Thermodynamics — WJEC A-Level Study Guide
Exam Board: WJEC | Level: A-Level
Master WJEC A-Level Thermodynamics by learning to construct perfect Born-Haber cycles and applying Gibbs Free Energy to predict reaction feasibility. This guide provides examiner insights, worked examples, and memory hooks to help you secure top marks.
## Overview
Thermodynamics in A-Level Chemistry is the study of energy changes in chemical reactions. For WJEC candidates, this topic (Unit 1.8) is a cornerstone of physical chemistry, focusing on two powerful tools: **Born-Haber cycles** and **Gibbs Free Energy**. Mastering this area is crucial as it not only carries significant marks but also provides a fundamental understanding of why some reactions happen and others don't. Born-Haber cycles allow us to calculate lattice enthalpies, a measure of ionic bond strength, by applying Hess's Law to a series of discrete energy steps. Gibbs Free Energy, meanwhile, combines enthalpy and entropy to give us a definitive measure of reaction feasibility. Exam questions are often multi-step calculations requiring precision, clear working, and a firm grasp of the underlying definitions. Expect to be tested on your ability to draw accurate cycles, manipulate the Gibbs equation, and interpret the results in context.

## Key Concepts
### Concept 1: Enthalpy Changes & Hess's Law
At the heart of thermodynamics are the specific, defined changes in heat energy, known as enthalpy changes (ΔH). You must know the formal definitions for several key processes. Hess's Law underpins this all, stating that the total enthalpy change for a reaction is independent of the route taken. This principle is what allows us to construct energy cycles to find unknown values.
**Example**: If we know the energy needed to turn sodium solid into gas (atomisation) and then into an ion (ionisation), and we know the energy change for chlorine molecules becoming ions (bond dissociation and electron affinity), we can use Hess's Law to find the Lattice Enthalpy of sodium chloride, provided we also know its overall enthalpy of formation.
### Concept 2: The Born-Haber Cycle
A Born-Haber cycle is a specific type of enthalpy cycle used to determine the lattice enthalpy of an ionic compound. It's a visual representation of Hess's Law, breaking down the formation of an ionic lattice into a series of steps. Marks are awarded for meticulous accuracy.
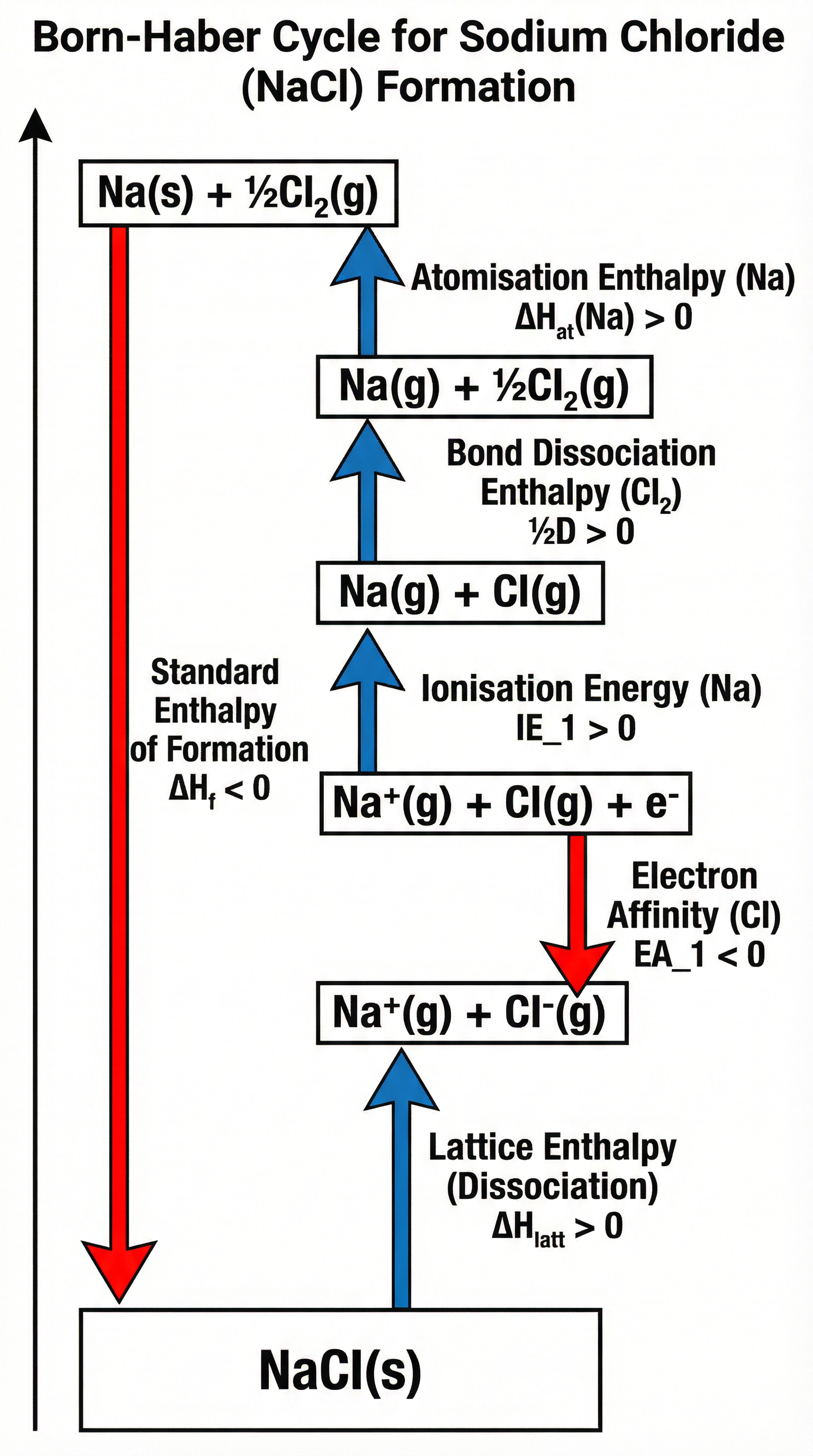
**Key Steps in a Born-Haber Cycle (for NaCl):**
1. **Start**: Elements in standard states (Na(s) + ½Cl₂(g)).
2. **Atomisation of Metal**: Na(s) → Na(g) (Endothermic)
3. **Atomisation of Non-Metal**: ½Cl₂(g) → Cl(g) (Endothermic - this is half the bond dissociation enthalpy)
4. **First Ionisation Energy of Metal**: Na(g) → Na⁺(g) + e⁻ (Endothermic)
5. **First Electron Affinity of Non-Metal**: Cl(g) + e⁻ → Cl⁻(g) (Exothermic)
6. **Lattice Enthalpy of Formation**: Na⁺(g) + Cl⁻(g) → NaCl(s) (Very Exothermic)
7. **Enthalpy of Formation**: The direct route from elements to the compound, linking the start and end points.
### Concept 3: Entropy (ΔS)
Entropy is a measure of the disorder or randomness of a system. The more ways particles and their energy can be arranged, the higher the entropy. A positive ΔS means the system has become more disordered (e.g., a solid melting to a liquid, or a reaction producing more moles of gas). A negative ΔS means the system has become more ordered.
**Example**: The reaction CaCO₃(s) → CaO(s) + CO₂(g) has a large positive entropy change because a mole of gas (CO₂) is produced, which is far more disordered than the solid reactant.
### Concept 4: Gibbs Free Energy (ΔG) & Feasibility
Gibbs Free Energy (ΔG) is the ultimate arbiter of whether a reaction is feasible. It combines the enthalpy change (ΔH) and the entropy change (ΔS) at a given temperature (T). A reaction is considered **feasible** (or spontaneous) if **ΔG ≤ 0**.
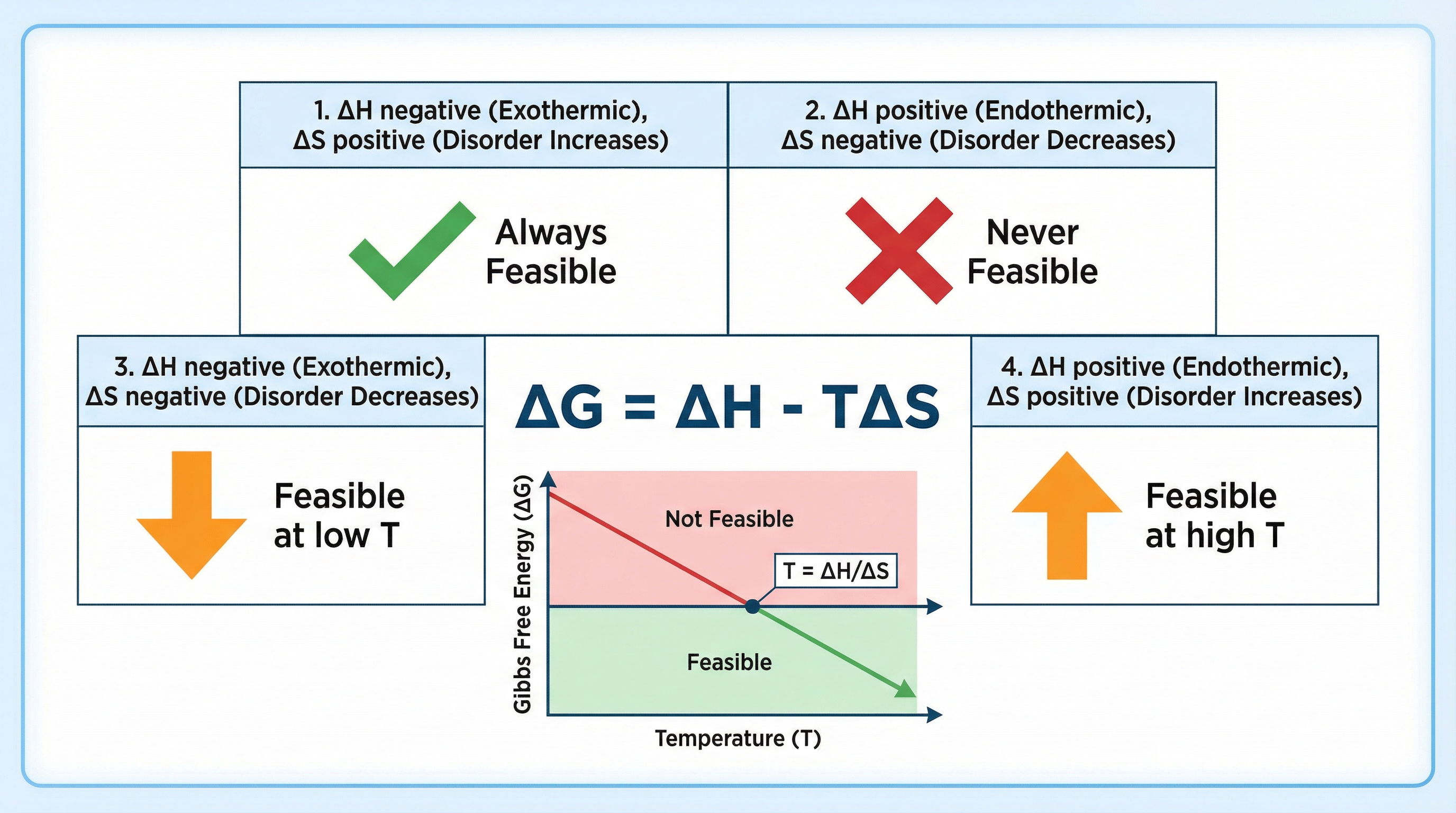
## Mathematical/Scientific Relationships
- **Gibbs Free Energy Equation**: `ΔG = ΔH - TΔS`
- **ΔG**: Gibbs Free Energy change (kJ mol⁻¹). **Must memorise**.
- **ΔH**: Enthalpy change (kJ mol⁻¹). **Must memorise**.
- **T**: Temperature in Kelvin (K). **Must memorise**.
- **ΔS**: Entropy change (J K⁻¹ mol⁻¹). **Must memorise**.
- **Temperature of Feasibility**: `T = ΔH / ΔS`
- This is derived from the Gibbs equation by setting ΔG = 0. It calculates the exact temperature at which a reaction becomes feasible. **Must memorise**.
## Unit Conversions
The most common mistake is failing to align units. Examiners test this deliberately.
- **Enthalpy (ΔH)** is almost always given in **kJ mol⁻¹**.
- **Entropy (ΔS)** is almost always given in **J K⁻¹ mol⁻¹**.
- **Action**: You MUST convert ΔS to kJ K⁻¹ mol⁻¹ by **dividing by 1000** before using the Gibbs equation. Explicitly write this conversion in your working to secure method marks.