Bonding, structure and properties — WJEC GCSE Study Guide
Exam Board: WJEC | Level: GCSE
Master the microscopic world that determines the macroscopic properties of all materials. This topic is the foundation of Chemistry, explaining why diamonds are hard, why metals conduct electricity, and how nanoparticles are revolutionising modern technology.
## Overview
Welcome to Bonding, Structure and Properties, a foundational topic that explains the behaviour of every substance in the universe. This topic is about understanding how atoms connect to one another and how those microscopic connections dictate the macroscopic properties we observe, such as melting points, electrical conductivity, and hardness.
Examiners love this topic because it tests your ability to link microscopic structure to macroscopic behaviour. You will frequently encounter 4-to-6 mark extended response questions asking you to compare the properties of two substances based on their bonding and structure. It is crucial to use precise terminology here—words like "ions," "molecules," "atoms," and "intermolecular forces" must be used accurately to secure marks. This topic connects heavily to quantitative chemistry and the periodic table, as the position of an element determines the type of bonding it will undergo.
Listen to our revision podcast for an audio summary:

## Key Concepts
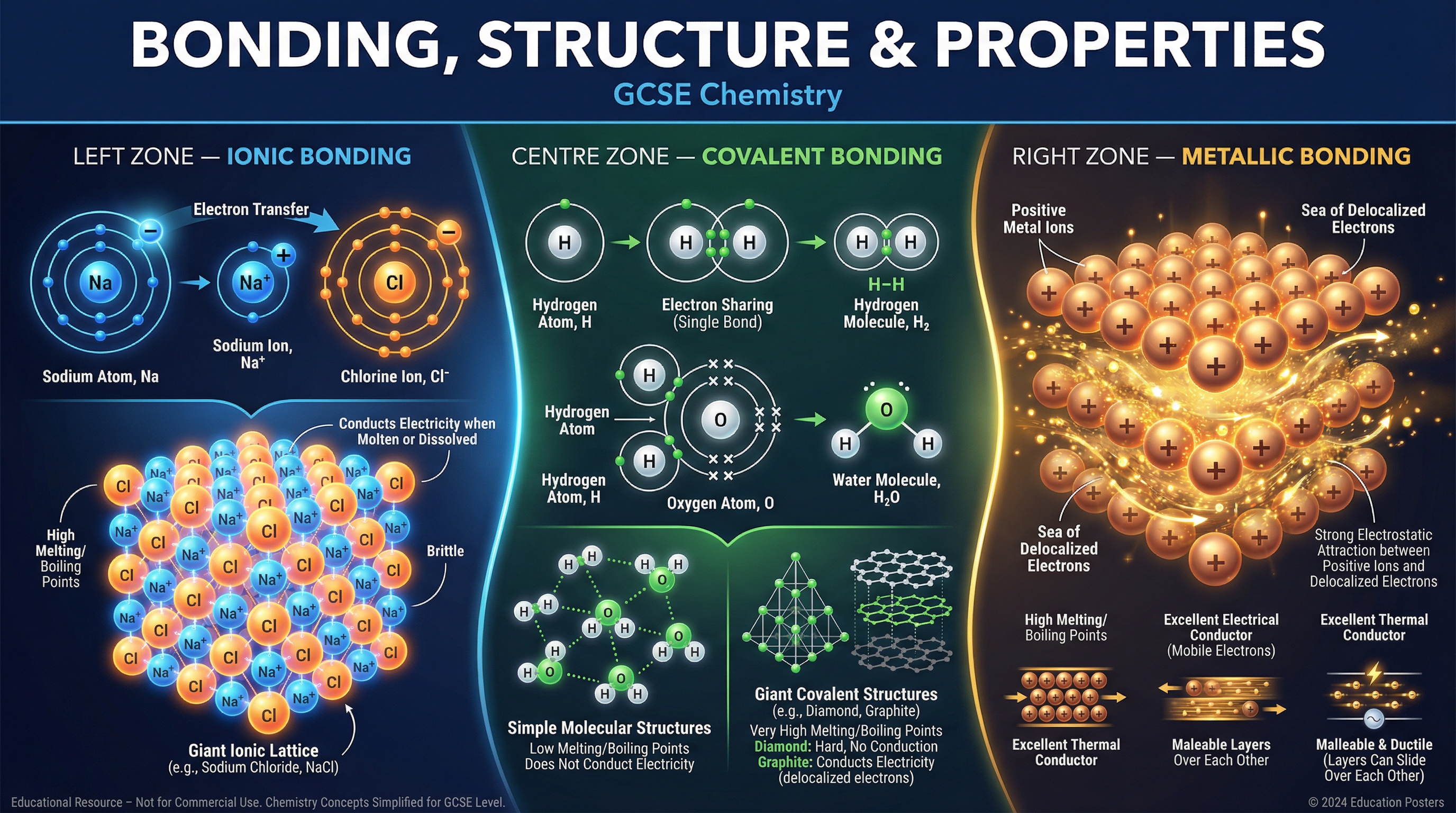
### Concept 1: Ionic Bonding and Giant Ionic Lattices
Ionic bonding occurs between metals and non-metals. The metal atoms lose electrons to form positively charged ions (cations), and the non-metal atoms gain these electrons to form negatively charged ions (anions). Both achieve a full outer electron shell, a stable configuration.
The key examiner phrase is: **"Electrostatic attraction between oppositely charged ions."**
These ions arrange themselves into a **giant ionic lattice**, a regular 3D repeating pattern.
**Properties:**
* **High Melting and Boiling Points:** Because there are many strong electrostatic forces between the oppositely charged ions throughout the giant lattice, a large amount of energy is required to overcome them.
* **Electrical Conductivity:** Solid ionic compounds *do not* conduct electricity because the ions are fixed in the lattice and cannot move. However, when molten (liquid) or dissolved in water (aqueous), the ions are free to move and carry charge.
**Example:** Sodium chloride (NaCl) forms when sodium transfers one electron to chlorine. The resulting Na+ and Cl- ions form a lattice with a melting point of 801°C.
### Concept 2: Covalent Bonding and Simple Molecules
Covalent bonding occurs between non-metal atoms. Instead of transferring electrons, atoms **share pairs of electrons** to achieve a full outer shell. The positively charged nuclei of the bonded atoms are strongly attracted to the shared pair of electrons.
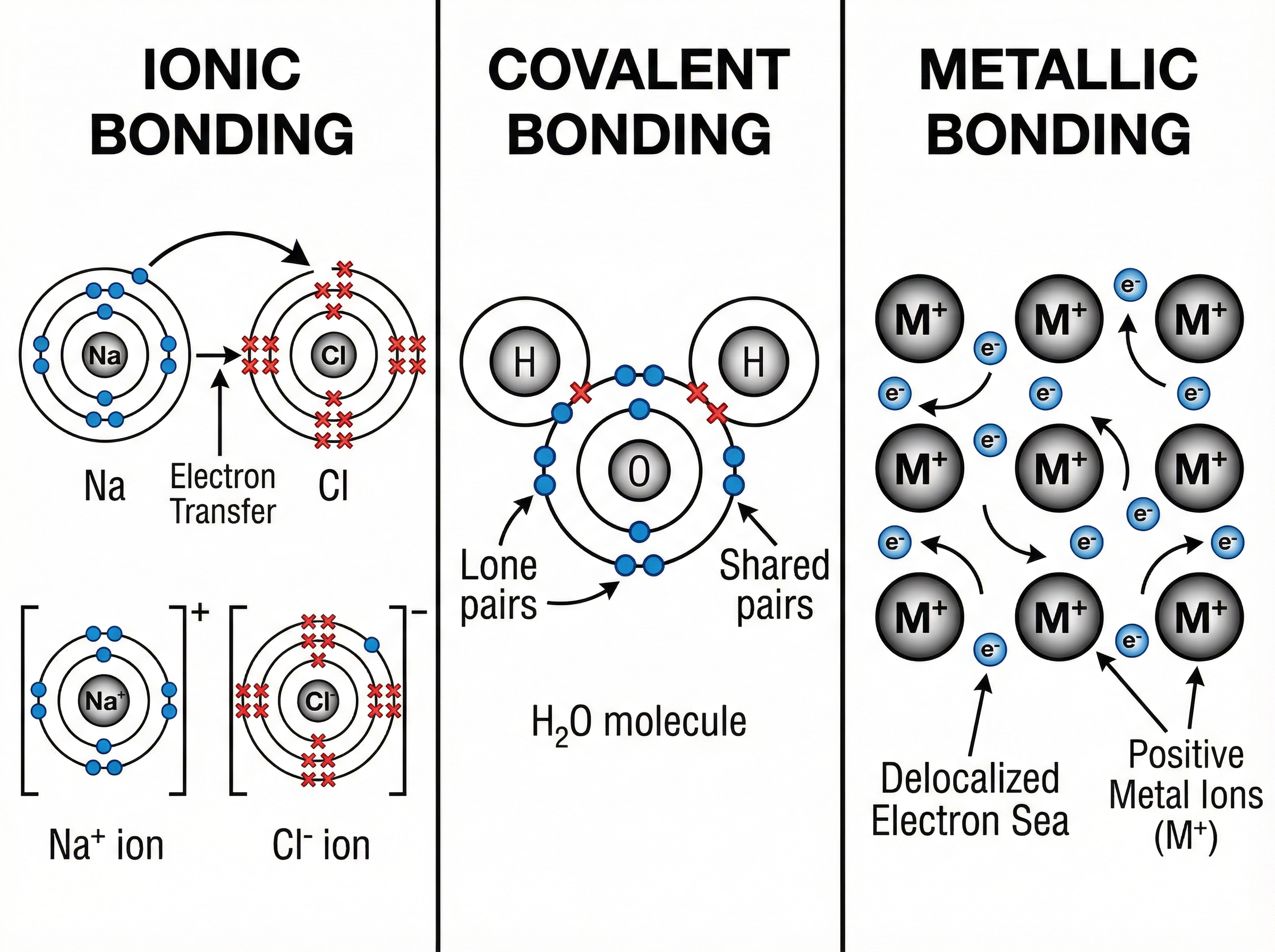
Many covalent substances form **simple molecular structures**, such as water ($H_2O$), carbon dioxide ($CO_2$), and methane ($CH_4$).
**Properties:**
* **Low Melting and Boiling Points:** While the covalent bonds *within* the molecules (intramolecular bonds) are very strong, the forces *between* the molecules (**intermolecular forces**) are weak. It is these weak intermolecular forces that are overcome when the substance melts or boils, requiring little energy.
* **No Electrical Conductivity:** Simple molecules have no overall charge and no delocalised electrons or mobile ions, so they cannot carry a charge in any state.
### Concept 3: Giant Covalent Structures
Some covalent substances form **giant covalent structures** (macromolecules), where millions of atoms are joined by covalent bonds in a continuous network.
**Properties:**
* **Very High Melting and Boiling Points:** To melt these substances, you must break the strong covalent bonds throughout the entire structure, which requires a massive amount of energy.
* **Variable Conductivity:** Most do not conduct electricity (e.g., diamond, silicon dioxide) because all outer electrons are used in bonding. Graphite is the major exception.
### Concept 4: Metallic Bonding
Metals consist of a giant structure of atoms arranged in a regular pattern. The outer shell electrons of metal atoms are **delocalised**—they are free to move throughout the whole structure.
The key examiner phrase is: **"Electrostatic attraction between a lattice of positive metal ions and a sea of delocalised electrons."**
**Properties:**
* **High Melting and Boiling Points:** The metallic bonds are strong, requiring significant energy to overcome.
* **Good Electrical and Thermal Conductors:** The delocalised electrons are free to move through the structure and carry electrical charge and thermal energy.
* **Malleable and Ductile:** The layers of positive ions can slide over each other without breaking the metallic bonds, allowing metals to be bent and shaped.
### Concept 5: Carbon Allotropes
Carbon can form several different giant covalent structures, known as allotropes, each with unique properties due to their specific arrangement of atoms.
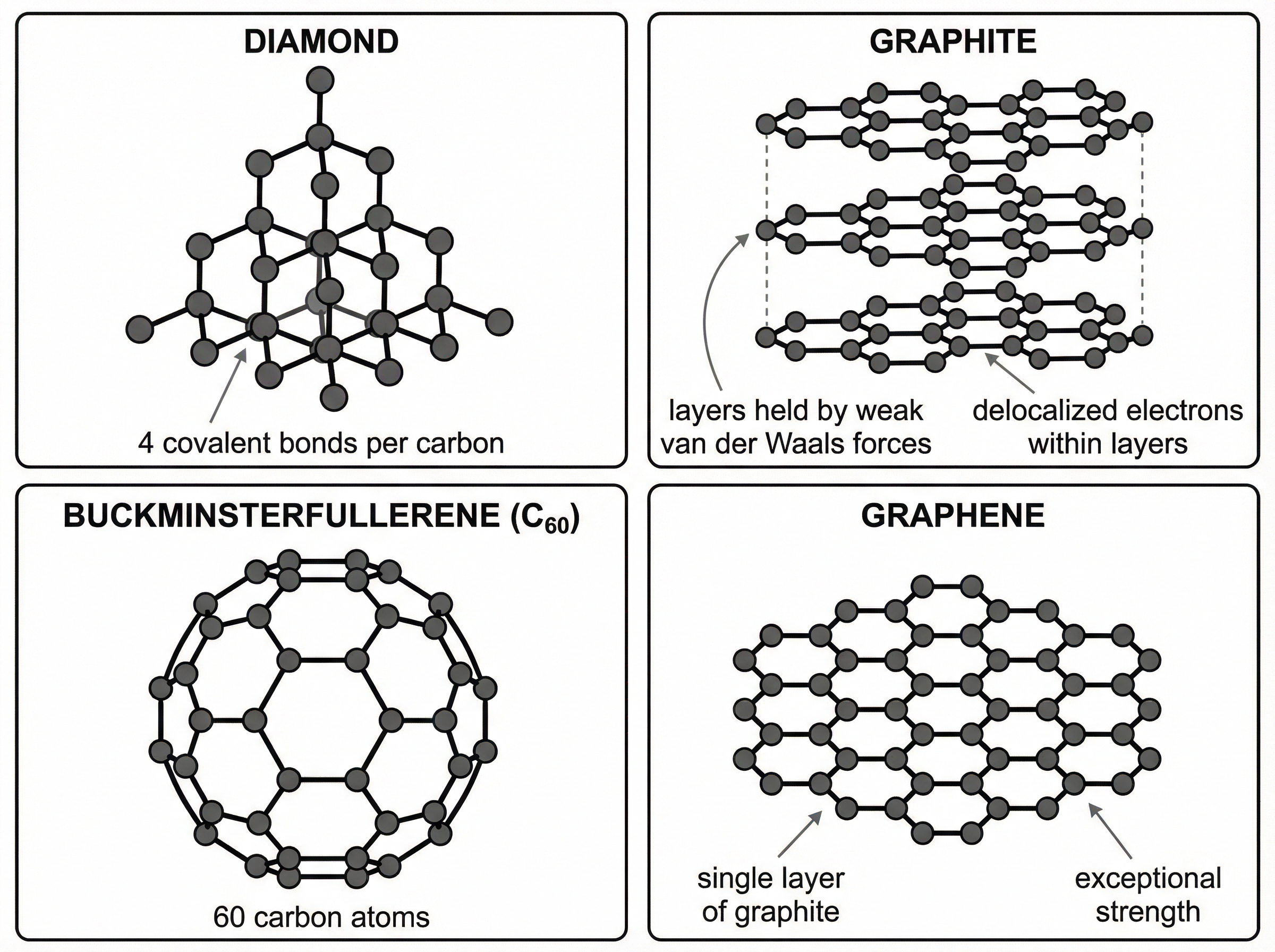
* **Diamond:** Each carbon atom forms four strong covalent bonds in a rigid tetrahedral structure. It is very hard, has a very high melting point, and does not conduct electricity.
* **Graphite:** Each carbon atom forms only three covalent bonds, creating layers of hexagonal rings. The fourth electron is delocalised, allowing graphite to conduct electricity and thermal energy. The layers are held together by weak intermolecular forces, so they can slide over each other, making graphite soft and slippery.
* **Graphene:** A single layer of graphite, just one atom thick. It is incredibly strong, transparent, and an excellent conductor of electricity.
* **Fullerenes:** Molecules of carbon atoms with hollow shapes, like spheres or tubes (nanotubes). Buckminsterfullerene ($C_{60}$) has a spherical shape. They are useful for drug delivery, as lubricants, and as catalysts.
### Concept 6: Nanoparticles
Nanoparticles are extremely small, with dimensions between 1 and 100 nanometres ($1 imes 10^{-9} m$ to $100 imes 10^{-9} m$). They contain only a few hundred atoms.
The crucial concept here is the **surface area to volume ratio**. As a particle decreases in size, its surface area to volume ratio increases significantly. This means a much greater proportion of the atoms are on the surface, making nanoparticles highly reactive compared to the bulk material.
**Applications and Risks:** Used in suncreams, cosmetics, electronics, and targeted drug delivery. However, their small size means they can enter the bloodstream and cells, and their long-term health and environmental impacts are not fully understood.
## Mathematical/Scientific Relationships
* **Standard Form:** You must be comfortable converting between normal numbers and standard form, especially for nanoparticles. E.g., $1 nm = 1 imes 10^{-9} m$.
* **Surface Area to Volume Ratio:**
$$ ext{Ratio} =
rac{ ext{Surface Area}}{ ext{Volume}}$$
*Calculate the surface area of a cube (6 × side²), then the volume (side³), and divide them.*
## Practical Applications
* **Alloys:** Pure metals are often too soft for everyday use because their regular layers slide easily. Alloys are mixtures of a metal with other elements (often different sized atoms). This distorts the regular layers, making it harder for them to slide, resulting in a harder, stronger material (e.g., steel, bronze).
* **Smart Materials:** Shape memory alloys (like Nitinol) can return to their original shape when heated, useful for dental braces and glasses frames.