Chemistry of acids — WJEC GCSE Study Guide
Exam Board: WJEC | Level: GCSE
Master the fundamental reactions, pH scale, and titration calculations that examiners test every year. This topic forms the backbone of quantitative chemistry and is essential for securing top grades in your GCSE.
## Overview
The Chemistry of Acids is a cornerstone topic in GCSE Chemistry. It bridges the gap between qualitative observations (what happens when things react) and quantitative analysis (exactly how much of each substance is reacting). Examiners love this topic because it allows them to test multiple Assessment Objectives simultaneously — from recalling basic definitions (AO1) to applying knowledge to novel contexts (AO2) and analyzing experimental data (AO3).
Understanding acids is crucial because these reactions form the basis for many industrial processes, from manufacturing fertilizers to treating wastewater. In your exam, you can expect questions ranging from simple 1-mark word equations to complex 6-mark titration calculations or experimental method descriptions. By mastering the core principles — the definition of an acid, the four key reactions, and the concept of neutralisation — you'll be well-prepared to tackle even the most challenging synoptic questions.
Listen to the companion podcast to reinforce your learning:

## Key Concepts
### Concept 1: Defining Acids and Alkalis
In aqueous solution, acids are substances that produce hydrogen ions (H⁺). This is the precise definition examiners are looking for. Conversely, alkalis are soluble bases that produce hydroxide ions (OH⁻) in aqueous solution.
The pH scale is a measure of the acidity or alkalinity of a solution, running from 0 to 14. A pH of 7 is neutral (e.g., pure water). Solutions with a pH less than 7 are acidic, while those with a pH greater than 7 are alkaline.
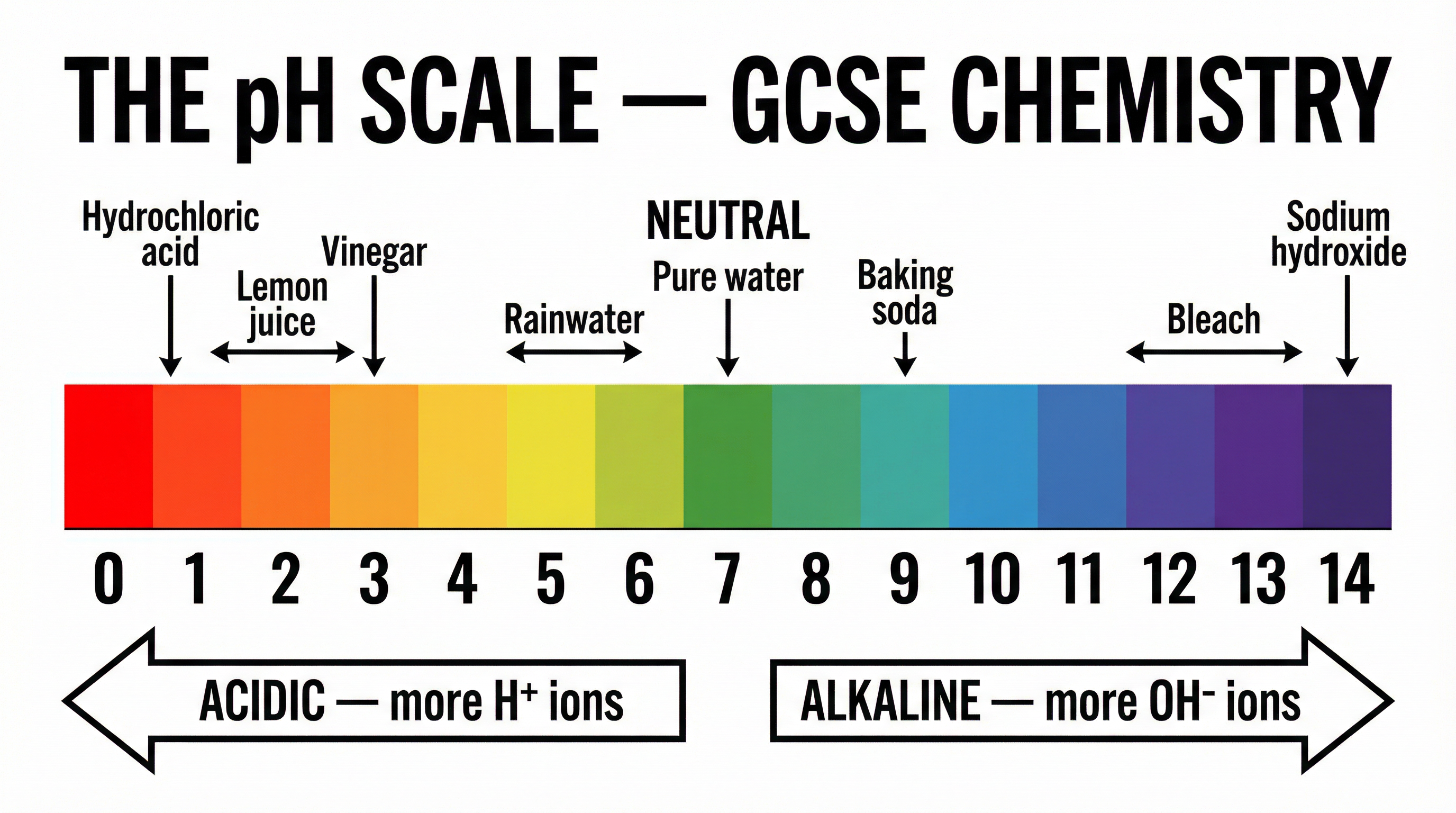
**Crucial Higher Tier Concept:** The pH scale is logarithmic. This means that a decrease of one pH unit corresponds to a tenfold increase in the concentration of hydrogen ions.
**Example**: If a solution of hydrochloric acid has a pH of 1, and it is diluted so the pH becomes 2, the concentration of H⁺ ions has decreased by a factor of 10. If the pH changes from 1 to 3, the concentration decreases by a factor of 100 (10²).
### Concept 2: Strong vs. Weak Acids
A common pitfall is confusing 'strong' with 'concentrated'. They mean entirely different things in chemistry:
* **Strong acids** (e.g., hydrochloric, nitric, sulfuric acid) completely ionise in aqueous solution. Every molecule dissociates to release an H⁺ ion.
* **Weak acids** (e.g., ethanoic, citric, carbonic acid) only partially ionise in aqueous solution. Only a small fraction of the molecules dissociate.
Concentration, on the other hand, refers to the amount of substance (in moles) dissolved in a given volume of solution (usually 1 dm³). Therefore, you can have a dilute solution of a strong acid, or a concentrated solution of a weak acid.
### Concept 3: The Four Key Acid Reactions
You must be able to recall the products of these four fundamental reactions and write balanced symbol equations for them.
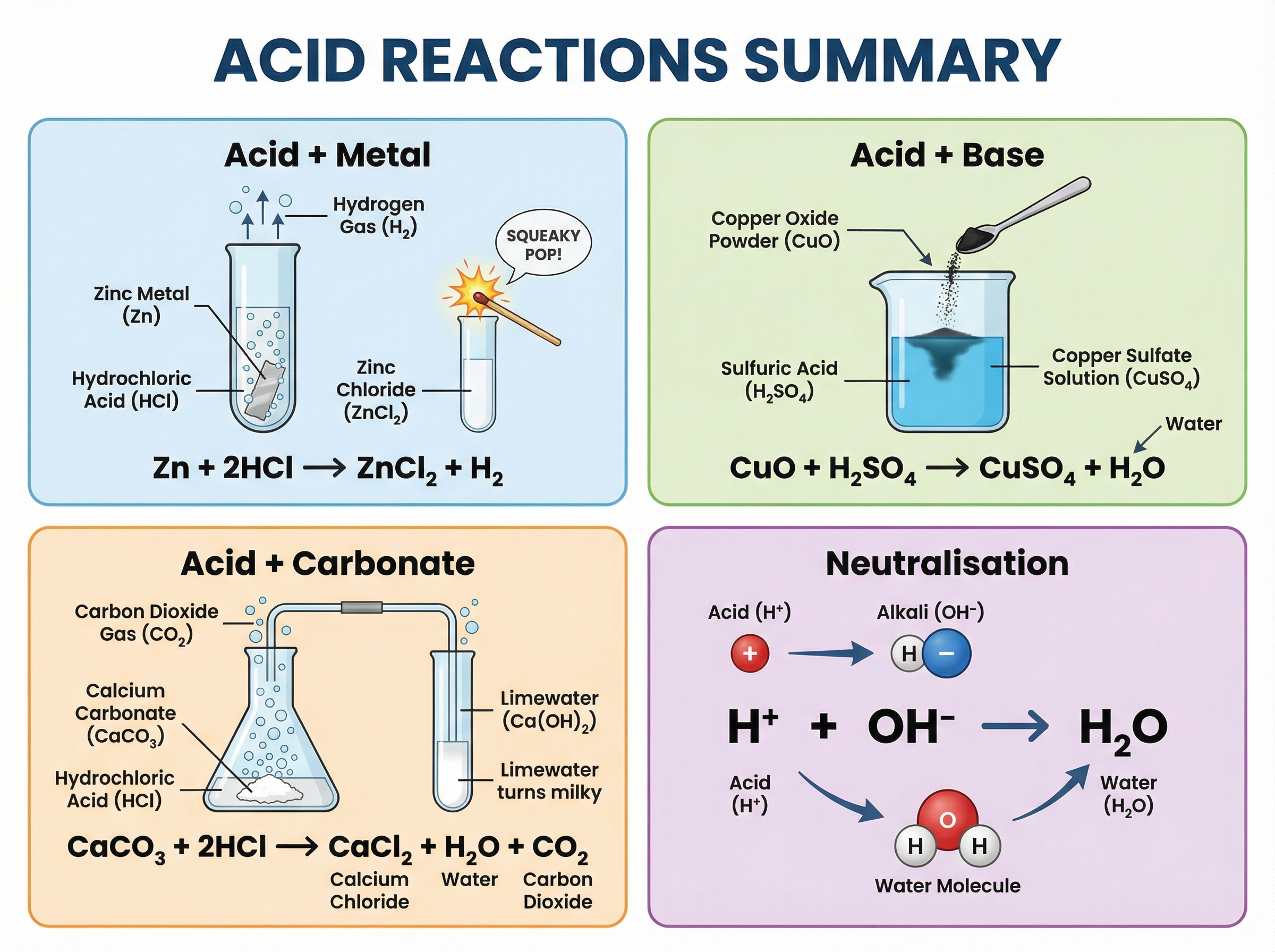
1. **Acid + Metal → Salt + Hydrogen**
* *Example*: 2HCl(aq) + Mg(s) → MgCl₂(aq) + H₂(g)
* *Observation*: Effervescence (bubbling). The hydrogen gas produced can be tested using a lit splint, which results in a 'squeaky pop'.
2. **Acid + Base → Salt + Water** (Neutralisation)
* *Example*: H₂SO₄(aq) + CuO(s) → CuSO₄(aq) + H₂O(l)
* *Note*: Bases are typically metal oxides or metal hydroxides.
3. **Acid + Alkali → Salt + Water** (Neutralisation)
* *Example*: HCl(aq) + NaOH(aq) → NaCl(aq) + H₂O(l)
* *Ionic Equation*: The fundamental ionic equation for all neutralisation reactions is: **H⁺(aq) + OH⁻(aq) → H₂O(l)**. Memorise this.
4. **Acid + Metal Carbonate → Salt + Water + Carbon Dioxide**
* *Example*: 2HNO₃(aq) + CaCO₃(s) → Ca(NO₃)₂(aq) + H₂O(l) + CO₂(g)
* *Observation*: Effervescence. The carbon dioxide produced turns limewater milky/cloudy.
### Concept 4: Titrations (Required Practical)
Titrations are used to accurately determine the concentration of an unknown acid or alkali by reacting it with a solution of known concentration.
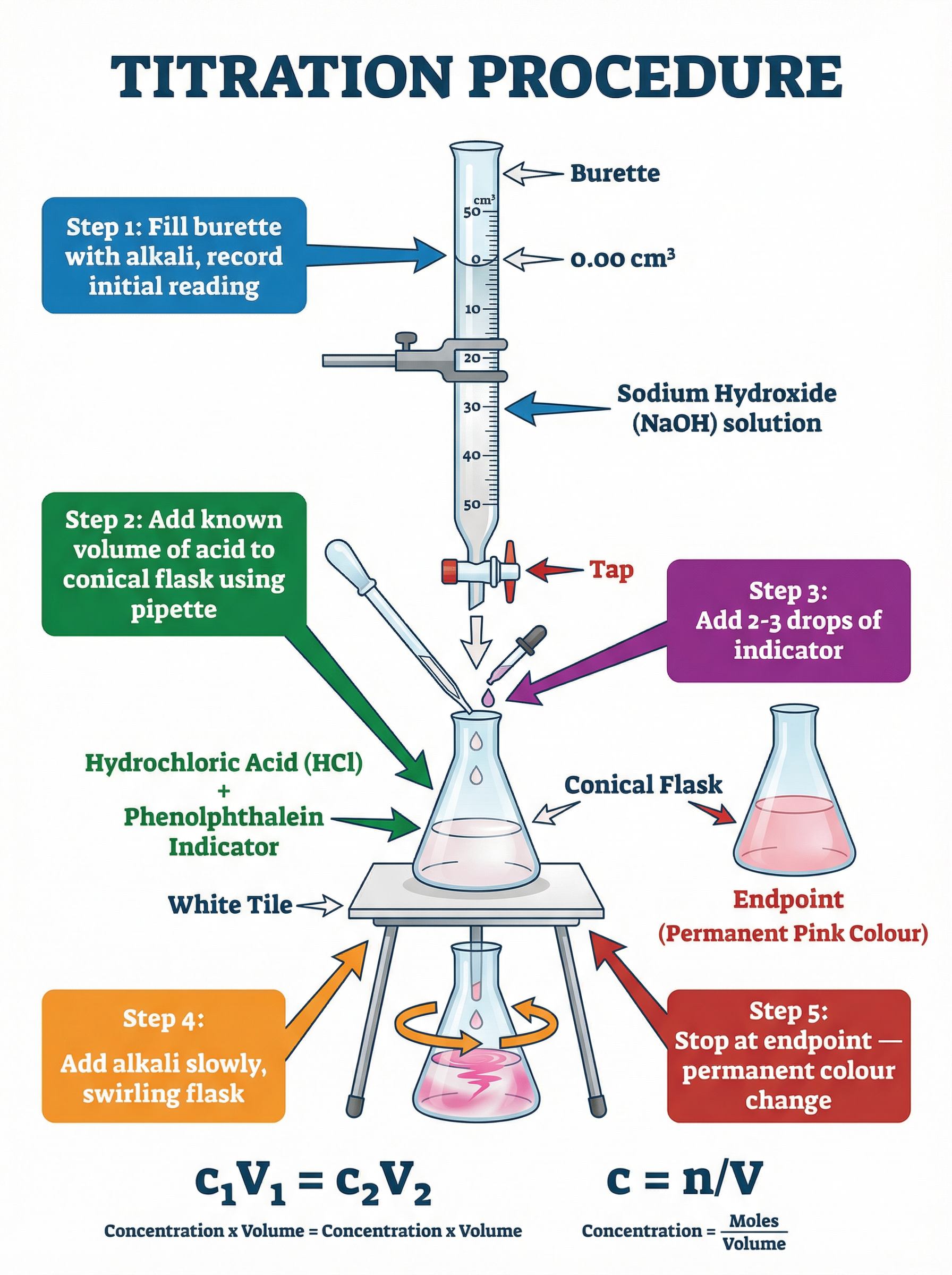
**The Method:**
1. Use a pipette to transfer a known volume (e.g., 25.0 cm³) of the unknown alkali into a conical flask.
2. Add a few drops of a suitable indicator (e.g., phenolphthalein or methyl orange).
3. Fill a burette with the acid of known concentration and record the initial reading.
4. Slowly add the acid from the burette to the conical flask while swirling.
5. Stop adding acid when the indicator permanently changes colour (the endpoint).
6. Record the final burette reading and calculate the titre (volume of acid added).
7. Repeat the titration until you obtain concordant results (within 0.10 cm³ of each other).
## Mathematical/Scientific Relationships
* **Concentration (c) = Moles (n) / Volume (V)**
* *c* is concentration in mol/dm³
* *n* is amount of substance in moles (mol)
* *V* is volume in cubic decimetres (dm³)
* *Must memorise*: Yes.
* *Crucial Conversion*: To convert cm³ to dm³, you **must divide by 1000**. (e.g., 25 cm³ = 0.025 dm³).
* **Mass (m) = Moles (n) × Molar Mass (Mr)**
* Used to convert between moles and mass if the question asks for concentration in g/dm³.
## Practical Applications
**Preparing a Soluble Salt (Required Practical)**
To prepare a pure, dry sample of a soluble salt (like copper sulfate) from an insoluble base (copper oxide):
1. Warm the dilute acid (sulfuric acid) gently using a Bunsen burner.
2. Add the insoluble base (copper oxide) to the acid a little at a time, stirring constantly, until it is in excess (some unreacted solid remains at the bottom). This ensures all the acid has reacted.
3. Filter the mixture using filter paper and a funnel to remove the excess unreacted base. The filtrate is the salt solution.
4. Pour the filtrate into an evaporating basin and heat it gently over a water bath to evaporate some of the water until crystals start to form (the crystallisation point).
5. Leave the basin in a cool place for the remaining water to evaporate slowly, allowing large crystals to form.
6. Pat the crystals dry with filter paper.