Carbon compounds — WJEC GCSE Study Guide
Exam Board: WJEC | Level: GCSE
Master the chemistry of carbon compounds, from the fractional distillation of crude oil to the creation of complex polymers. This topic is heavily examined every year and forms the foundation for understanding fuels, plastics, and the petrochemical industry.

## Overview
This topic explores the fundamental chemistry of carbon compounds, focusing on crude oil as our primary source of hydrocarbons. It is a cornerstone of GCSE Chemistry because it links simple molecular structures to massive global industries: fuels and plastics. Examiners consistently test this topic to assess your ability to draw structures accurately, predict chemical reactions, and explain industrial processes like fractional distillation and cracking.
Understanding carbon chemistry is crucial because it connects directly to environmental topics (like combustion and climate change) and materials science (like polymerisation). Exam questions typically range from straightforward 1-mark recall of functional groups to complex 6-mark extended response questions requiring you to compare addition and condensation polymerisation or explain how boiling points change within a homologous series.
Listen to the full revision podcast here:

## Key Concepts
### Concept 1: Crude Oil and Fractional Distillation
Crude oil is a finite resource found in rocks, formed over millions of years from the remains of ancient biomass, mainly plankton. It is a complex mixture of hydrocarbons—compounds containing *only* carbon and hydrogen atoms. Because it's a mixture, the different hydrocarbons must be separated to be useful, a process achieved through **fractional distillation**.
The process works because different hydrocarbons have different boiling points. The crude oil is heated until it vaporises, and the vapour enters a fractionating column that is hot at the bottom and cooler at the top. As the vapours rise, they cool and condense back into liquids at their specific boiling points. **Crucially**, examiners want you to link molecular size to boiling point: larger molecules have stronger intermolecular forces, requiring more energy to overcome, hence higher boiling points. They condense lower down the column.
**Example**: Bitumen (used for roads) has very large molecules and condenses at the hot bottom of the column. Petrol (used for cars) has smaller molecules and condenses higher up where it is cooler.
### Concept 2: Cracking
Fractional distillation produces many large, heavy hydrocarbons that are not in high demand as fuels. However, there is a massive demand for smaller, lighter fractions like petrol. To solve this supply-and-demand issue, the petrochemical industry uses **cracking** to break down large alkane molecules into smaller, more useful alkanes and alkenes.
There are two main methods:
1. **Catalytic cracking**: Vaporise the heavy hydrocarbon and pass it over a hot zeolite catalyst.
2. **Steam (thermal) cracking**: Vaporise the heavy hydrocarbon, mix with steam, and heat to a very high temperature.
Cracking is a thermal decomposition reaction. It *always* produces at least one alkene because there are not enough hydrogen atoms to fully saturate all the new smaller molecules. This is vital because alkenes are the starting materials (monomers) for making plastics.
### Concept 3: The Homologous Series
A homologous series is a 'family' of organic compounds that have the same general formula, the same functional group, and similar chemical properties. Each successive member differs by a $-CH_2-$ unit. You must know four series:
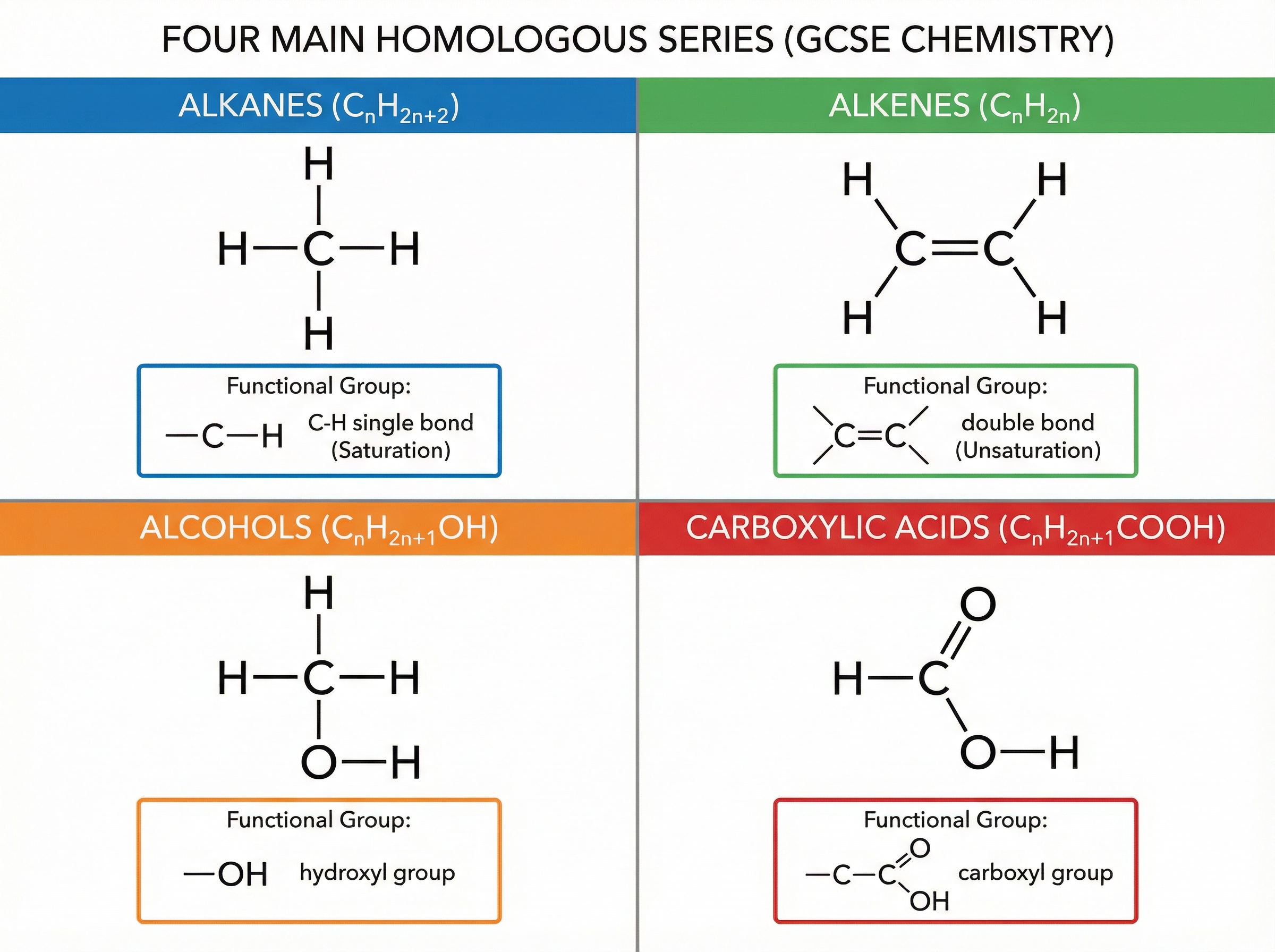
1. **Alkanes**: Saturated hydrocarbons (only single bonds). General formula $C_n H_{2n+2}$. They are generally unreactive but combust well.
2. **Alkenes**: Unsaturated hydrocarbons containing a $C=C$ double bond. General formula $C_n H_{2n}$. They are more reactive and will decolourise orange bromine water.
3. **Alcohols**: Contain the hydroxyl functional group ($-OH$). General formula $C_n H_{2n+1}OH$. They combust, react with sodium to produce hydrogen, and can be oxidised to carboxylic acids.
4. **Carboxylic Acids**: Contain the carboxyl functional group ($-COOH$). General formula $C_n H_{2n+1}COOH$. They are weak acids that partially ionise in water.
### Concept 4: Polymerisation
Polymerisation is the joining together of many small molecules (monomers) to form a very large molecule (polymer).
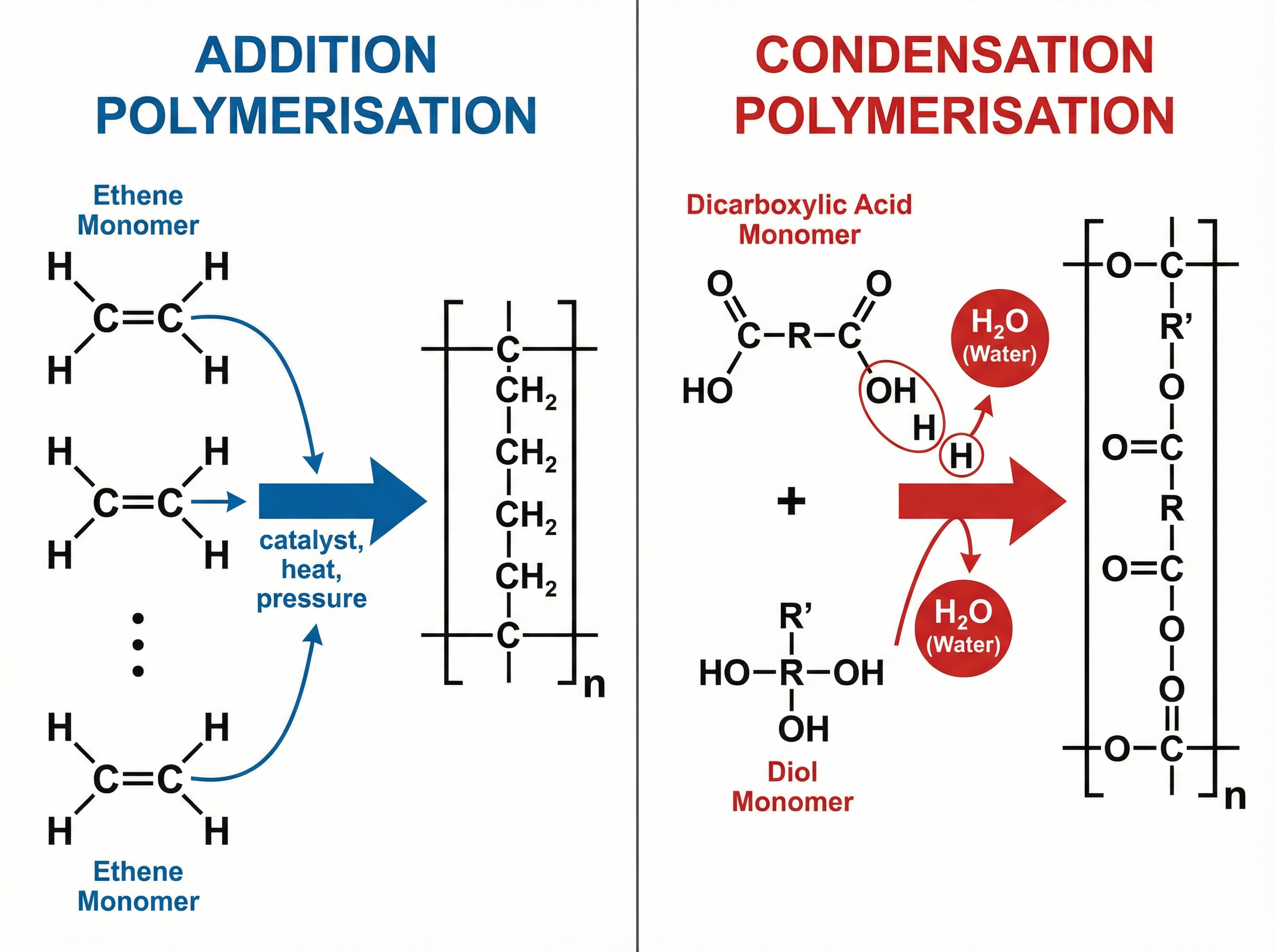
**Addition Polymerisation**: Involves monomers with a $C=C$ double bond (alkenes). The double bond opens up to connect to neighbouring molecules. *No other product is formed*. For example, ethene monomers join to form poly(ethene).
**Condensation Polymerisation**: Involves monomers with two functional groups. When these monomers react and join, they lose small molecules such as water ($H_2O$) or hydrogen chloride ($HCl$). For example, a dicarboxylic acid reacts with a diol to form a polyester and water.
## Mathematical/Scientific Relationships
- **Alkane General Formula**: $C_n H_{2n+2}$
- **Alkene General Formula**: $C_n H_{2n}$
- **Alcohol General Formula**: $C_n H_{2n+1}OH$
- **Carboxylic Acid General Formula**: $C_n H_{2n+1}COOH$
*Note: In these formulas, 'n' represents the number of carbon atoms. You must memorise these formulas as they are not provided in the exam.*
## Practical Applications
- **Combustion of Fuels**: Alkanes are our primary fuels. Complete combustion (plenty of oxygen) produces $CO_2$ and $H_2O$. Incomplete combustion (limited oxygen) produces carbon monoxide ($CO$) or carbon soot ($C$), which has serious health and environmental impacts.
- **Testing for Alkenes**: The bromine water test is a standard chemical test. Adding orange bromine water to an alkene causes an addition reaction across the double bond, turning the solution colourless. Alkanes will not react, and the solution remains orange.