Energy changes in chemistry — WJEC GCSE Study Guide
Exam Board: WJEC | Level: GCSE
Master the core concepts of Energy Changes in Chemistry, from identifying exothermic and endothermic reactions to mastering reaction profiles and calculating bond energies. This topic is essential for your GCSE exams, combining conceptual understanding with vital mathematical skills.
## Overview
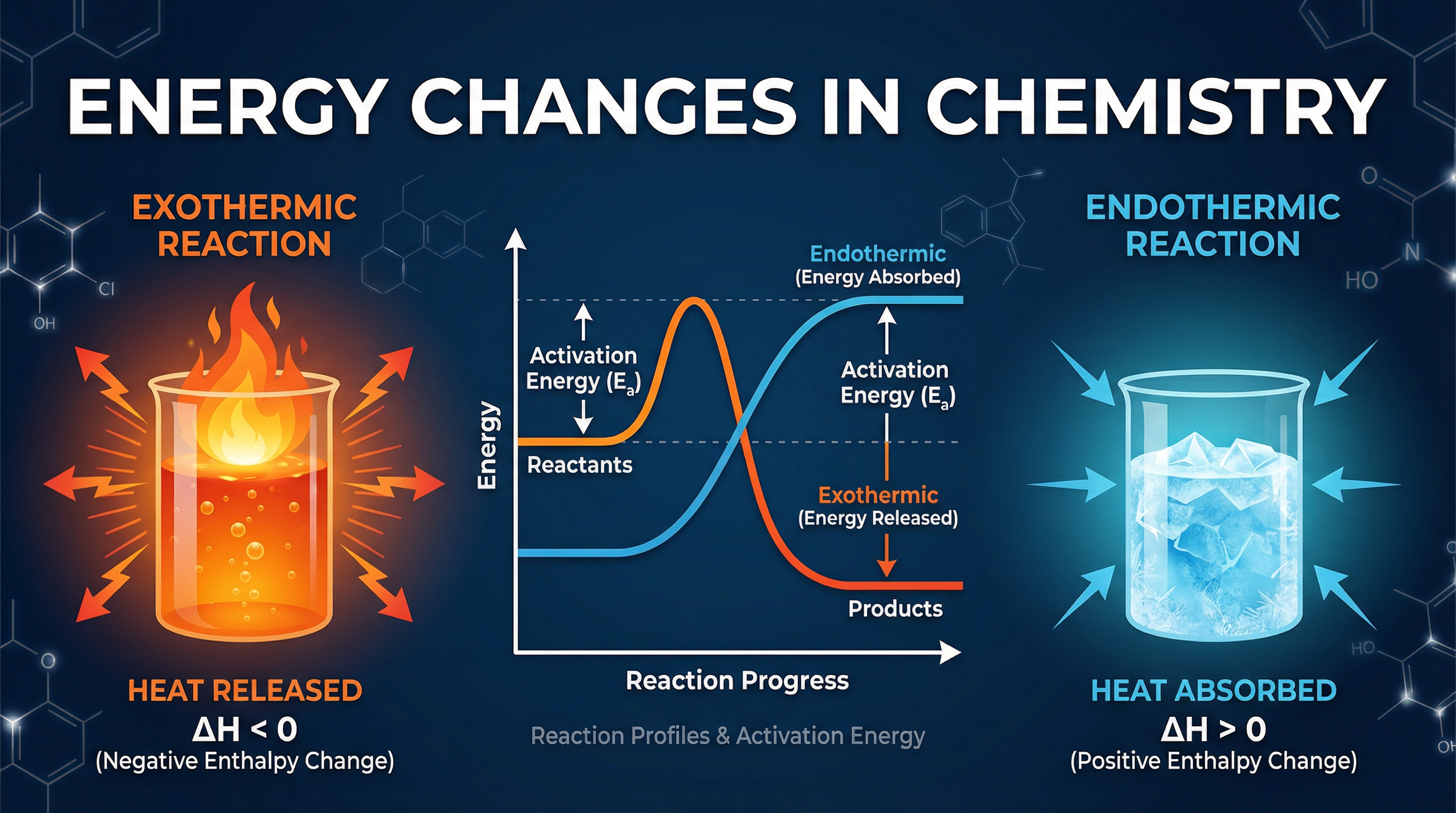
Energy Changes in Chemistry is a fundamental topic that explores the relationship between chemical reactions and the transfer of energy. Every chemical reaction involves energy: breaking bonds in reactants requires an input of energy, while forming new bonds in products releases energy. Understanding which of these processes dominates allows us to classify reactions as either **exothermic** or **endothermic**.
This topic is crucial because it bridges theoretical chemistry with real-world applications, from designing self-heating cans to developing clean energy solutions like hydrogen fuel cells. Examiners frequently test this area through a mix of qualitative descriptions, graphical interpretations (reaction profiles), and quantitative calculations (bond energies). Mastering this topic will secure you significant marks across multiple assessment objectives.
Listen to our comprehensive revision podcast for a deep dive into these concepts:

## Key Concepts
### Concept 1: Exothermic and Endothermic Reactions
In chemistry, we must always consider the direction of energy transfer between the chemical **system** (the reactants and products) and the **surroundings** (everything else, including the thermometer and the beaker).
An **exothermic** reaction is one where energy is transferred *from* the system *to* the surroundings. Because the surroundings gain energy, their temperature **increases**. This occurs because more energy is released when new bonds form in the products than is required to break the bonds in the reactants.
**Example**: Combustion, neutralisation, and oxidation reactions are typically exothermic. Hand warmers use the exothermic oxidation of iron to release heat.
An **endothermic** reaction is one where energy is taken *in* from the surroundings *to* the system. Because the surroundings lose energy, their temperature **decreases**. This occurs because more energy is required to break the bonds in the reactants than is released when new bonds form in the products.
**Example**: Thermal decomposition and the reaction of citric acid and sodium hydrogencarbonate are endothermic. Sports injury cold packs use an endothermic reaction to cool down quickly.
### Concept 2: Reaction Profiles
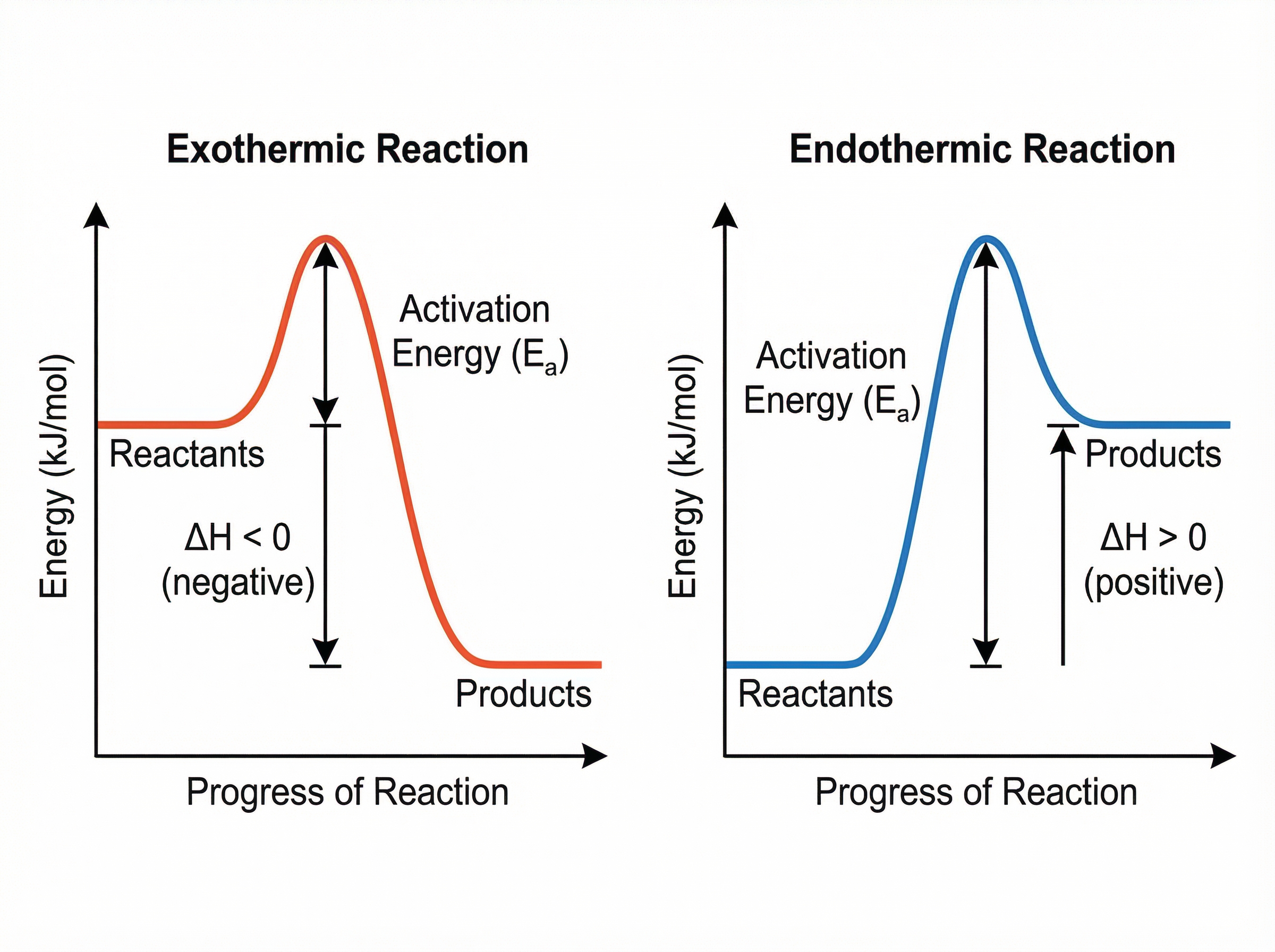
Reaction profiles (or energy level diagrams) are graphical representations of the energy changes during a chemical reaction. They show the relative energies of reactants and products, the activation energy, and the overall energy change.
- **Activation Energy ($E_a$)**: The minimum amount of energy that colliding particles must possess for a reaction to occur. On a profile, it is always an upward arrow from the reactants' energy level to the peak of the curve.
- **Overall Energy Change ($\Delta H$)**: The difference in energy between the reactants and the products.
For an **exothermic** profile, the products are at a *lower* energy level than the reactants. The $\Delta H$ arrow points downwards, indicating a negative energy change.
For an **endothermic** profile, the products are at a *higher* energy level than the reactants. The $\Delta H$ arrow points upwards, indicating a positive energy change.
### Concept 3: Bond Energy Calculations
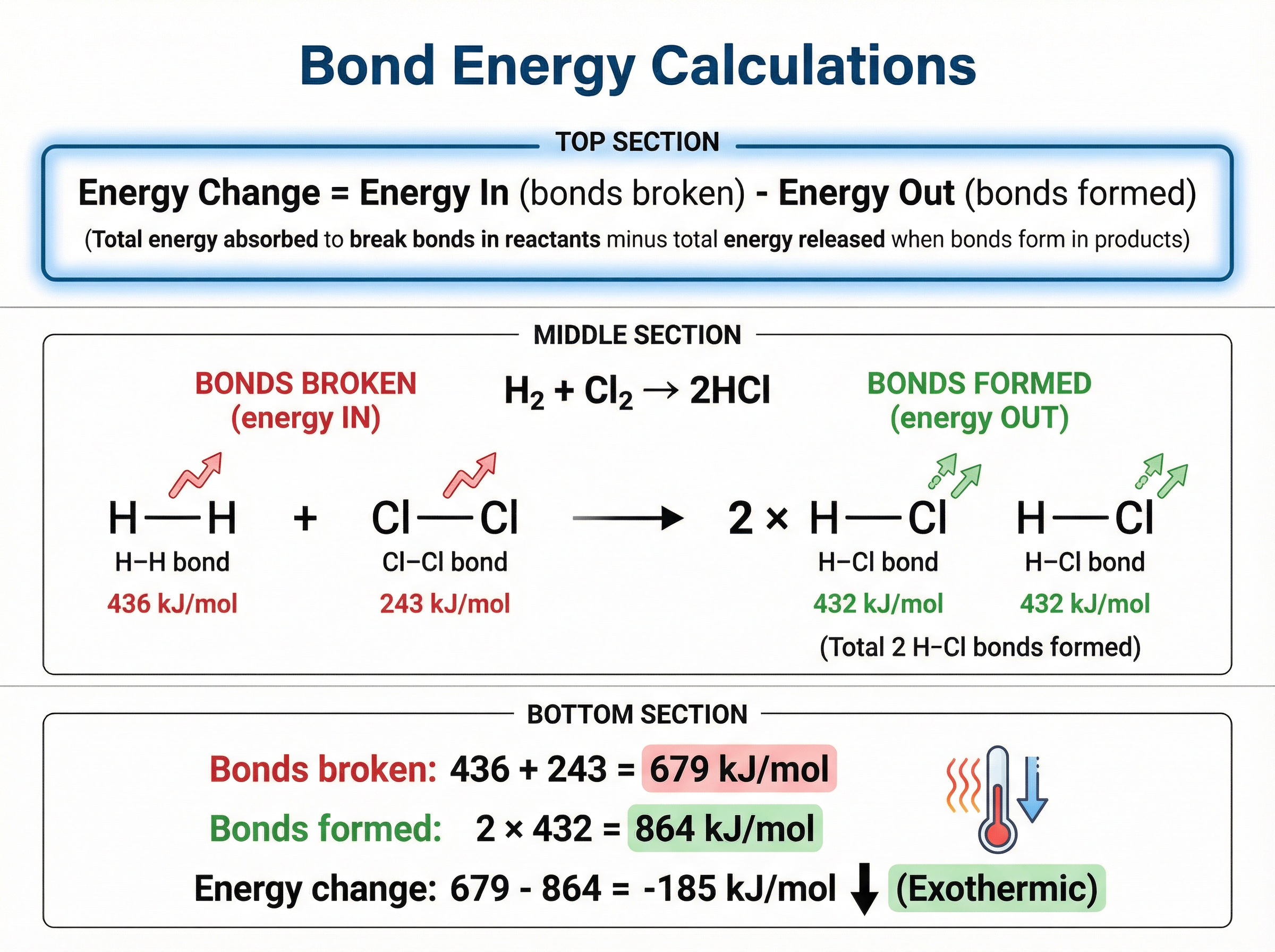
During a chemical reaction, old bonds are broken and new bonds are formed. We can calculate the overall energy change if we know the specific **bond energies** (the energy required to break one mole of a particular bond, measured in kJ/mol).
1. **Energy In**: Calculate the total energy required to break all bonds in the reactants. (Endothermic process)
2. **Energy Out**: Calculate the total energy released when all bonds in the products are formed. (Exothermic process)
3. **Overall Change**: Subtract 'Energy Out' from 'Energy In'.
If the result is negative, the reaction is exothermic. If positive, it is endothermic.
### Concept 4: Chemical Cells and Fuel Cells
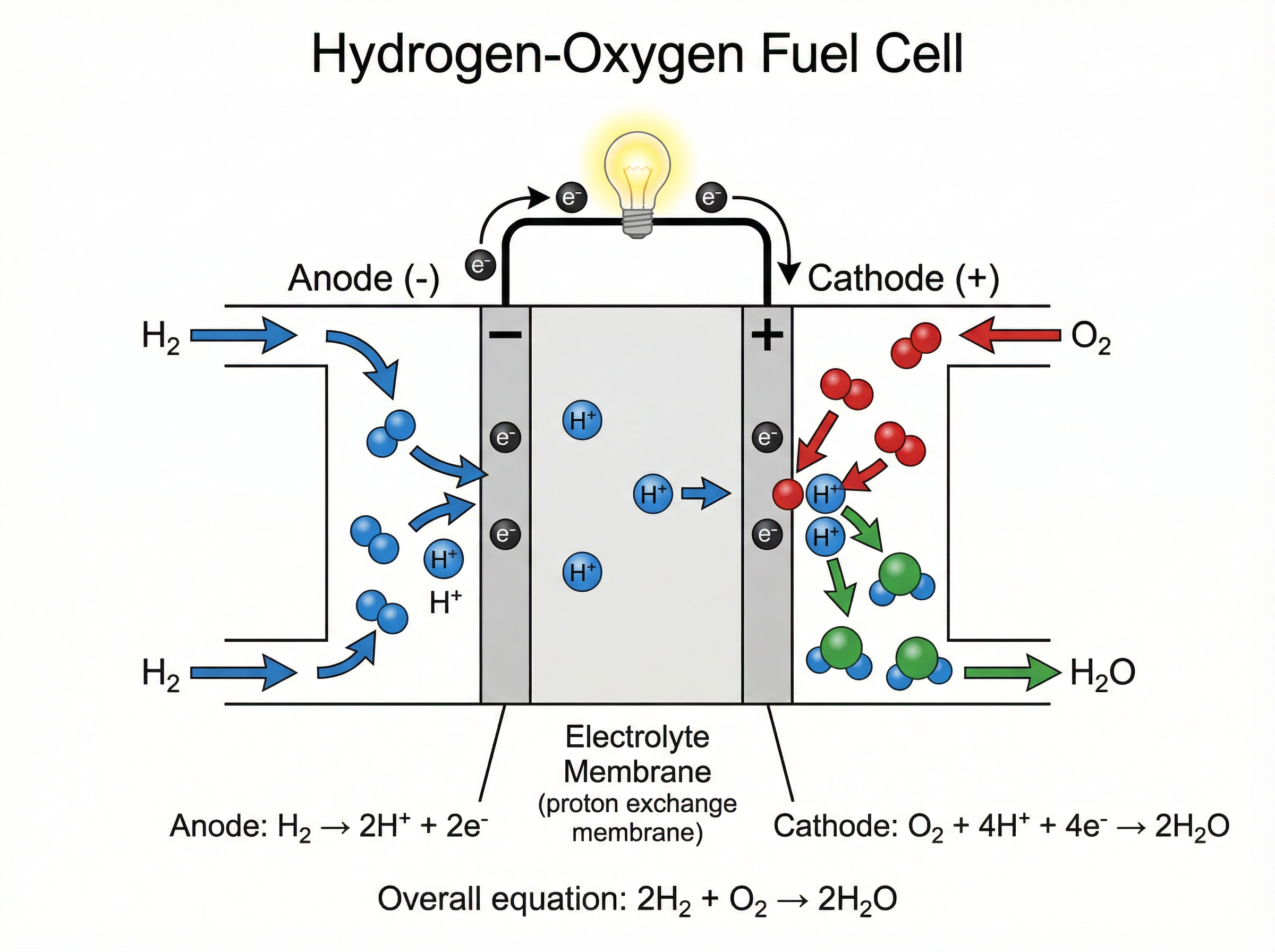
**Chemical Cells**: A simple cell can be made by connecting two different metals in contact with an electrolyte. The difference in reactivity between the metals generates a potential difference (voltage). However, these cells are non-rechargeable; once one of the reactants is depleted, the reaction stops.
**Fuel Cells**: A fuel cell produces a continuous voltage as long as it is supplied with an external fuel (like hydrogen) and oxygen. In a hydrogen-oxygen fuel cell, hydrogen is oxidised at the anode, and oxygen is reduced at the cathode. The only product is water, making it a clean energy alternative to combustion engines. However, the hydrogen fuel is often produced from fossil fuels, and it is difficult to store and transport.
## Mathematical/Scientific Relationships
**Overall Energy Change = Total Energy to Break Bonds - Total Energy Released Forming Bonds**
- **Symbolic Form**: $\Delta H = \Sigma \text{Bonds Broken} - \Sigma \text{Bonds Formed}$
- **Units**: kJ/mol (kilojoules per mole)
- **Must memorise**: Yes. This formula is not typically provided on the data sheet.
**Overall Equation for Hydrogen Fuel Cell**:
$2H_2 + O_2 \rightarrow 2H_2O$
**Half-Equations for Hydrogen Fuel Cell**:
- **Anode (Oxidation)**: $H_2 \rightarrow 2H^+ + 2e^-$
- **Cathode (Reduction)**: $O_2 + 4H^+ + 4e^- \rightarrow 2H_2O$
## Practical Applications
**Required Practical: Investigating Temperature Changes**
Students are required to investigate the variables that affect temperature changes in reacting solutions, such as the reaction between an acid and an alkali (neutralisation).
- **Apparatus**: Polystyrene cup (acts as an insulator to reduce heat loss to the surroundings), beaker (for stability), thermometer with a lid (to prevent heat loss via convection).
- **Method**: Measure a set volume of acid into the cup. Record the initial temperature. Add a set volume/mass of the second reactant (e.g., alkali or metal). Stir and record the maximum (or minimum) temperature reached. Calculate the temperature change.
- **Common Errors**: The biggest source of error is heat loss to the surroundings. Using a polystyrene cup with a lid minimizes this, making the maximum temperature recorded more accurate.
- **Examiner Focus**: Examiners often ask why a polystyrene cup is used instead of a glass beaker, or ask candidates to plot the temperature changes on a graph and extrapolate lines to find the theoretical maximum temperature change.