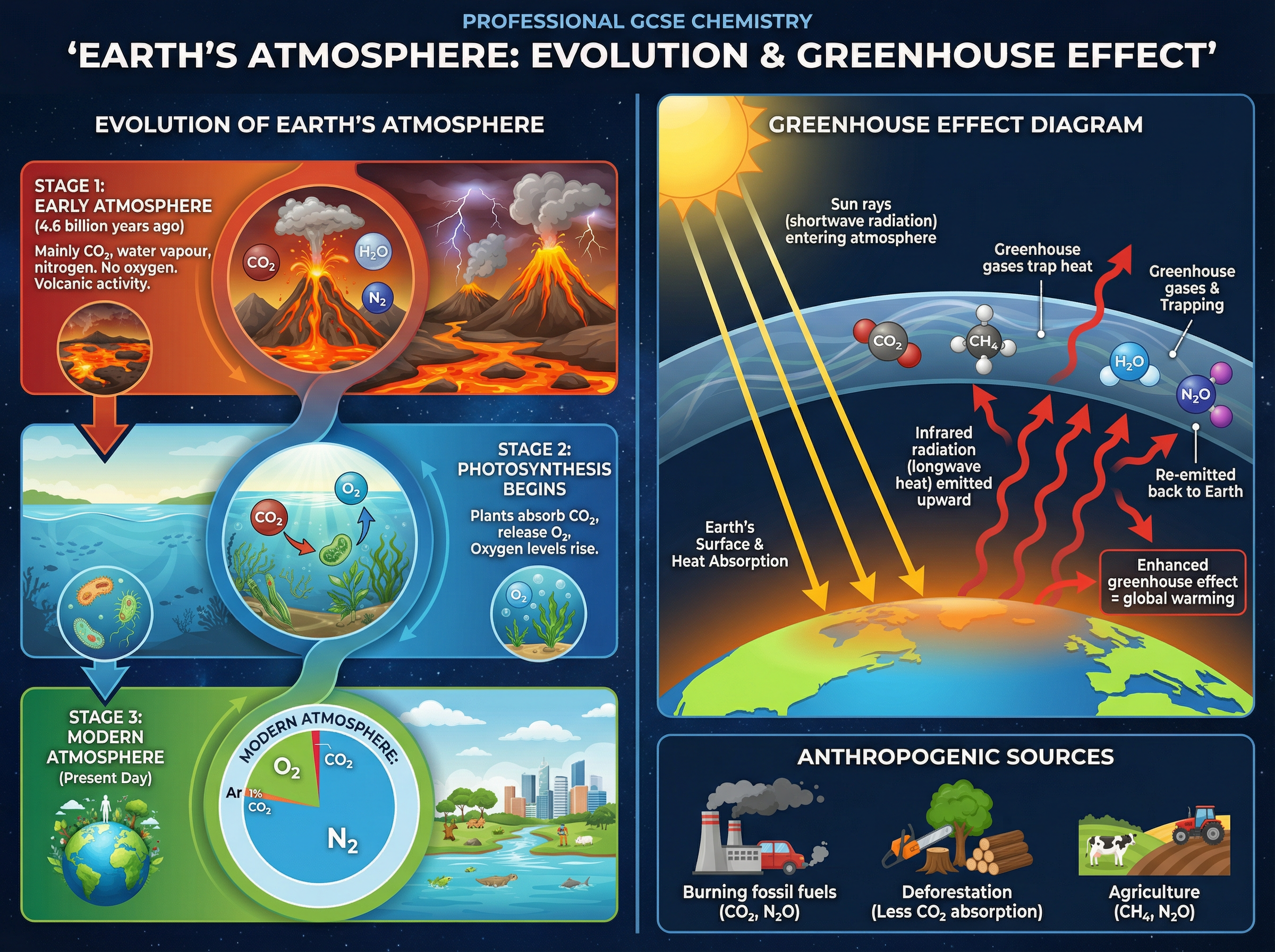
The Earth's early atmosphere was formed by intense volcanic activity, consisting mostly of carbon dioxide, water vapour, and nitrogen, with little to no oxygen. As the Earth cooled, water vapour condensed to form oceans, and a significant amount of CO₂ dissolved into them.
The evolution of algae and plants introduced photosynthesis, which gradually consumed CO₂ and released oxygen (O₂), eventually creating the oxygen-rich atmosphere we have today (roughly 78% nitrogen, 21% oxygen, and trace amounts of other gases).
The greenhouse effect is a natural and necessary phenomenon where gases like CO₂, methane (CH₄), and water vapour trap infrared radiation emitted by the Earth, keeping the planet warm enough to support life. However, human activities—such as burning fossil fuels and intensive agriculture—have artificially increased the concentration of these gases, leading to an enhanced greenhouse effect and global climate change.

## Mathematical/Scientific Relationships
- **General Formula for Alkanes**: CₙH₂ₙ₊₂ (Used to determine the molecular formula of any alkane given the number of carbon atoms, n).
- **General Formula for Alkenes**: CₙH₂ₙ (Used for alkenes with one double bond).
- **Haber Process Equation**: N₂(g) + 3H₂(g) ⇌ 2NH₃(g) (Essential to memorize for balancing equations and explaining equilibrium shifts).
## Practical Applications
Understanding these concepts is crucial for modern chemical engineering. Life Cycle Assessments (LCAs) are used by companies to evaluate the total environmental impact of a product, from raw material extraction to disposal. By analyzing LCAs, chemists can make informed decisions about whether to use single-use plastics or biodegradable alternatives, and how to improve recycling processes to conserve finite resources and reduce energy consumption.