Chemical formulae, equations and amount of substance — WJEC GCSE Study Guide
Exam Board: WJEC | Level: GCSE
Mastering chemical formulae and the mole concept is the key to unlocking quantitative chemistry. This topic bridges the atomic world with the macroscopic world, forming the foundation for calculating reacting masses, gas volumes, and concentrations.
## Overview

Chemical formulae, equations, and amount of substance form the mathematical backbone of GCSE Chemistry. This topic is fundamentally about counting atoms and molecules—not individually, but in vast, measurable quantities called moles. Understanding these concepts is critical because they allow chemists to predict exactly how much product a reaction will yield or how much reactant is needed, which is essential in everything from industrial manufacturing to pharmacology.
Examiners consistently test this area across multiple papers. You will encounter straightforward 1-2 mark questions on balancing equations and calculating relative formula mass, building up to challenging 4-6 mark synoptic questions involving limiting reactants, empirical formulae, and gas volumes. Mastering the mole concept here will pay dividends when you study titrations, electrolysis, and energy changes.

## Key Concepts
### Concept 1: Chemical Formulae and Relative Formula Mass (Mr)
A chemical formula tells you precisely which elements are in a compound and their ratio. For instance, $H_2SO_4$ contains two hydrogen atoms, one sulfur atom, and four oxygen atoms per molecule. The small subscript numbers are fixed—changing them changes the substance entirely.
The **Relative Formula Mass ($M_r$)** is the sum of the relative atomic masses ($A_r$) of all atoms in the formula. Examiners will provide a periodic table, so you simply need to identify the $A_r$ values and add them up.
*Why does this work?* Because atoms of different elements have different masses, comparing their masses directly requires a standard reference (Carbon-12). The $M_r$ gives us a single value that represents the mass of one 'unit' of that substance relative to others.
**Example**: Calculate the $M_r$ of Calcium Carbonate ($CaCO_3$).
$A_r$ values: $Ca = 40, C = 12, O = 16$
$M_r = 40 + 12 + (3 \times 16) = 40 + 12 + 48 = 100$
### Concept 2: Balancing Chemical Equations
The Law of Conservation of Mass dictates that no atoms are lost or made during a chemical reaction. Therefore, the total mass of the products must equal the total mass of the reactants. To represent this, chemical equations must be balanced by placing large numbers (coefficients) in front of the formulae.
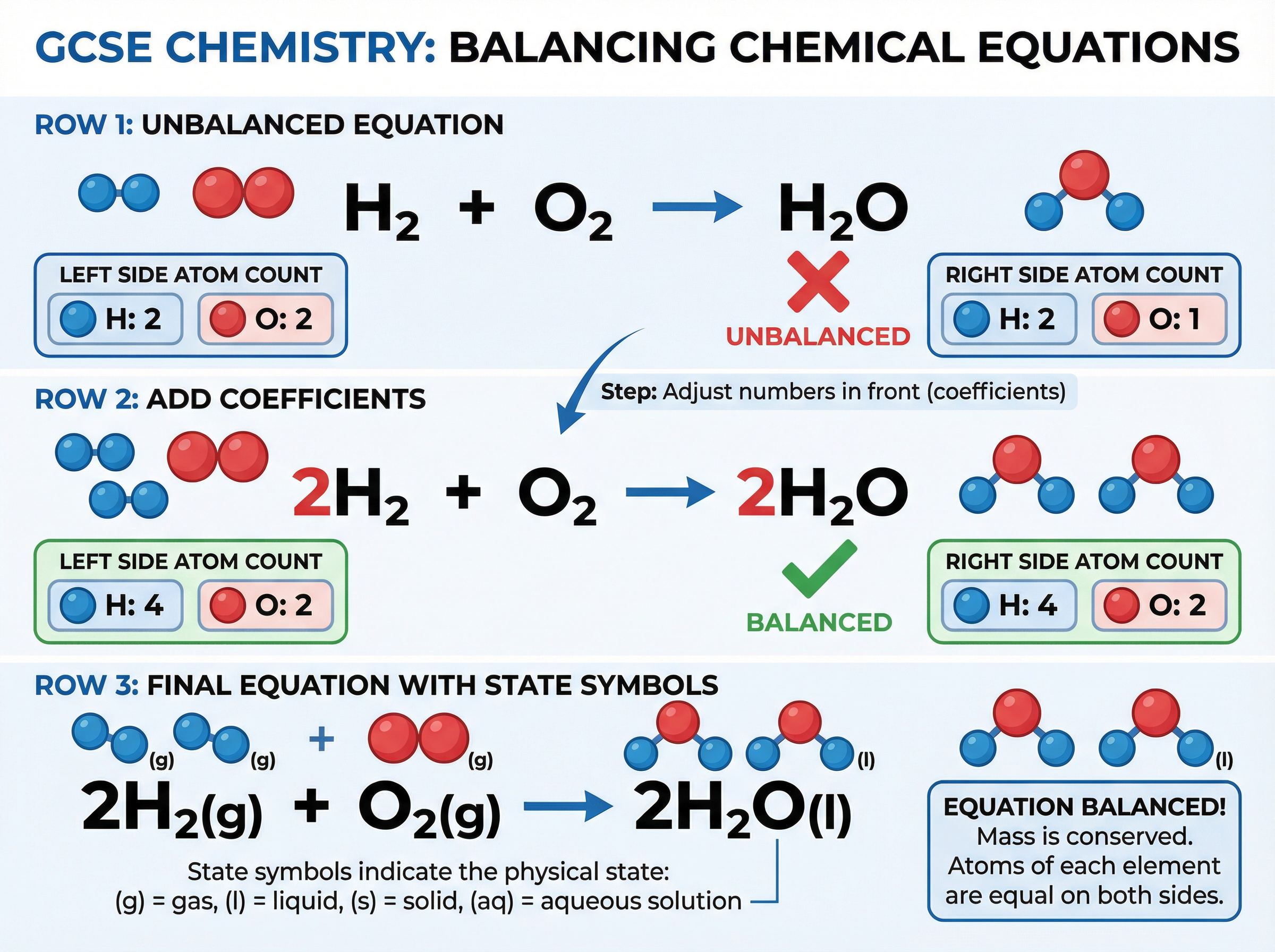
*Why does this work?* A chemical reaction is simply the rearrangement of existing atoms. If you start with 4 hydrogen atoms, you must end with 4 hydrogen atoms. Examiners will penalize you if you alter the small subscript numbers, as this indicates a fundamental misunderstanding of chemical compounds.
### Concept 3: The Mole and Avogadro's Constant
The 'mole' is the standard unit for the amount of substance. One mole of any substance contains exactly $6.02 \times 10^{23}$ particles (atoms, molecules, or ions). This specific number is known as the **Avogadro constant**.
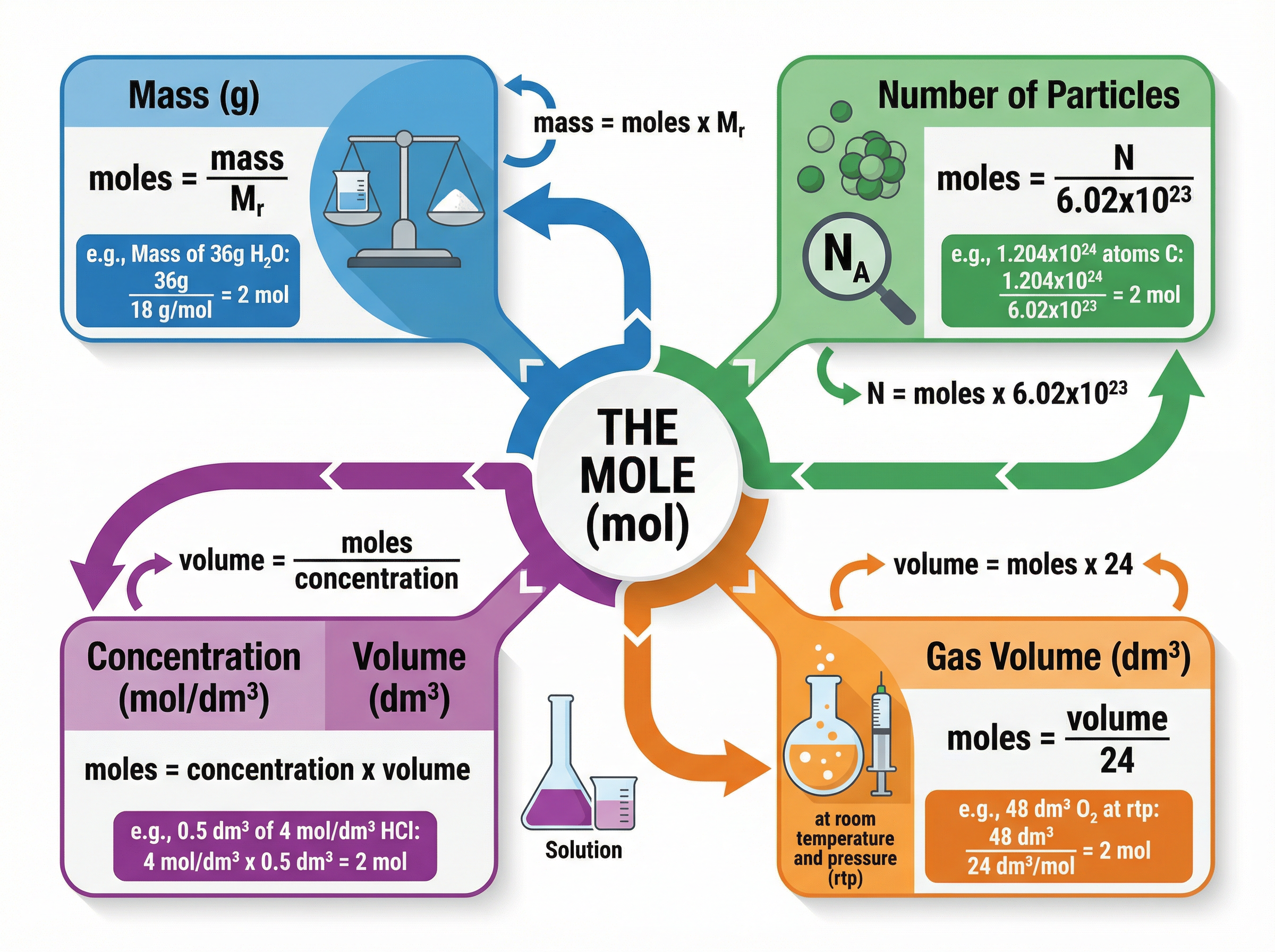
The genius of the mole concept is this: the mass of one mole of a substance in grams is numerically equal to its relative formula mass ($M_r$). Therefore, 1 mole of carbon ($A_r = 12$) has a mass of 12g, and 1 mole of water ($M_r = 18$) has a mass of 18g.
### Concept 4: Reacting Masses and Stoichiometry
Using balanced equations and the mole concept, we can calculate the exact masses of reactants needed or products formed. The coefficients in a balanced equation represent the molar ratio of the substances involved.
If the equation is $2Mg + O_2 \rightarrow 2MgO$, it tells us that 2 moles of magnesium react with 1 mole of oxygen to produce 2 moles of magnesium oxide.
### Concept 5: Limiting Reactants (Higher Tier)
In many reactions, one reactant is completely used up while the other is in excess. The reactant that is completely consumed is the **limiting reactant** because it determines the maximum amount of product that can be formed.
*Why does this happen?* Think of making bicycles: if you have 10 frames and 16 wheels, you can only make 8 bicycles. The wheels are the limiting factor, and you will have 2 frames left over in excess.
### Concept 6: Molar Gas Volume
At room temperature and pressure (rtp: 20°C and 1 atmosphere), equal volumes of different gases contain the same number of molecules. Specifically, one mole of *any* gas occupies a volume of $24 dm^3$ (or $24,000 cm^3$). This allows for quick conversions between the volume of a gas and the number of moles.
## Mathematical/Scientific Relationships
To succeed in this topic, you must memorize and confidently apply these core formulas:
1. **Moles from Mass**: $Moles = \frac{Mass (g)}{M_r}$
*(Must memorise)*
2. **Number of Particles**: $Particles = Moles \times (6.02 \times 10^{23})$
*(Avogadro constant given, but relationship must be memorised)*
3. **Moles of Gas at rtp**: $Moles = \frac{Volume (dm^3)}{24}$
*(Must memorise. Note: if volume is in $cm^3$, divide by 24,000 instead)*
4. **Concentration (mol/dm³)**: $Concentration = \frac{Moles}{Volume (dm^3)}$
*(Must memorise)*
## Practical Applications
While this topic is heavily theoretical, it is applied in almost every chemical practical. When preparing a standard solution for a titration, you must calculate the exact mass of solid required to achieve a specific concentration. In industrial chemistry, calculating reacting masses ensures that expensive reactants are not wasted and that the theoretical yield is known, which is crucial for determining the efficiency (percentage yield) of a manufacturing process.